Clear Sky Science · en
Peritumoural adipose tissue drives immune evasion in colorectal cancer via adipose–mesenchymal transformation
Why Belly Fat Around Tumors Matters
Colorectal cancer grows inside the wall of the intestine, but it sits right next to a thick cushion of belly fat. Most people think of this fat as passive padding. This study reveals something surprising: the fat wrapped around the tumor can quietly steal cancer-fighting immune cells away from the tumor, helping the cancer hide from the immune system. Understanding this hidden tug-of-war suggests new ways to predict who will respond to immunotherapy and how to make these treatments work better.
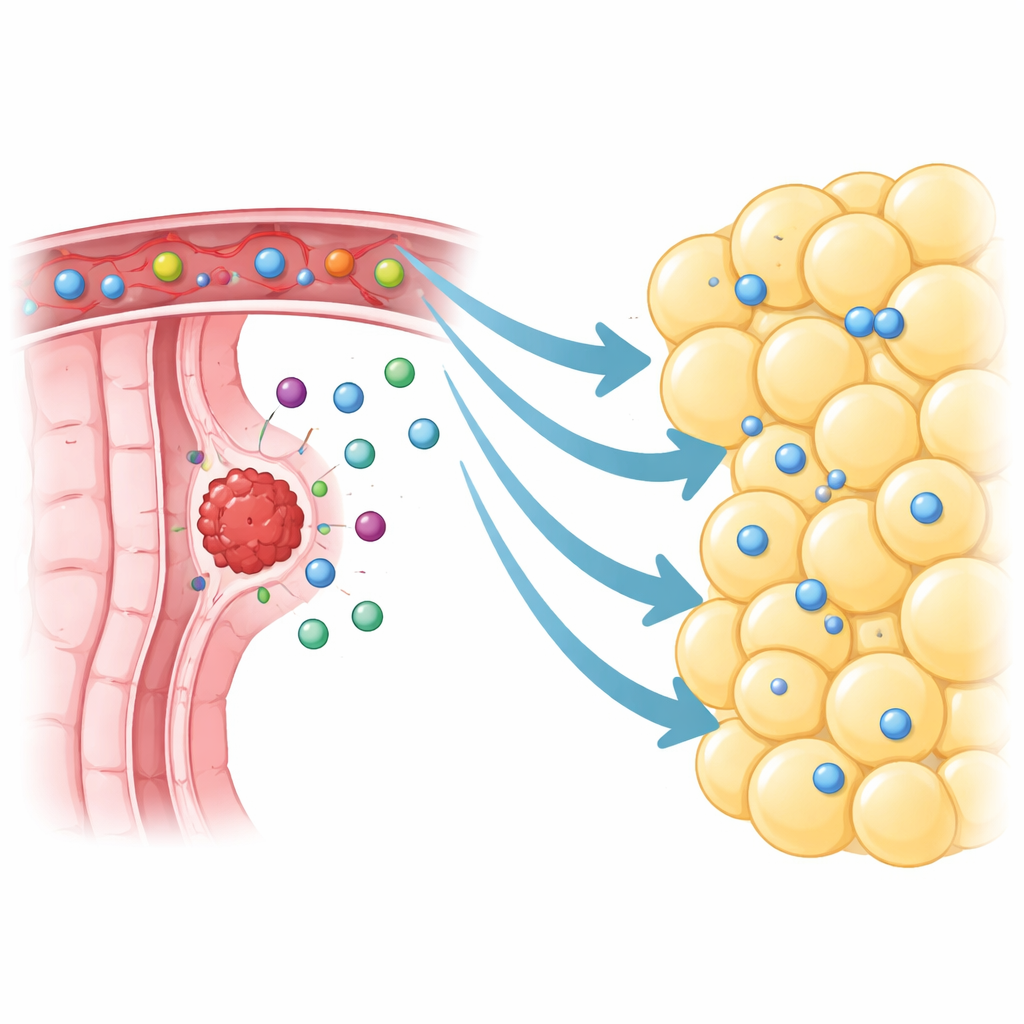
A Busy Immune Neighborhood Next to the Tumor
The researchers focused on visceral fat directly surrounding colorectal tumors, called peritumoural visceral adipose tissue. Using single-cell RNA sequencing, they mapped more than 370,000 individual cells from tumors, nearby fat, distant fat, and normal tissue. Instead of being quiet and inert, the fat next to tumors was teeming with immune cells, especially T cells and B cells. They even found organized clusters resembling mini lymph nodes, signs that this tissue can host active immune reactions. Compared with fat taken farther away, the peritumoural fat held many more lymphocytes, including memory and tumor-specific killer T cells that, in principle, should be patrolling for cancer.
