Clear Sky Science · en
Adaptor-mediated recruitment of three dyneins to dynactin enhances force generation
How Cells Haul Heavy Cargo
Inside every cell, tiny molecular machines constantly haul cargo along microscopic tracks, keeping the cell alive and organized. This paper explores how one of the cell’s main “freight engines,” a motor called dynein, automatically shifts into a higher gear when it encounters extra resistance. Understanding this built‑in power boost helps explain how nerve cells move large structures over long distances, and why defects in these systems can contribute to brain disorders.
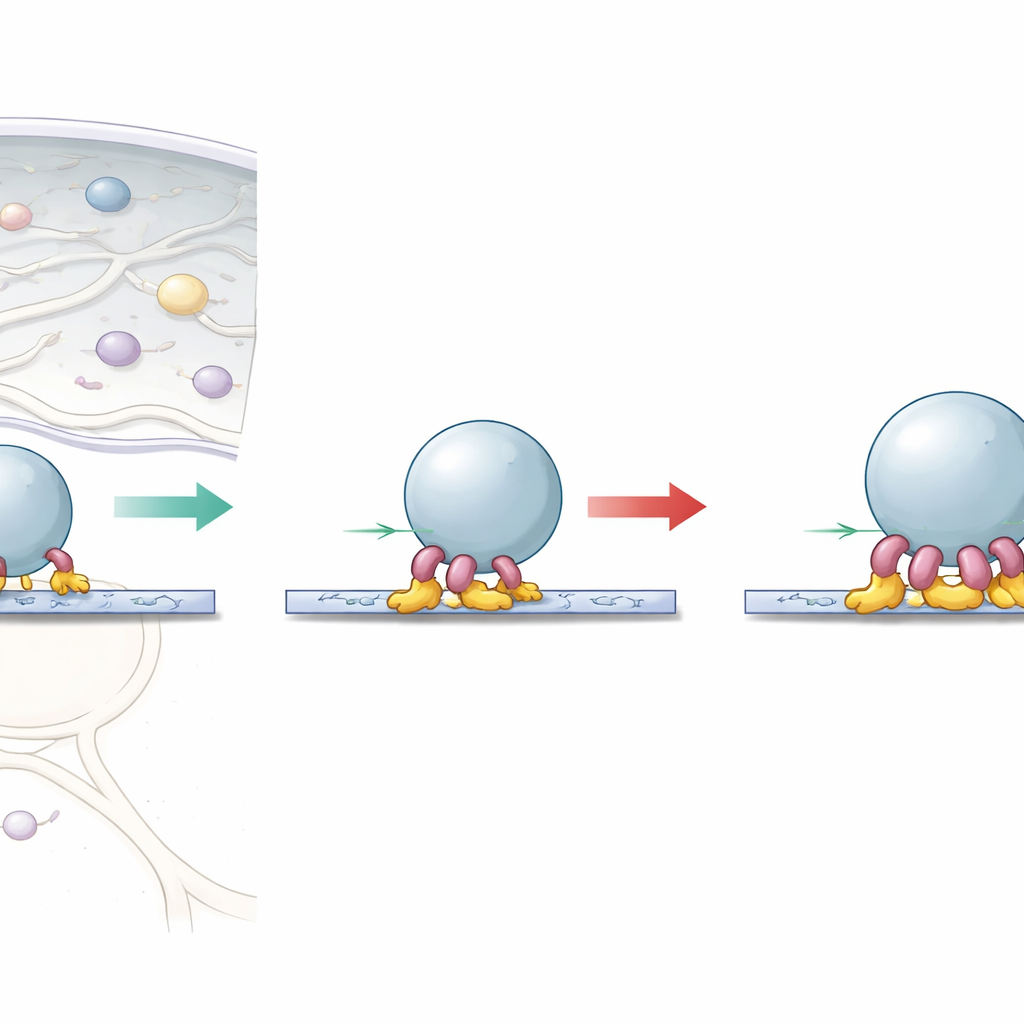
The Cell’s Backward‑Pulling Engines
Cells are crisscrossed by stiff protein rails known as microtubules. Dynein is a motor that walks along these rails, usually carrying cargo from the outer regions of the cell toward the center. It rarely works alone. A long adaptor protein called BicD2 helps attach dynein to cargo, while a scaffold complex called dynactin helps keep the motor moving steadily. Together, dynein, dynactin and BicD2 form a transport unit that can pull against opposing forces inside the crowded cell. Another helper protein, Lis1, is crucial for brain development, but its exact role in controlling dynein’s pulling power has been unclear.
A Built‑In Handbrake That Limits Force
The researchers used ultra‑sensitive optical tweezers—essentially laser “handles” that can measure forces on a single cargo‑coated bead—to watch these transport units pulling along microtubules. They discovered that a unit containing just one dynein motor has two distinct strength settings. In a relaxed state, the motor often stalls at a modest force, as if a handbrake were partly on. With help from Lis1, or from specific mutations that hold dynein in an open, active shape, this handbrake is released and the same single motor can pull considerably harder before it stalls. This suggests that dynein naturally flips into a folded, self‑inhibited form that limits its force, and that Lis1’s main job is to keep the motor in its fully active configuration.
