Clear Sky Science · en
RNA-specific local translation is patterned by condensates for multinucleate cell growth
How big cells keep distant tasks in sync
Cells in our bodies are usually tiny, self-contained units with one nucleus. But some organisms, including certain fungi and muscle cells, are huge compartments filled with many nuclei sharing the same cytoplasm. In these giants, signals and molecules can take a long time to diffuse from place to place, raising a puzzle: how do they coordinate growth and division across such vast distances without losing control? This study tackles that question in a multinucleate fungus by uncovering how tiny droplets inside the cell locally tune protein production right where it is needed.
Tiny droplets that organize a giant cell
In the filamentous fungus Ashbya gossypii, long tube-like cells called hyphae contain many nuclei that divide out of sync while growth occurs mainly at the tips. Earlier work showed that a protein named Whi3 forms microscopic droplets, or condensates, with specific messenger RNAs (mRNAs) that code for key regulators of the cell cycle and tip growth. Here, the authors set out to understand what these condensates actually do. Using fast three-dimensional imaging, they found that Whi3 condensates change in size and number depending on where they are in the cell and on the local state of growth and nuclear division. Large condensates cluster near slowly growing hyphal tips, while smaller, more variable condensates surround nuclei at particular stages of the cell cycle. Mutant fungi that cannot form normal condensates grow faster at the tips and show more synchronized nuclear divisions, hinting that condensate properties help coordinate these processes.
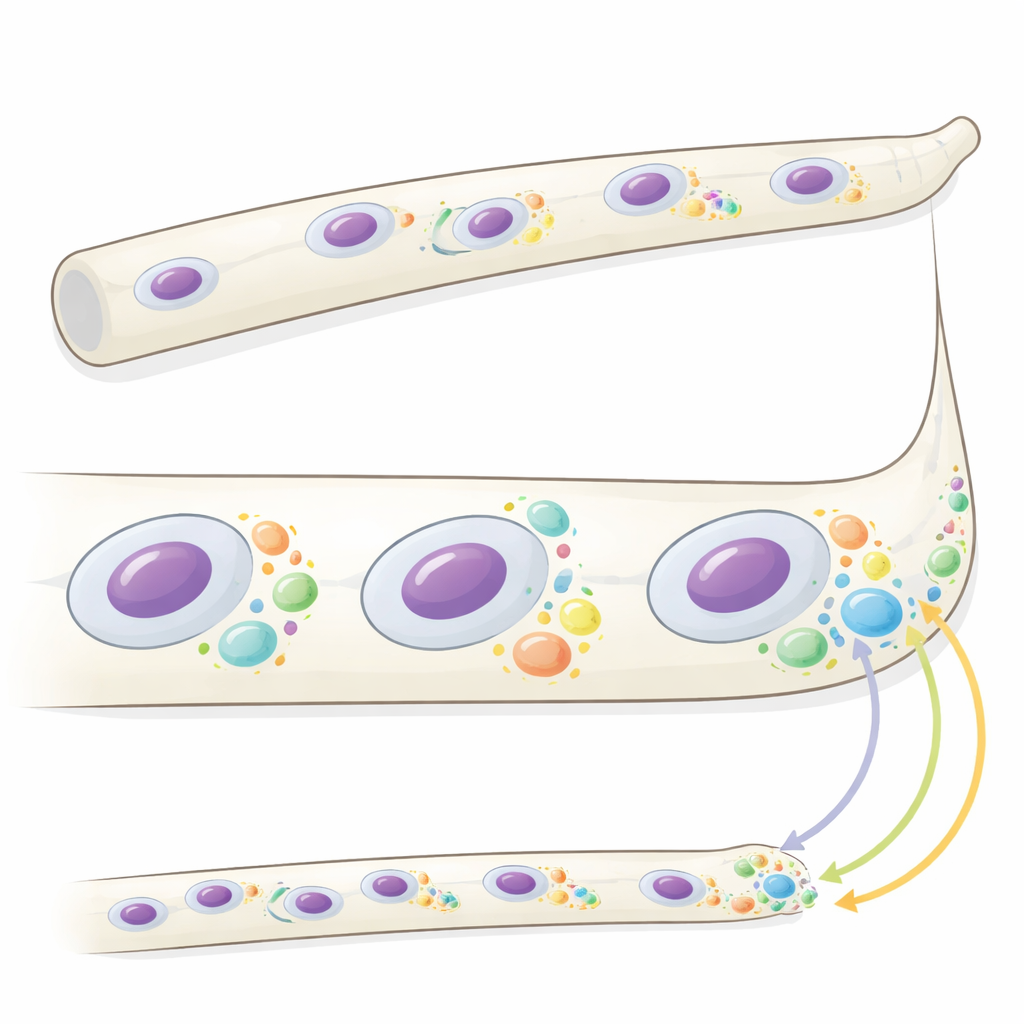
Local protein production written in space and time
To see whether Whi3 condensates control where proteins are made, the team measured translation — the process of turning mRNA into protein — for two important Whi3-bound messages. One, CLN3, encodes a cyclin that helps drive cells through the division cycle; the other, BNI1, encodes a factor that shapes growth at the hyphal tip. Using a sensitive fluorescence-based method that detects active ribosomes on individual endogenous mRNA molecules, the researchers showed that CLN3 is translated mainly near certain nuclei and only during specific phases of their cycle, especially around mitotic nuclei. BNI1 mRNA clusters at hyphal tips, but its translation there is usually sparse and variable. When Whi3’s behavior is altered by a phospho-mimic mutation, BNI1 translation becomes strongly enriched at the tips, and tip growth speeds up, revealing that Whi3 can either repress or permit translation depending on its state and location.