When Helpful Immune Cells End Up in the Wrong Place
To see whether this immune-rich fat was helping or hurting, the team used mouse models where tumors were implanted near a fat pad resembling the human situation. They then surgically removed this nearby fat or chemically dissolved it. Counterintuitively, taking away the peritumoural fat slowed tumor growth. Tumors from mice that had lost this fat contained more CD4 and CD8 T cells, including T cells that recognized tumor antigens. Additional experiments tracking donor immune cells showed that, when the fat was present, many of these cells accumulated in the fat pad instead of in the tumor. Removing the fat redirected the cells into the tumor, strengthening the anti-cancer response. In other words, the fat acted as a competitive reservoir that soaked up immune cells and kept them from reaching their true target.
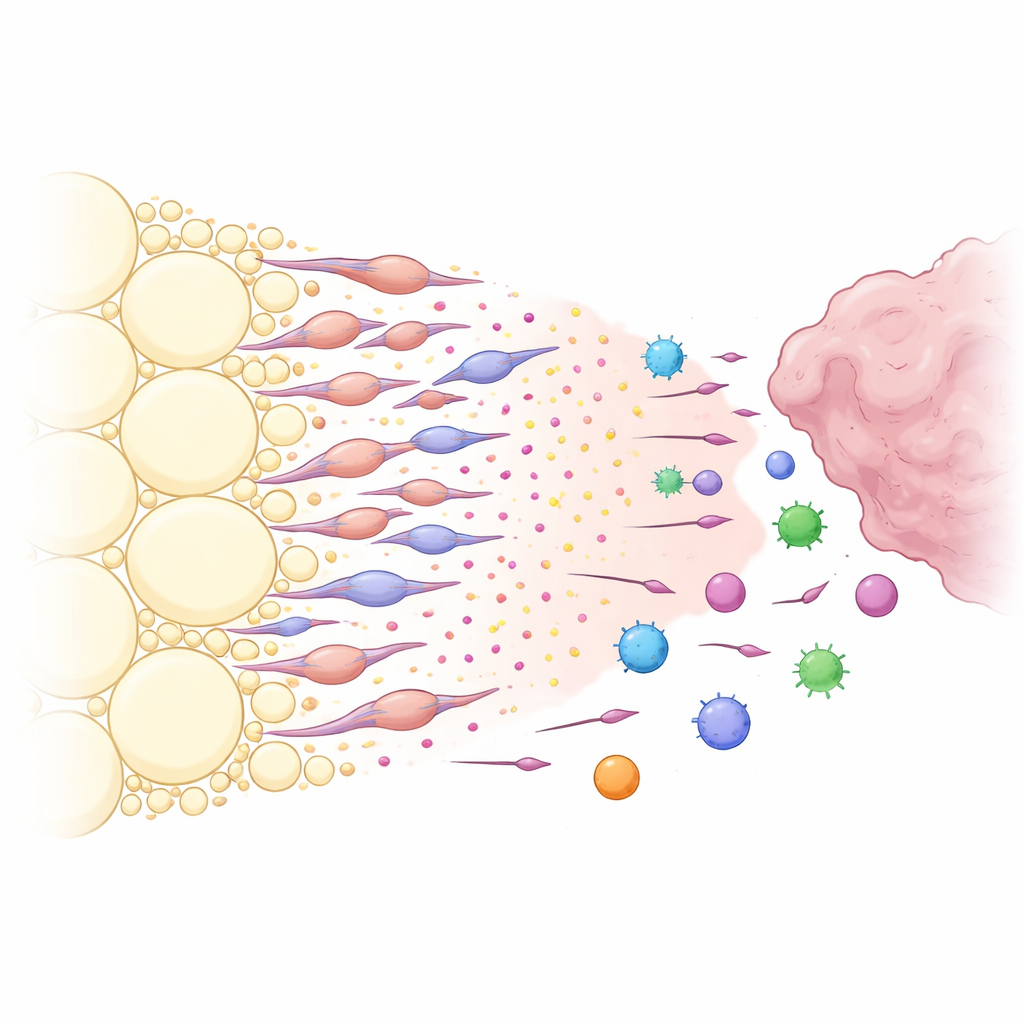
A Chemical Lure Built by Transformed Fat Cells
What made the peritumoural fat so good at capturing immune cells? By analyzing communication signals between cell types, the researchers identified a powerful chemical pathway centered on the molecule CXCL12 and its receptor CXCR4. Stromal cells in the fat near the tumor produced high levels of CXCL12, while many immune cells expressed CXCR4, making them sensitive to this signal. Blocking CXCL12 locally in the fat, or genetically deleting it in the fat stroma, caused tumors to shrink and allowed more CXCR4-positive immune cells to enter the tumor instead of being diverted. The team then discovered that the main CXCL12 producers were not ordinary fat cells, but a special group of fat-derived fibroblast-like cells they termed adipose-derived cancer-associated fibroblasts. Tumor-released factors, especially the growth factor TGF-β1, pushed local adipose stromal cells to transform into these fibroblast-like cells, which in turn secreted large amounts of CXCL12 and other growth-promoting signals.
Turning a Shield into a Weak Spot
