Clear Sky Science · en
Stereoselective total synthesis of skew-tetramantane
A Tiny Twist inside Diamond
Diamonds are famous for their sparkle and hardness, but chemists are just as fascinated by the tiny building blocks that make up diamond’s crystal lattice. This article describes how researchers have, for the first time, built in the laboratory a remarkably small, twisted fragment of diamond called skew-tetramantane. Understanding and controlling such perfectly defined “nano-diamonds” could open the door to new materials for electronics, quantum technologies and medicine.
From Flat Carbon Sheets to 3D Diamond Cages
Carbon can arrange itself in very different ways. In flat sheets like graphene, carbon atoms form a honeycomb of hexagons. In three dimensions, they can adopt the tightly packed framework of diamond. Chemists have long been able to make many flat, ring-shaped molecules that mimic pieces of graphene, and even twist them into spiral forms called helicenes. In contrast, building equally precise three-dimensional diamond fragments—known as diamondoids—has proved far harder. Only the three smallest cages, related to the drug-like molecule adamantane, could be made reliably, while larger, more complex cages had to be painstakingly separated from fossil fuels.
Why Skew-Tetramantane Is a Special Diamond Fragment
Among the known diamondoids, skew-tetramantane occupies a special place. It is a rigid, highly stable cage that can be viewed as the smallest chiral “σ-helicene” inside the diamond lattice: its three-dimensional shape can twist in one of two mirror-image ways, much like left and right hands. In nature, skew-tetramantane appears only in minuscule amounts within petroleum and natural gas, and obtaining pure samples requires multiple rounds of high-end separation techniques. Traditional synthetic routes based on high-temperature rearrangements generate huge numbers of fleeting intermediates and tend to favor other isomers, making a targeted preparation of skew-tetramantane essentially impossible.
A Stepwise Plan to Grow a Diamond Cage
To solve this problem, the authors devised a rational “cage extension” strategy. Instead of shuffling carbon atoms under harsh conditions, they started from a smaller, well-known diamondoid called triamantane and planned to bolt on a four-carbon “cap” at a specific face of the cage. Each new carbon–carbon bond had to be formed with exquisite control over position and three-dimensional shape to ensure that only the desired skew-tetramantane framework emerged. The team