Clear Sky Science · en
Native H2 pathways enable biocompatible hydrogenation of metabolic alkenes in bacteria
Turning Microbes into Tiny Green Factories
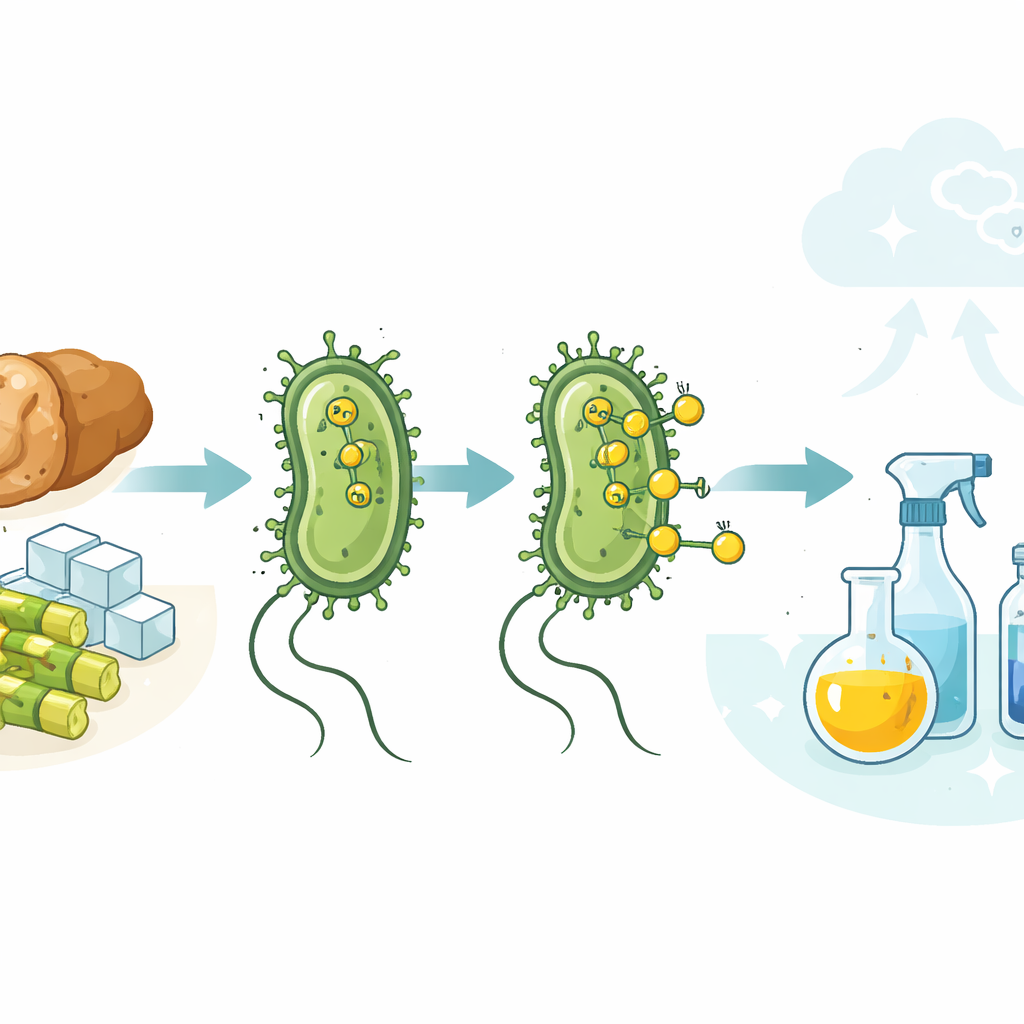
Imagine if common gut bacteria could help replace fossil fuels in making everyday chemicals—from food preservatives to plastics—while even cleaning up food waste. This study shows that ordinary laboratory strains of Escherichia coli can do just that: they naturally make hydrogen gas inside their cells, and that hydrogen can be harnessed on the cell surface to power industrial-style chemical reactions under gentle, water-based conditions. By pairing living microbes with a solid metal catalyst, the researchers outline a path toward cleaner, carbon‑saving manufacturing.
