Clear Sky Science · en
Lysine-11 ubiquitination drives type-I/III interferon induction by cGAS–STING and Toll-like receptors 3 and 4
How Our Cells Sound the Alarm
When viruses or dangerous molecules invade the body, our cells must decide within minutes whether to launch powerful antiviral alarm signals called interferons. These signals can save us from severe infections—but when misfired, they also drive damaging autoimmune diseases. This study uncovers a previously hidden molecular “switchboard” inside cells that helps decide when to turn interferon responses on, and when to hold them back. Understanding this switchboard offers clues for better vaccines, antiviral therapies and treatments for interferon-driven disorders.
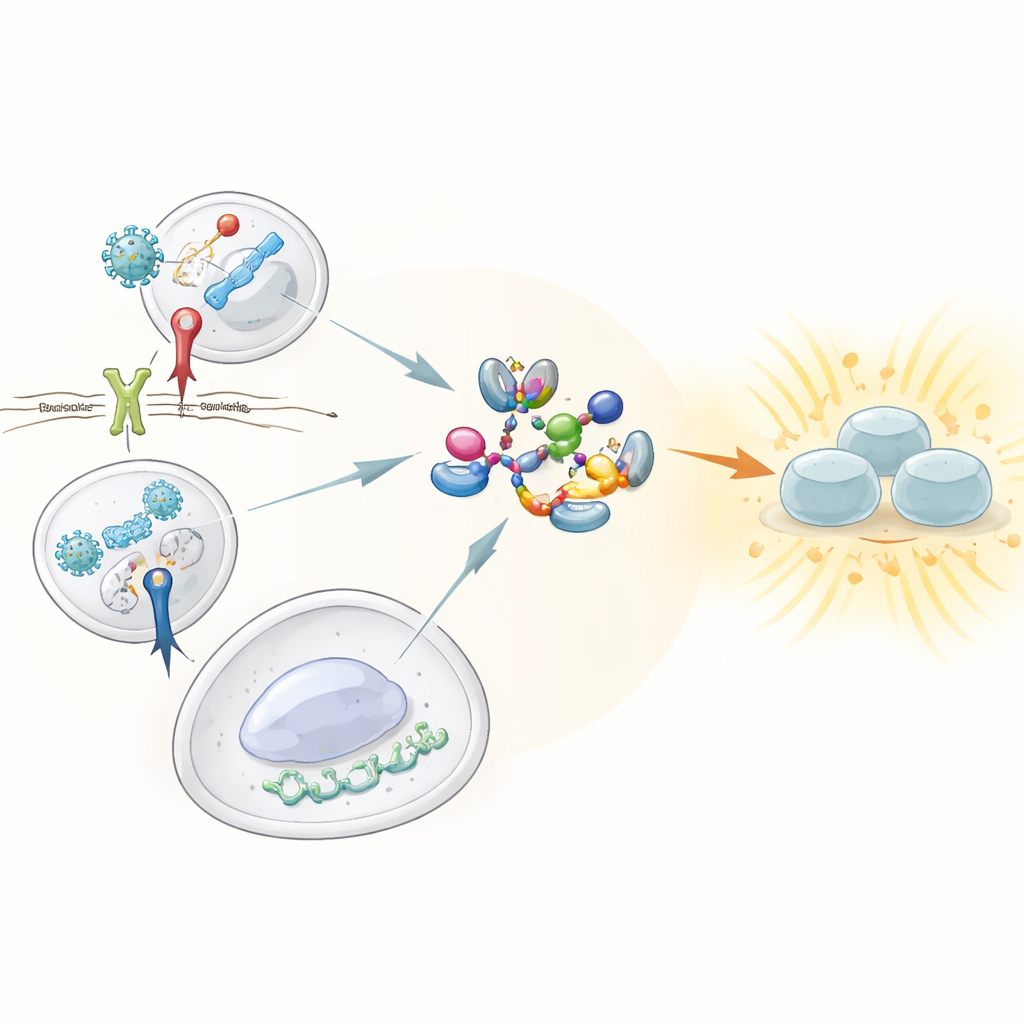
Guardians That Sense Trouble
Our innate immune system relies on sentinels known as pattern recognition receptors, which patrol for telltale features of viruses and bacteria. Among them, three stand out for their ability to trigger strong interferon responses: Toll-like receptor 3 (TLR3), which detects double-stranded viral RNA; Toll-like receptor 4 (TLR4), which senses a component of bacterial cell walls; and the cGAS–STING pathway, which spots stray DNA in the wrong place inside cells. All three must activate a central enzyme called TBK1, which in turn switches on the transcription factor IRF3 to drive the production of type I and III interferons. Until now, how these sensors converged on TBK1 to flip it into its active state was not well understood.