Minimal parts, many outputs
To test whether this tuning is an intrinsic property of the condensate components, the authors recreated the system in cell-free extracts. They attached a luciferase reporter to the regulatory regions of CLN3 or BNI1 mRNAs and mixed these RNAs with purified Whi3 at different concentrations. At low levels, where no droplets form, translation changed little. At higher levels that drive condensate formation, CLN3-linked translation was strongly repressed as Whi3 concentration and condensate size increased. BNI1 behaved differently: moderate condensate levels enhanced its translation, whereas more abundant or larger condensates flipped the system into repression. Mutant versions of Whi3 that formed smaller or fewer droplets, or RNAs with fewer Whi3 binding sites, shifted these responses, often relieving repression and boosting translation. These experiments show that the same basic ingredients — Whi3, its target RNAs, and their condensates — can generate a continuum of translation states simply by altering concentration, interaction strength, or droplet size.
Where translation happens: at the droplet’s edge
Bulk assays average over many molecules and cannot reveal where translation occurs within or around condensates. To visualize this directly, the team used a “MoonTag” reporter that lights up when new protein chains emerge from ribosomes. In vitro, they saw MoonTag signals accumulate on and within Whi3–RNA droplets, proving that these condensates can indeed be active translation sites rather than mere storage depots. Strikingly, both ribosomes and nascent-protein signals were strongest at the droplet interface, forming a bright ring around each condensate. Smaller condensates, with more surface area relative to their volume, supported more translation per RNA than larger droplets, which were more repressive overall. Changing RNA valency or Whi3’s charge state shifted whether this translation-permissive zone sat at the surface or deeper inside, indicating that subtle molecular features of the condensate tune how easily translation machinery can access resident RNAs.