Why Hydrogen Matters for Everyday Products
Hydrogen gas sits at the heart of modern chemistry. It is used to turn liquid oils into spreads, upgrade crude fuels, and build many ingredients for medicines and plastics. Today, almost all of that hydrogen comes from fossil fuels, mainly natural gas and coal, releasing large amounts of carbon dioxide. At the same time, many microbes naturally vent hydrogen as they break down sugars when oxygen is scarce. The puzzle the authors tackle is how to tap this gentle, biological hydrogen stream to drive the same kinds of reactions that industry now performs with high pressures, organic solvents and fossil‑derived gas.
Letting Bacteria Power a Metal Catalyst
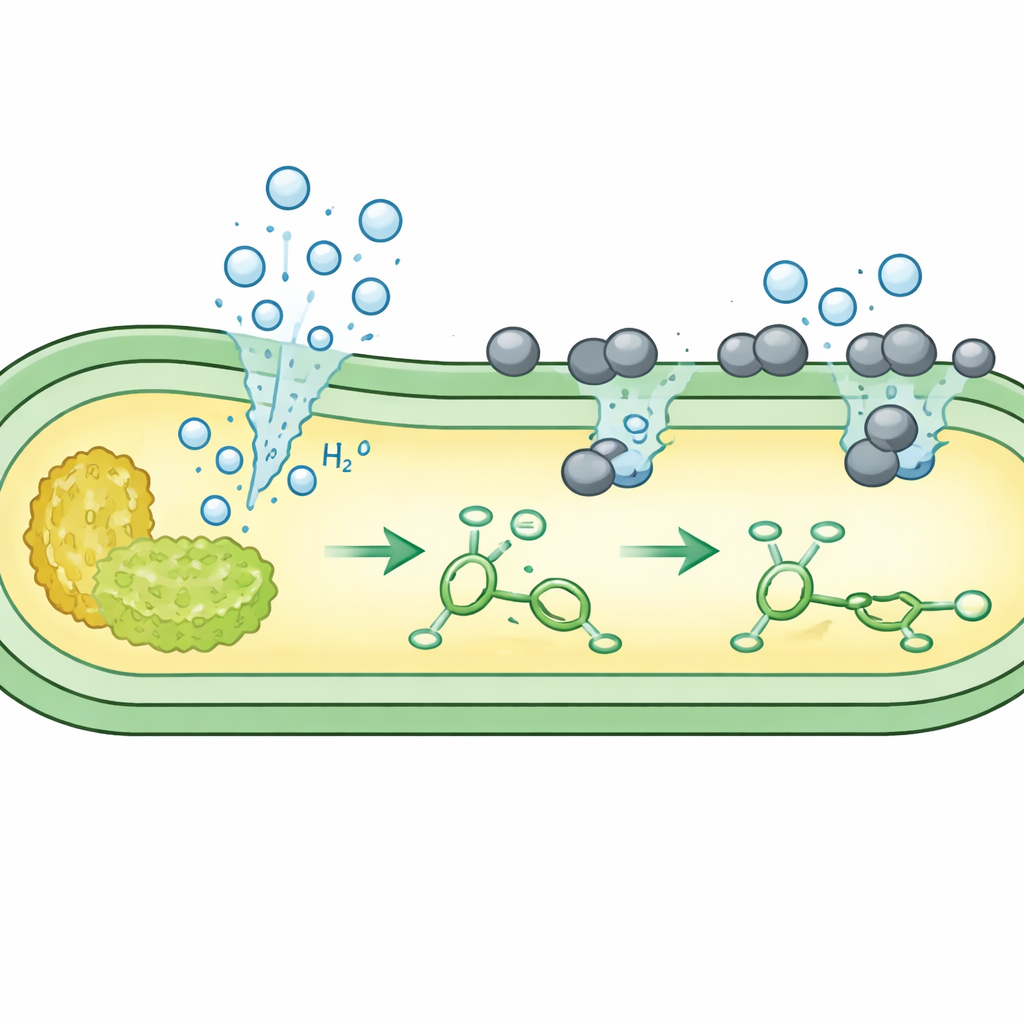
The team focused on simple hydrogen‑adding reactions, where a double bond in a molecule is “saturated” with hydrogen. They grew several unmodified E. coli strains in sugar‑based media, then added a finely dispersed palladium catalyst that is compatible with living cells. Under oxygen‑free conditions, the bacteria’s native metabolism converted sugar into formate and then into hydrogen gas at the inner face of the cell membrane. Microscopy showed that the positively charged catalyst particles nestle onto the negatively charged cell surface, where they meet the escaping hydrogen. There, the metal uses microbial hydrogen to reduce a test molecule, caffeic acid, to its saturated product in very high yields—often better than those obtained with a previous, heavily engineered strain.