A Hidden Molecular Connector
The researchers discovered that a protein called ANKIB1 acts as a crucial connector between these sensors and TBK1. ANKIB1 is an enzyme that attaches small molecular tags, known as ubiquitin chains, to other proteins. Importantly, it builds a specific type of chain, based on a linkage called “K11,” which had previously been better known for roles in cell division than in immunity. In human cells lacking ANKIB1, stimulation of TLR3 or activation of STING no longer led to proper TBK1 and IRF3 activation, and interferon genes barely turned on. Restoring ANKIB1 brought the response back, showing that this single enzyme is both necessary and sufficient to drive this branch of the antiviral alarm.
Building a Docking Platform for Relay Proteins
Closer inspection revealed how ANKIB1 exerts its influence. When TLR3 or STING sense danger, they assemble multi-protein platforms called signalosomes. ANKIB1 is recruited to these platforms through a region that recognizes ubiquitin. Once there, it decorates several key components—including the adaptor proteins TRIF and STING themselves, as well as other signaling molecules—with K11-linked ubiquitin chains. These chains do not simply mark proteins for destruction; instead, they act like molecular Velcro. Another protein, Optineurin (OPTN), is drawn specifically to these modified sites. Serving as a relay adaptor, OPTN then brings TBK1 into the complex so it can be activated and pass the signal onward to IRF3 and interferon genes. When OPTN was removed, TBK1 activation collapsed, underscoring that K11-linked tagging by ANKIB1 and OPTN binding form a single, essential axis.
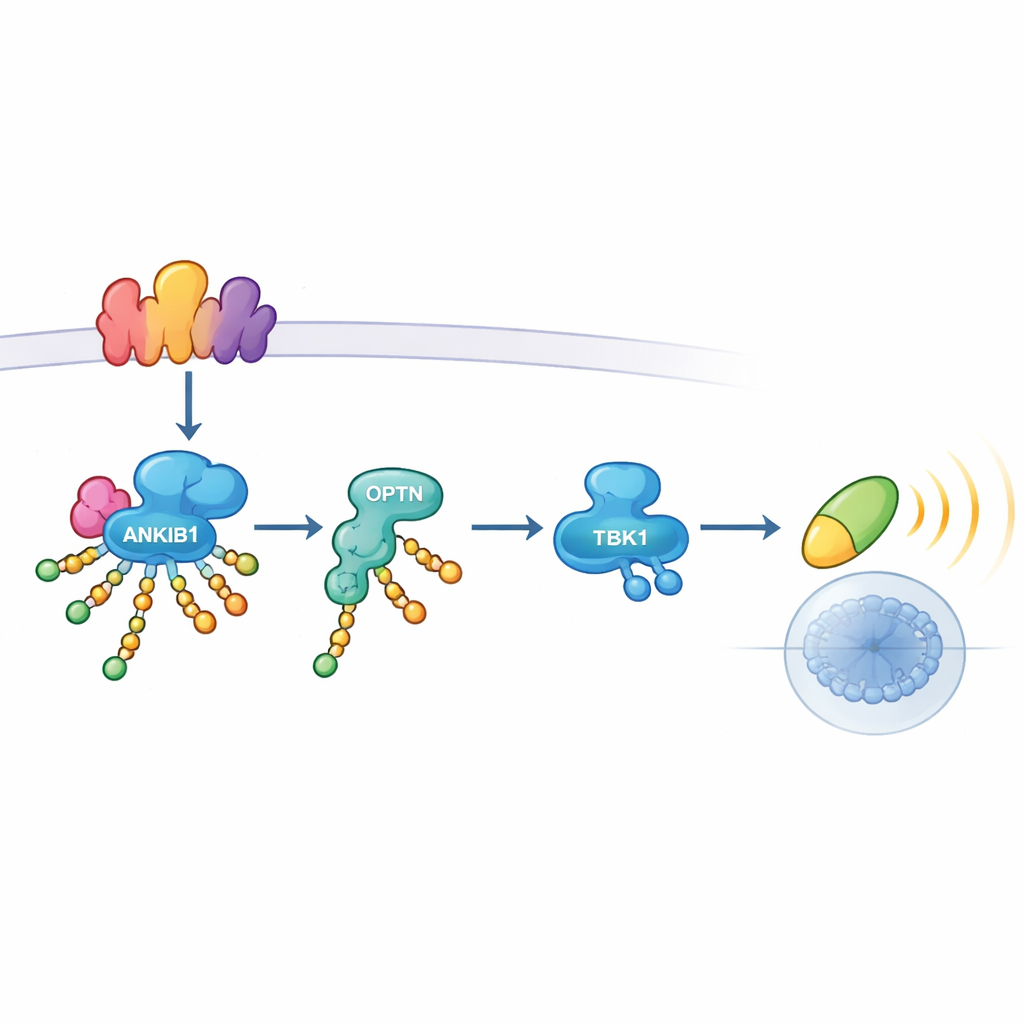
When This Pathway Matters—and When It Does Not