Adding Extra Engines Under Strain
When the team looked at more complex assemblies, they saw that transport units did not have just one possible stall force but several distinct plateaus. Two dyneins working together produced a higher level of force, and in some conditions a third dynein could join, pushing the stall force even higher. The key to recruiting that third motor turned out to be a second BicD2 adaptor molecule that latches onto a part of the extra dynein. Under backward tension—when the cargo is strongly resisted—this auxiliary adaptor is more likely to engage, allowing a third dynein to dock onto the dynactin scaffold. Mutating the contact point between this extra adaptor and the third dynein sharply reduced the highest force state, confirming that this interaction is essential for building a three‑motor team.
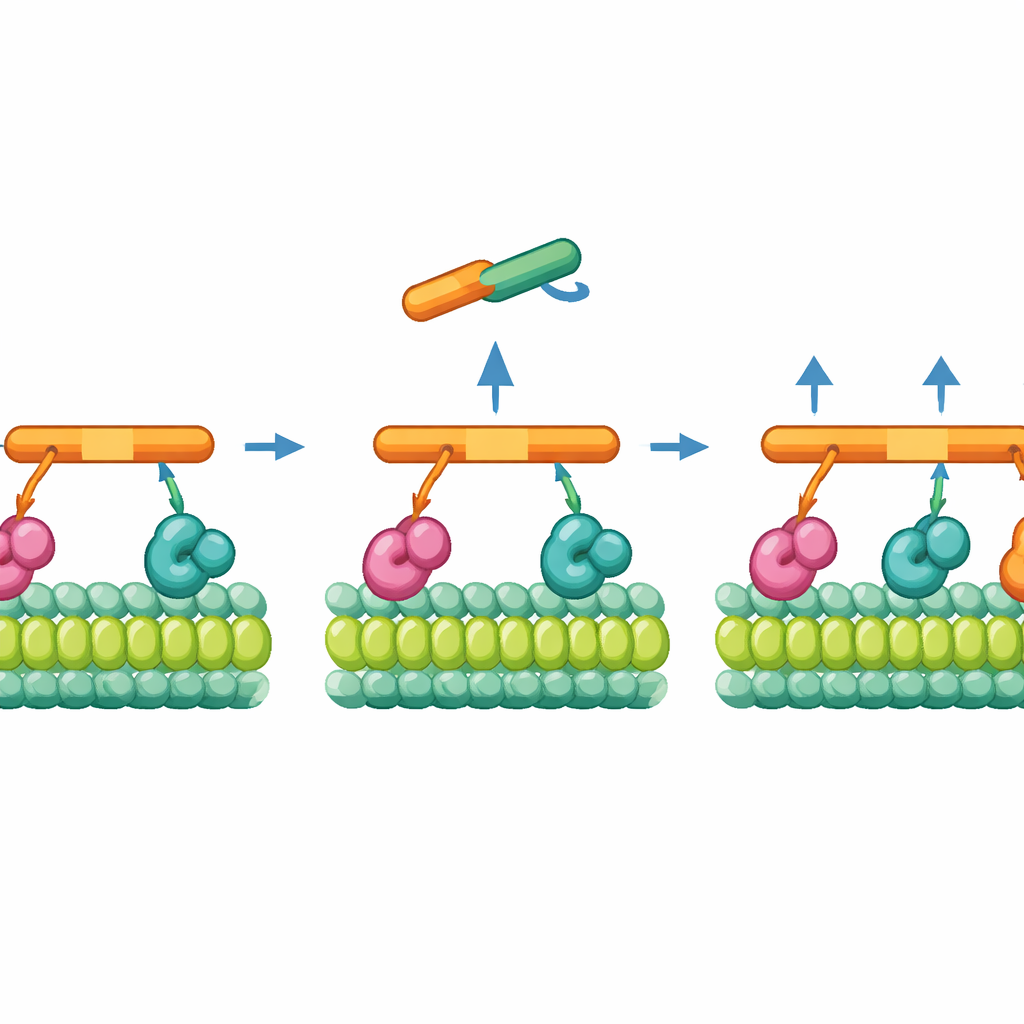
How Load Changes the Way Motors Step
Beyond measuring overall force, the authors also tracked how far the cargo moved with each tiny step. Under normal pulling conditions, dynein teams advanced in mostly uniform, nanometer‑sized increments, consistent with a compact, tightly coordinated group of motors. As the load increased and a third dynein joined, the steps became slightly smaller and motion slowed, suggesting more complex coordination when three engines share the job. The motors also showed brief forward‑and‑back motions that resembled a driver feathering the gas and brake, hinting at a stochastic, rather than perfectly synchronized, stepping pattern that nonetheless keeps cargo moving in the right direction.
Why This Matters for Healthy Cells
Overall, the study reveals that dynein transport units are not fixed machines but adaptive teams. A self‑inhibitory shape limits how hard a lone motor can pull, Lis1 and mechanical load help flip the system into more powerful states, and an extra adaptor allows a third motor to join when resistance is high. In everyday terms, the cell’s freight engines can sense when the load gets heavier and automatically add more engines to the train, ensuring that cargo still reaches its destination. This flexible response to changing mechanical demands helps explain how cells maintain reliable transport in complex environments, and offers new clues to how disruptions in these regulators might underlie certain neurodevelopmental diseases.
Citation: Rao, L., Liu, X., Arnold, M. et al. Adaptor-mediated recruitment of three dyneins to dynactin enhances force generation. Nat Cell Biol 28, 480–491 (2026). https://doi.org/10.1038/s41556-026-01877-0
Keywords: dynein motor, intracellular transport, molecular motors, cell mechanics, Lis1 adaptor