Expanding the Menu of Products and Feedstocks
Once the basic reaction worked, the researchers tried a broad range of molecules containing carbon–carbon double bonds, including plant‑derived acids and long‑chain fatty compounds. Many were smoothly converted to their saturated forms, some in nearly quantitative yields. They also built E. coli strains that produce extra hydrogen by diverting more metabolic flux into the native hydrogen‑forming pathway. These boosted strains enabled similar conversions with less metal catalyst. In a striking twist, the team replaced pure sugar with liquefied waste bread as the food source. Enzymes broke the stale bread into glucose, which the bacteria fermented just as effectively, turning a common food waste into both biological hydrogen and value‑added chemicals.
Making Both Ingredient and Fuel Inside One Cell
Next, the authors asked whether a single bacterium could make not only the hydrogen “reagent” but also the unsaturated “substrate” it is meant to modify. They engineered E. coli so that sugar first flows into pathways that produce aromatic building blocks such as cinnamic and coumaric acids, and into another pathway that produces hydrogen. When the culture had built up these internal metabolites, they added the palladium catalyst. At the cell surface, the catalyst used the in situ hydrogen to convert the newly formed double‑bonded metabolites into fully saturated products such as hydrocinnamic acid, desaminotyrosine and adipic acid, a key precursor for nylon. In some designs, this cometabolism achieved almost complete conversion, all within the same living culture.
Measuring the Climate Payoff
To see whether this clever biochemistry actually helps the planet, the team performed a life‑cycle assessment comparing their hybrid “chemo‑microbial” approach with standard hydrogenation routes driven by fossil‑based or electrolytic hydrogen. When biological hydrogen and in‑cell production of substrates were combined in a single, heat‑efficient process, total greenhouse‑gas emissions dropped markedly. Using waste bread as feedstock pushed the system even further: by avoiding landfill or incineration and turning discarded food into chemicals, some scenarios became carbon‑negative overall, meaning they remove more greenhouse gases than they emit.
What This Means for Future Manufacturing
In simple terms, the study shows that everyday bacteria can be persuaded to act as both the power source and the ingredient factory for important chemical reactions, while a solid metal on their surface quietly finishes the job. Because everything happens in water, at near‑body temperature and using renewable or waste sugars, this approach could one day offer cleaner alternatives to traditional petrochemical plants. With further engineering of microbial pathways and catalysts, this living‑cell platform may enable a new generation of sustainable processes that turn renewable carbon and discarded food into useful products with a much smaller climate footprint.
Citation: White, M.F.M., Trotter, C.L., Steele, J.F.C. et al. Native H2 pathways enable biocompatible hydrogenation of metabolic alkenes in bacteria. Nat. Chem. 18, 535–543 (2026). https://doi.org/10.1038/s41557-025-02052-y
Keywords: microbial hydrogen, green chemistry, biocatalysis, waste-to-chemicals, sustainable manufacturing