Clear Sky Science · en
Hetero[3.1.1]propellanes
A New Shape for Future Medicines
Most pills on the market are built from flat, ring-shaped pieces of carbon known as benzene rings. These trusty components work well, but they can sometimes make drugs too greasy, poorly soluble, or prone to unwanted side effects. This paper introduces an entirely new family of tiny three‑dimensional building blocks—called hetero[3.1.1]propellanes—that chemists can use to design medicines with more favorable properties. 
Why Chemists Want to Escape Flat Designs
Drug molecules must fit snugly into the complex three‑dimensional shapes of proteins in the body. Flat aromatic rings, like benzene, often limit how precisely a molecule can be positioned in space and can increase its tendency to dissolve in fats rather than in water. In recent years, chemists have turned to compact, cage‑like carbon frameworks that mimic the geometry of benzene but are more three‑dimensional and less oily. Two such shapes, called bicyclo[1.1.1]pentanes and bicyclo[3.1.1]heptanes, can stand in for para‑ and meta‑substituted benzene rings in drugs. However, the purely carbon version of bicyclo[3.1.1]heptane remains relatively greasy, limiting the performance gains it offers.
Adding Heteroatoms to Tune Drug‑Like Behavior
The authors propose a simple idea with big consequences: replace one carbon atom in the bicyclo[3.1.1]heptane framework with a different element such as oxygen, nitrogen, or sulfur. These “heteroatoms” can make molecules less lipophilic, more water‑soluble, and easier for the body to process, while preserving the crucial three‑dimensional geometry that helps a drug engage its target. Yet, despite decades of study on related carbon frameworks, no one had succeeded in making the corresponding small, highly strained precursors known as hetero[3.1.1]propellanes, which are the ideal starting points for building a wide variety of such heterocycles.
Building a New Family of Molecular Cages
The team at Oxford and AbbVie developed a unified, scalable route to three members of this new family: oxygen‑, sulfur‑, and nitrogen‑containing [3.1.1]propellanes. Their strategy starts from a simple commercial molecule, 2,3‑dibromopropene, which undergoes a highly efficient reaction with a diazo compound under rhodium catalysis to form a key three‑membered ring on multigram scale. From this common intermediate, the researchers introduce oxygen, sulfur, or nitrogen to close a small heterocycle, then trigger a final ring‑forming step with a lithium reagent that snaps the second three‑membered ring into place. Remarkably, these delicate‑looking cages are more stable than their all‑carbon cousins and can be handled as bottled solutions for extended periods, making them practical reagents rather than fragile curiosities. 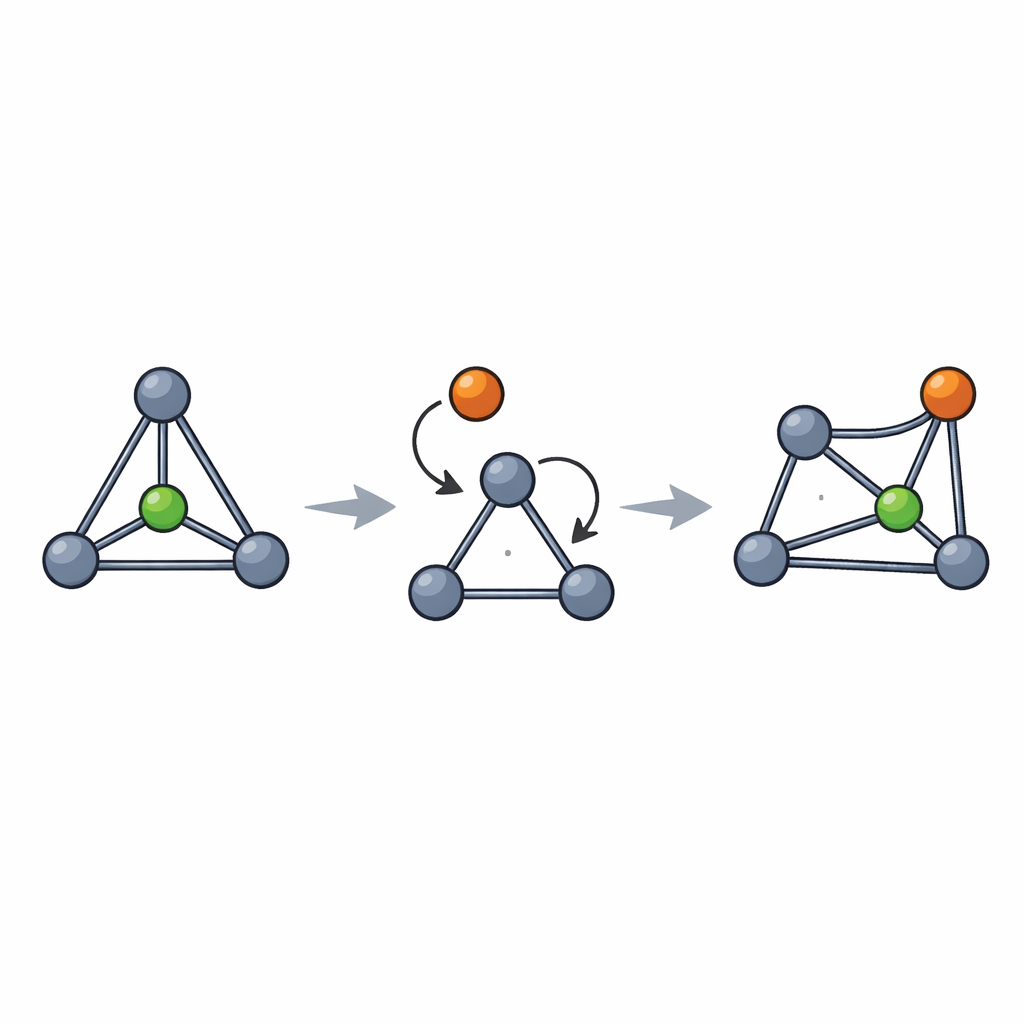
