Clear Sky Science · en
Hydrogen production from wastewater via ammonia gas recovery
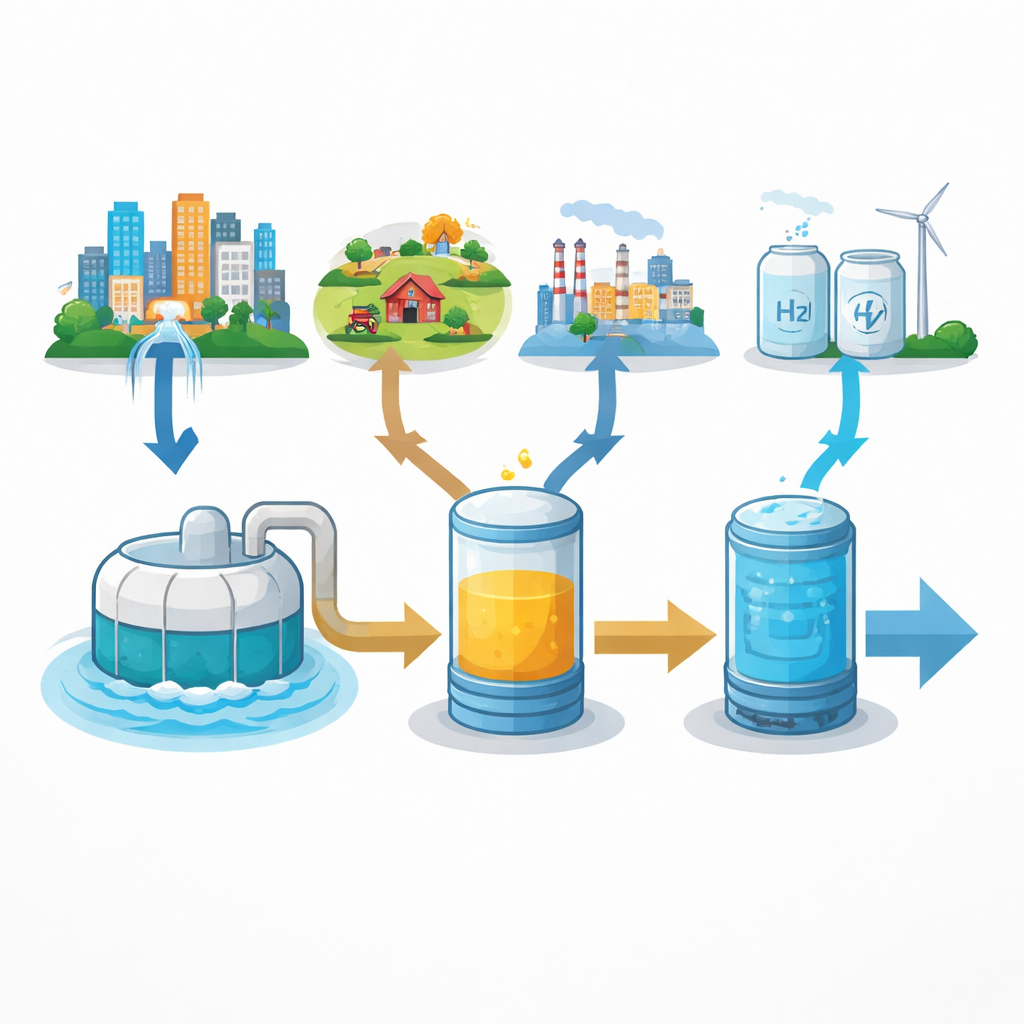
Turning Dirty Water into Clean Energy
Every day, cities, farms, and factories flush away wastewater rich in nitrogen. Today that nitrogen is mostly treated as a pollutant and lost to the air, even though it represents a vast, overlooked resource. This study explores a bold idea: instead of throwing that nitrogen away, we can capture it as ammonia and transform it into hydrogen, a clean fuel that only produces water when used. By connecting advanced treatment technologies, the authors show that wastewater could supply a sizable fraction of the world’s hydrogen without adding carbon pollution.