Because these transformed cells help funnel immune cells away from tumors, the researchers tested whether dismantling this “immune sink” could improve immunotherapy. In mice engineered so that the adipose-derived cancer-associated fibroblasts could be selectively eliminated, treatment with a PD-1–blocking antibody became far more effective: tumors grew less, and more tumor-specific and CXCR4-positive T cells infiltrated the cancer. Similarly, drugs or antibodies that blocked either CXCL12 or its receptor CXCR4 worked synergistically with PD-1 blockade in both subcutaneous and orthotopic colorectal cancer models. These combinations not only reduced tumor size but also reshaped the immune landscape inside the tumor.
Clues from Patient Scans and What Comes Next
The team then asked whether their findings mattered in real patients. In a group of people with locally advanced colorectal cancer receiving combined chemotherapy, radiotherapy, and PD-1 immunotherapy, those who had a complete response tended to have a smaller volume of peritumoural visceral fat on MRI before treatment. Quantitatively, the area of this fat predicted therapy response better than some standard tissue markers, and patients with large peritumoural fat volumes had much lower response rates. To a non-specialist, the takeaway is striking: not only the tumor itself, but also the fat immediately surrounding it, can determine how well immunotherapy works. By focusing on this fat—either measuring it to guide treatment decisions or targeting its transformed stromal cells and the CXCL12–CXCR4 signal—it may be possible to turn a protective shield for the tumor into a therapeutic weak point.
Citation: Wang, JH., Zheng, YQ., Qian, ZY. et al. Peritumoural adipose tissue drives immune evasion in colorectal cancer via adipose–mesenchymal transformation. Nat Cell Biol 28, 581–595 (2026). https://doi.org/10.1038/s41556-026-01885-0
Keywords: colorectal cancer, tumor microenvironment, visceral fat, immunotherapy, CXCL12 CXCR4