Snapping Open the Cage to Make Drug‑Like Scaffolds
Once the hetero[3.1.1]propellanes are in hand, the real power of the approach appears. Under mild radical conditions—where short‑lived reactive fragments are generated in situ—the central bond of the propellane cage can be selectively broken. This “strain‑release” ring‑opening converts the compact propellanes into a broad range of 3‑heterobicyclo[3.1.1]heptanes, each bearing a new substituent at its bridgehead positions. The authors show that many different carbon‑, nitrogen‑, sulfur‑, and selenium‑based radicals can be installed, and that some reactions can be driven by visible‑light photocatalysis. They even demonstrate late‑stage modification of complex molecules, grafting the new cage onto fragments such as sugars, small peptides, and an agrochemical, which illustrates the method’s flexibility.
Turning Concepts into Better Drug Candidates
Beyond demonstrating synthetic reach, the researchers connect their chemistry to real medicinal needs. They use an oxygen‑containing propellane to construct an analogue of the approved anticancer drug sonidegib, in which a flat benzene unit is replaced by the new three‑dimensional oxa‑bicyclo[3.1.1]heptane core. Prior studies have shown that this swap can improve solubility and other key properties without sacrificing shape. The new route offers a shorter, more modular way to access such analogues, allowing chemists to vary both the cage substituents and the surrounding parts of the drug late in the synthesis.
What This Means for Future Medicines
In essence, this work turns a theoretical curiosity—heteroatom‑containing [3.1.1]propellanes—into robust, scalable tools for medicinal chemistry. By providing a straightforward way to make and selectively open these tiny molecular cages, the authors unlock an unexplored region of chemical space where compact, three‑dimensional scaffolds can be tailored for solubility, stability, and precise fit to biological targets. For a layperson, the message is simple: by reshaping the tiny building blocks inside our medicines from flat plates into carefully engineered three‑dimensional frames that include “helper” atoms like oxygen and nitrogen, chemists gain new control over how drugs behave in the body, potentially leading to safer and more effective treatments.
Citation: Revie, R.I., Dasgupta, A., Biddick, Y. et al. Hetero[3.1.1]propellanes. Nat. Chem. 18, 502–508 (2026). https://doi.org/10.1038/s41557-026-02072-2
Keywords: drug design, bioisostere, heterocycle, strain-release chemistry, bicyclo[3.1.1]heptane