From Problem Waste to Useful Ammonia
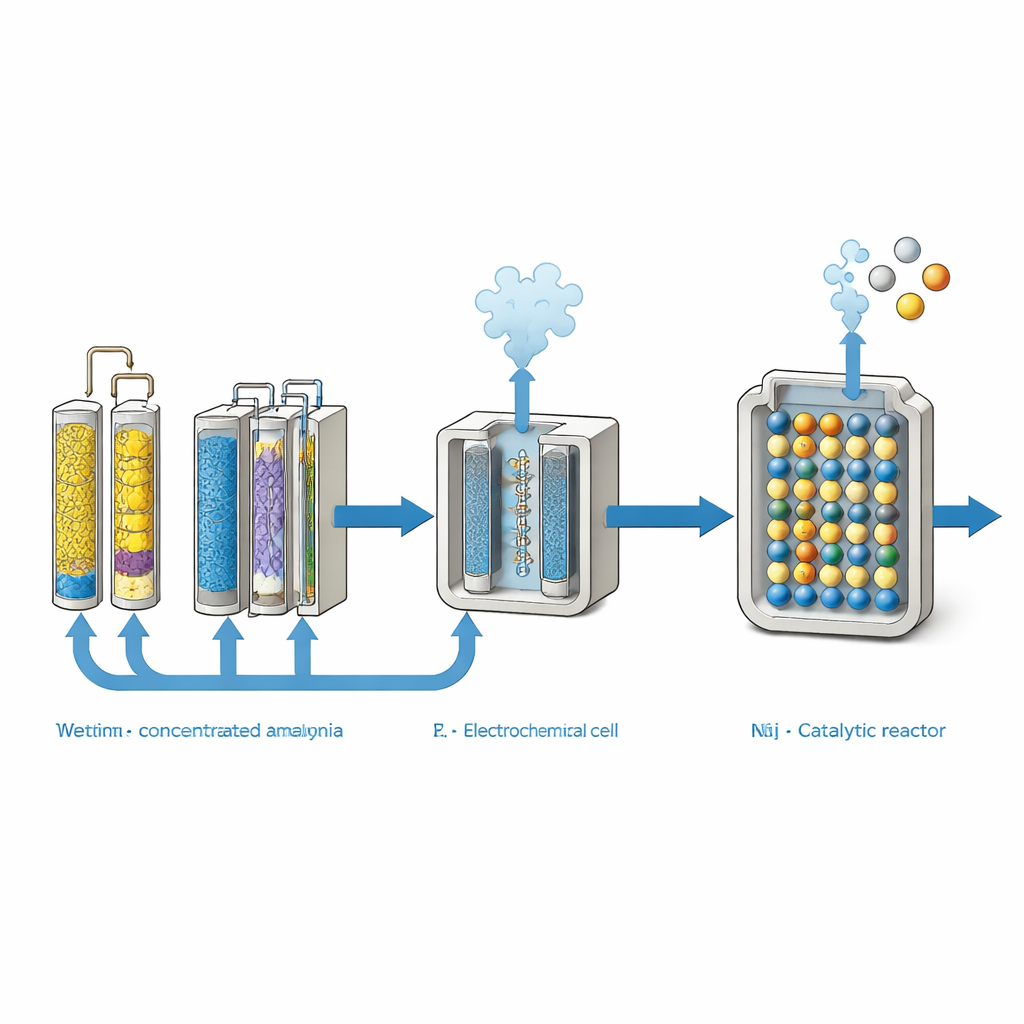
Modern wastewater treatment plants are designed to protect rivers and lakes from nutrient overload by converting reactive nitrogen into harmless nitrogen gas. That approach works for pollution control, but it wastes the energy and fertilizer value locked in nitrogen. The paper examines three existing techniques that can do something smarter: recover nitrogen from real waste streams in a form that can be reused. Gas stripping uses heat and high pH to drive dissolved ammonium into ammonia gas, which is captured in an acidic liquid. Membrane dialysis passes ammonia across special gas‑permeable barriers while keeping most other impurities behind. Electrodialysis relies on an electric field and ion‑selective membranes to pull ammonium into a concentrated stream. By re‑analyzing many published experiments on real wastewaters with a common yardstick, the authors compare how much ammonia each pathway can actually recover per liter of wastewater.