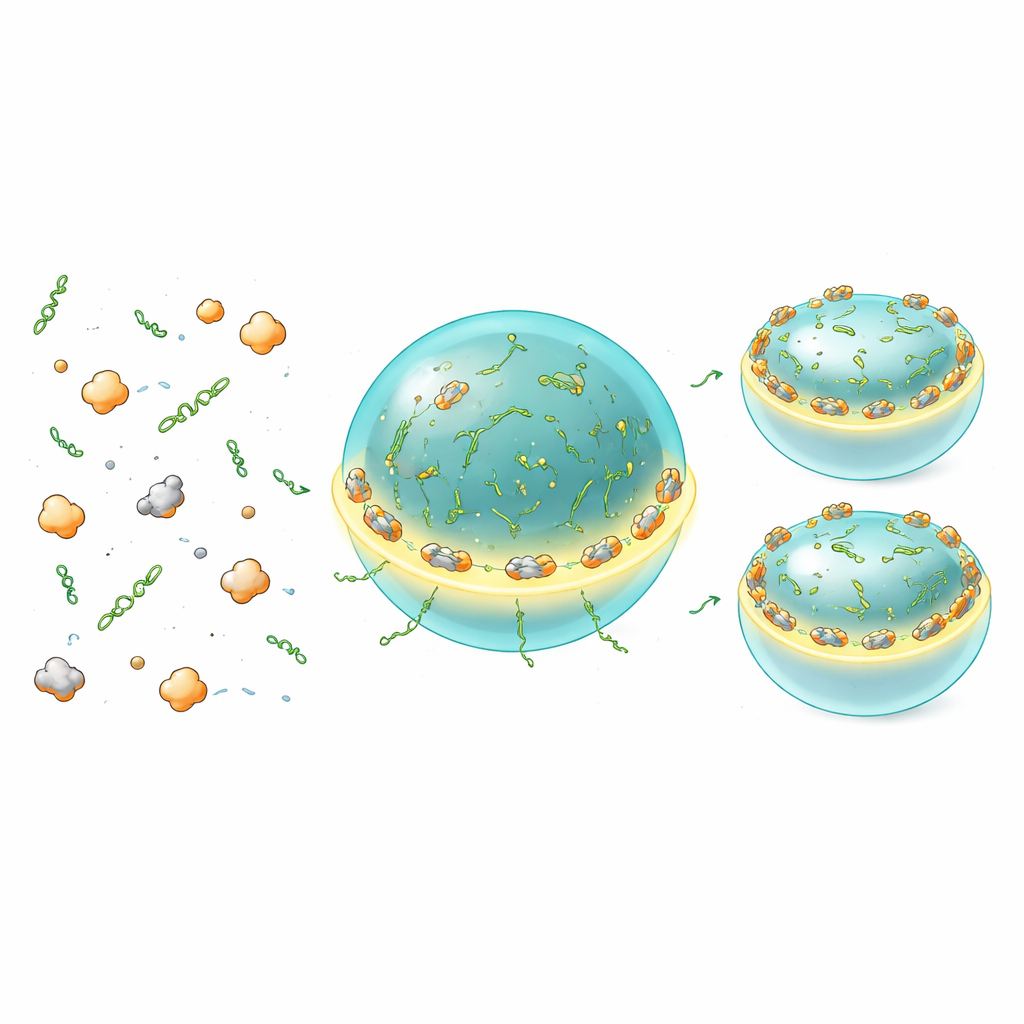
Fine-tuned dosing for balanced growth
Altogether, the work portrays Whi3–RNA condensates as adjustable valves that meter out locally produced protein instead of simple on–off switches. In the fungus, this means CLN3 cyclin can be produced in pulses around selected nuclei to keep their division cycles out of phase, while BNI1 can be made in intermittent bursts at hyphal tips to sustain growth yet prevent runaway expansion at a single site. When condensate formation or properties are disrupted, this nuanced control is lost: protein production becomes more uniform in space and time, nuclei divide synchronously, branching patterns change, and overall morphology is disturbed. For a general audience, the key message is that cells can use tiny phase-separated droplets as spatially and temporally tunable reactors, allowing huge, multinucleate cells to coordinate growth and division across long distances by locally sculpting when and where proteins are made.
Citation: Geisterfer, Z.M., Jalihal, A.P., Cole, S.J. et al. RNA-specific local translation is patterned by condensates for multinucleate cell growth. Nat Cell Biol 28, 507–519 (2026). https://doi.org/10.1038/s41556-026-01887-y
Keywords: biomolecular condensates, local translation, cell cycle control, fungal hyphae, phase separation