used visible-light photocatalysis to gently attach a handle to triamantane, and then exploited highly selective reactions in which a reactive carbene species inserts into one particular carbon–hydrogen bond among many nearly identical options. By carefully designing the length and orientation of the handle, and by choosing chiral rhodium catalysts that favor one mirror-image pathway, they steered the growing cage along a single, well-defined route. 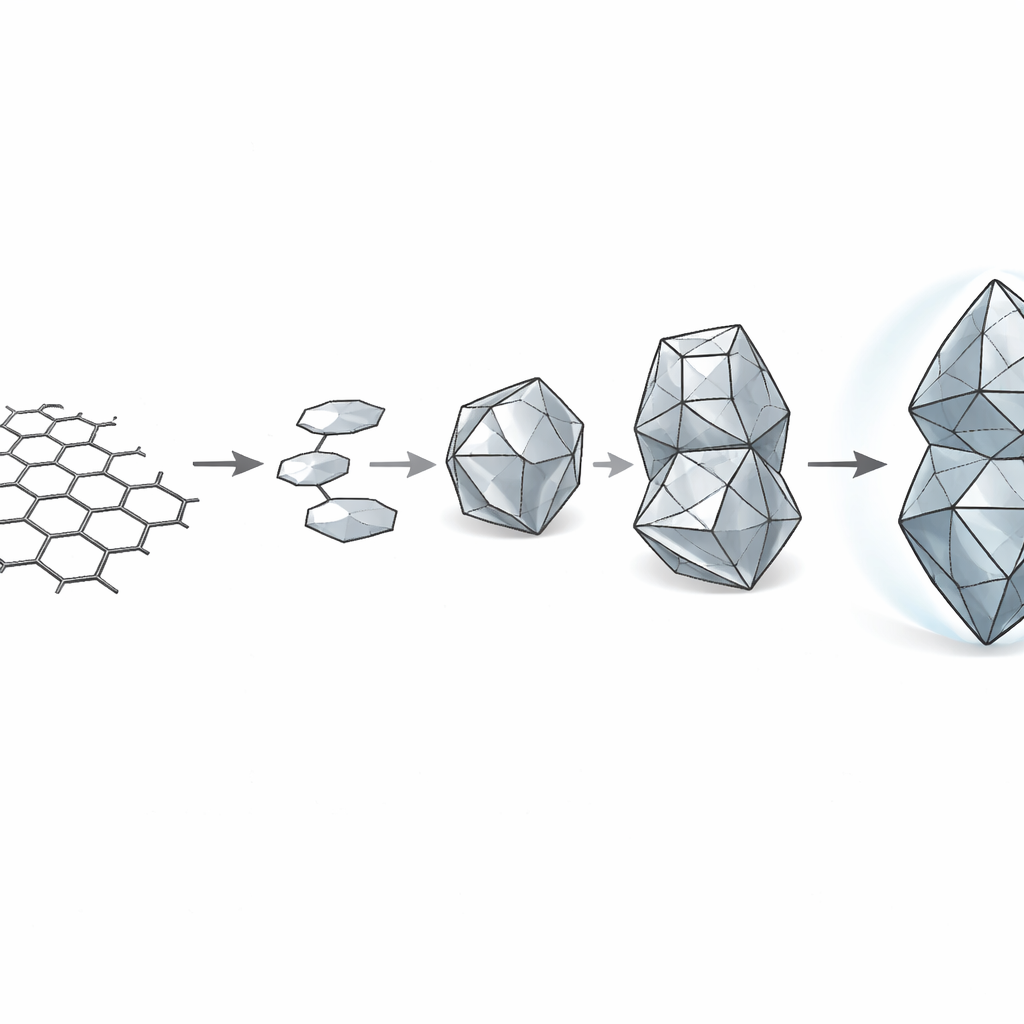
Guiding the Final Snap of the Cage
Once the partial cage was assembled, the challenge shifted to reshaping rings and closing the final gaps. The researchers used a controlled ring-expansion step, known as the Buchner–Curtius–Schlotterbeck rearrangement, to convert a five-membered ring into the six-membered ring pattern characteristic of the diamond lattice. Additional tailoring steps, including a gentle hydration that fixed the orientation of a key carbon atom, positioned the last carbon–hydrogen bond and the reactive carbene perfectly for the decisive intramolecular insertion. Under these carefully tuned conditions, the cage “snapped shut” into the skew-tetramantane framework with high selectivity, and the remaining temporary groups could be removed under mild, light-driven conditions. The final product matched skew-tetramantane isolated from fossil fuels in every measurable respect, including detailed high-field nuclear magnetic resonance data. 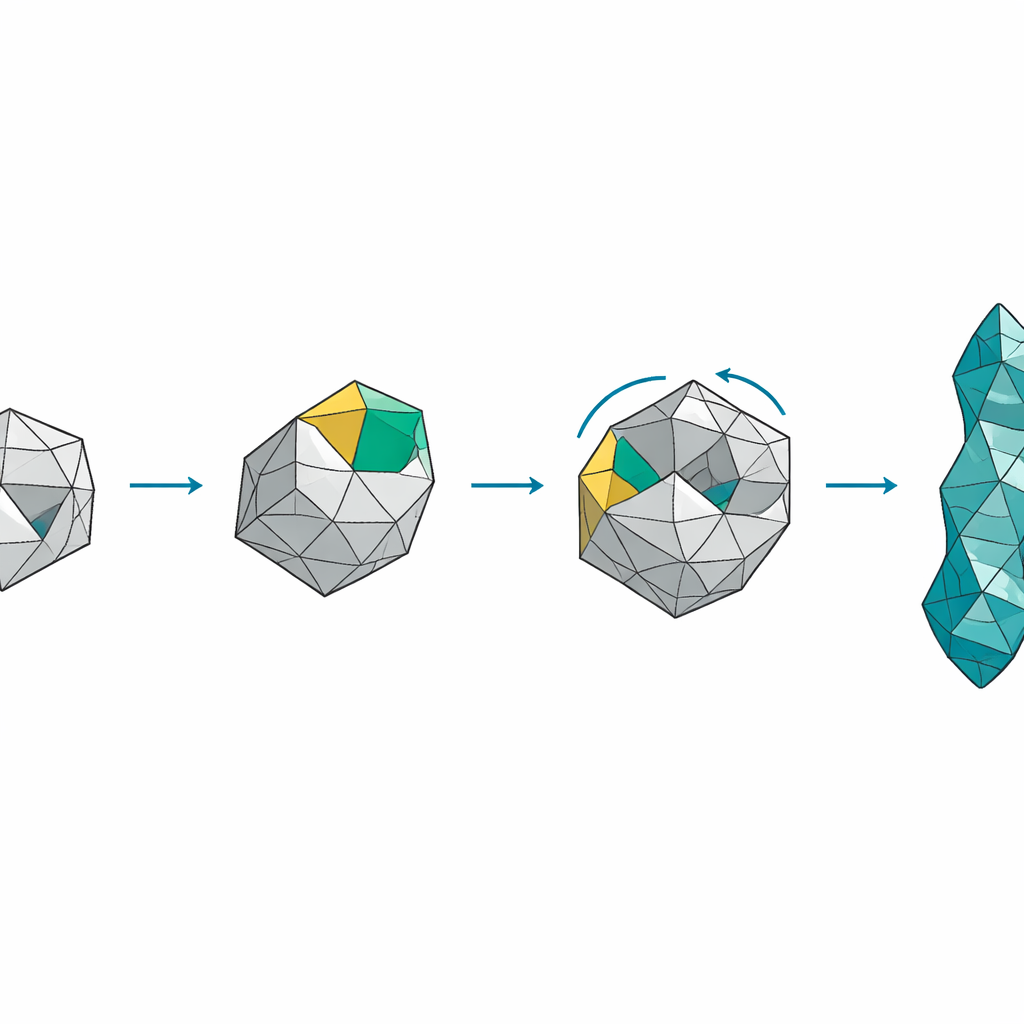
New Building Blocks for Future Technologies
In everyday terms, the researchers have learned to carve out and assemble a specific tiny twist of diamond with atomic precision, rather than sifting it from ancient oil. Their approach shows that higher diamondoids can be built up systematically at low temperature, guided by modern photocatalysis and finely tuned metal catalysts. This opens the possibility of crafting many different three-dimensional carbon cages with predictable shapes, stiffness and outward-pointing bonds. Such tailor-made nanodiamond fragments could serve as components in advanced optical materials, electronic devices, quantum bits, or as ultra-rigid scaffolds in pharmaceuticals and biomarkers, bringing the precision of molecular design to one of nature’s hardest substances.
Citation: Li, XY., Sparr, C. Stereoselective total synthesis of skew-tetramantane. Nat. Chem. 18, 597–602 (2026). https://doi.org/10.1038/s41557-025-02026-0
Keywords: diamondoids, skew-tetramantane, photocatalysis, carbene insertion, nanocarbon materials