Which Recovery Routes Work Best
When all the data are normalized, gas stripping emerges as the most efficient at pulling ammonium out of typical wastewaters, often recovering more than 90 percent of the nitrogen. However, its performance drops sharply as nitrogen levels get very high, because extra chemicals are needed and other dissolved salts interfere. Electrodialysis performs well but can suffer from competition with other charged ions and from mineral scaling on the membranes. Membrane dialysis shows a different strength: it remains highly effective even when nitrogen concentrations are extreme, thanks to the selective transport of ammonia gas across hydrophobic membranes. In the most demanding cases, membrane systems achieved the largest total amounts of recovered ammonia per liter of wastewater, making them especially attractive for livestock manure, landfill leachate, and concentrated industrial effluents.
Cracking Ammonia into Hydrogen
Capturing ammonia is only half the story. To turn it into usable fuel, the ammonia must be decomposed into hydrogen and nitrogen over a solid catalyst at high temperature. The authors comb through recent catalyst research and identify three main families: those based on the precious metal ruthenium, those based on cheaper metals such as nickel, and bimetallic alloys that combine different metals. Ruthenium catalysts stand out for reaching nearly complete ammonia conversion at lower temperatures around 500 °C, which cuts energy use and extends catalyst life. Nickel and alloy catalysts can also perform well but typically require hotter operation, increasing fuel consumption. Importantly, ammonia recovered through the electrochemical pathways is essentially free of sulfur, chlorine, and heavy metals, meaning it behaves like high‑purity commercial ammonia and is unlikely to poison these catalysts.
How Much Hydrogen Could Wastewater Provide?
By linking the best recovery and decomposition steps into a three‑stage chain—nitrogen capture as ammonium, electrochemical conversion to ammonia gas, and catalytic cracking to hydrogen—the study estimates how much hydrogen could, in theory, be generated from global wastewater flows. Depending on the type of wastewater and the technology combination, each liter can yield from roughly a tenth of a gram to more than a gram of hydrogen. Scaled up to worldwide municipal, domestic, livestock, food‑processing, and some industrial streams, this adds up to between 2.5 and 30.6 million tons of hydrogen per year. That is equivalent to about 44 percent of current global hydrogen production, achieved without burning fossil fuels and while improving wastewater treatment.
Weighing Costs and Environmental Benefits
The researchers also compare this new route with the long‑established Haber–Bosch process, which makes synthetic ammonia from natural gas and is responsible for most of the world’s fertilizer. In pure energy terms, recovering ammonia from wastewater and then cracking it to hydrogen still costs slightly more than conventional “gray” ammonia, but it is already in the same range as “blue” ammonia, which captures some of its carbon dioxide, and is cheaper than “green” ammonia produced from renewable electricity alone. When greenhouse gas emissions are counted, the wastewater‑based pathways look even better. With today’s electricity mix they can beat gray ammonia, and when powered by low‑carbon sources such as solar, membrane and electrodialysis routes can even outperform green ammonia in climate impact per kilogram of product.
What This Means for a Hydrogen Future
Seen as a whole, the work shows that nitrogen in wastewater is not just a disposal problem but a strategic resource. By choosing the right recovery process for each type of waste stream—often membrane dialysis for very strong liquids—and pairing it with efficient ruthenium‑based reactors, it becomes possible to produce large amounts of carbon‑free hydrogen while also reclaiming fertilizer. Some hurdles remain, including scaling up electrodialysis and electrochemical steps, managing impurities in full‑scale plants, and reducing the cost and scarcity concerns around ruthenium. Even so, the analysis suggests that with thoughtful engineering and renewable power, tomorrow’s treatment plants could double as clean‑energy refineries, turning what we flush away into a significant slice of the world’s hydrogen supply.
Citation: Yang, H., Lim, S.Y., Lee, G. et al. Hydrogen production from wastewater via ammonia gas recovery. npj Clean Water 9, 25 (2026). https://doi.org/10.1038/s41545-026-00558-7
Keywords: wastewater hydrogen, ammonia recovery, membrane dialysis, electrodialysis, ruthenium catalysts