The team next asked whether this mechanism is universal across all antiviral sensors. They found that ANKIB1 is vital for interferon induction through TLR3, the TRIF-dependent arm of TLR4, and the cGAS–STING pathway, all of which use OPTN as the main adaptor to TBK1. In contrast, another sensor, RIG-I, which detects viral RNA, relies on a different set of adaptor proteins and does not need ANKIB1 or OPTN to activate TBK1. Experiments in mice mirrored these cell-based findings. Animals lacking Ankib1 produced far less interferon after activating TLR3 or STING, were protected from a lethal interferon-driven inflammatory syndrome, yet became much more vulnerable to infection with herpes simplex virus 1, a DNA virus that depends heavily on cGAS–STING–driven interferon for control. Responses to influenza virus, which is mainly detected by RIG-I, remained largely intact.
Balancing Protection and Harm
Together, these results reveal ANKIB1-generated K11-linked ubiquitin chains as a central currency in a major branch of innate antiviral signaling. By creating docking sites for OPTN, ANKIB1 helps assemble the precise molecular relay that turns danger sensed by TLR3, TLR4 (through TRIF) and cGAS–STING into robust interferon production. This axis is double-edged: it is essential for defense against certain viruses, yet also fuels interferon-driven inflammatory disease when overactivated. Because ANKIB1’s catalytic activity and specific domains are required for this function, they present attractive targets for future drugs designed either to dampen harmful interferon responses in autoimmune conditions or to boost antiviral immunity where it is too weak.
Citation: Betrancourt, A., Cinko, M.T., Varanda, A.B. et al. Lysine-11 ubiquitination drives type-I/III interferon induction by cGAS–STING and Toll-like receptors 3 and 4. Nat Cell Biol 28, 608–621 (2026). https://doi.org/10.1038/s41556-026-01886-z
Keywords: innate immunity, interferon signaling, ubiquitination, cGAS–STING pathway, viral infection