Clear Sky Science · en
Live-vectored antigen cocktail confers protection against African swine fever virus (ASFV) Georgia 2007/1 challenge
Why a Pig Disease Matters to All of Us
African swine fever is a deadly virus in pigs that has swept across much of the world, wiping out herds and driving up pork prices. Because there is still no widely available safe vaccine, farmers must rely on drastic measures like mass culling to stop outbreaks. This study describes a new type of experimental vaccine that uses a harmless carrier virus to deliver a large collection of African swine fever virus pieces into pigs. The goal is to train the pigs’ immune systems to recognize and defeat the real virus before it can cause disease.
A Silent Killer in the Pig Barn
African swine fever virus causes a fast-moving hemorrhagic disease in domestic pigs and wild boar, often killing nearly every infected animal. The virus can spread through direct contact, contaminated feed, or pork products, and it survives well in the environment and in frozen meat. As the virus has expanded from Africa into Europe and Asia, it has become one of the greatest threats to global pork production. Traditional approaches to vaccination, such as weakening the virus and using it as a live vaccine, can sometimes protect pigs but carry serious risks. Weakened strains may revert to dangerous forms or recombine with other strains, and there is no easy way to distinguish vaccinated animals from those naturally infected.
A New Vaccine Built from Viral Building Blocks
To skirt these dangers, the researchers designed a vaccine that never exposes pigs to the full African swine fever virus. Instead, they broke the virus down into dozens of individual proteins and grouped them into 43 genetic “cassettes.” Each cassette was inserted into a modified adenovirus, a different virus that can safely infect pig cells. These adenoviruses are engineered to replicate only in a controlled way and to manufacture several African swine fever proteins inside pig cells. When a pig is injected with a cocktail containing all 43 constructs, its cells briefly behave as factories, producing many different viral fragments that the immune system can learn to recognize.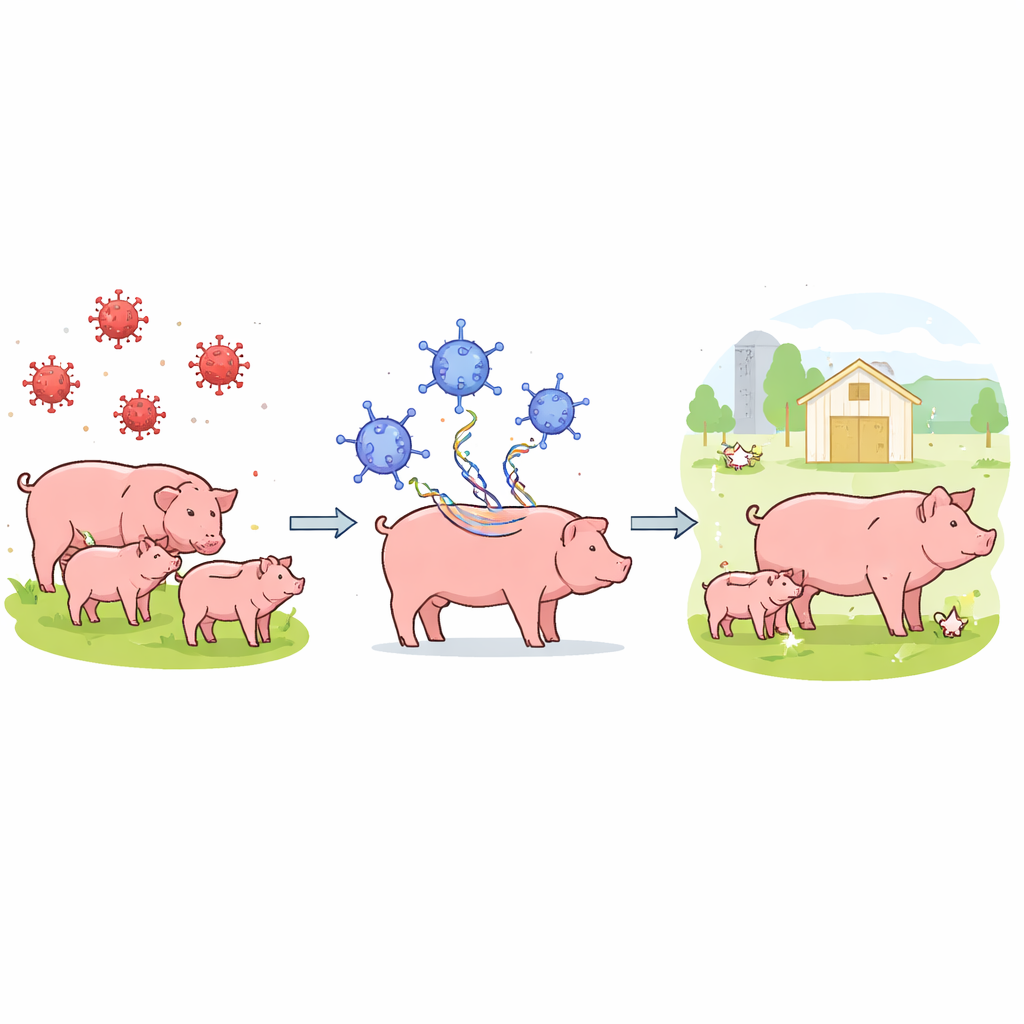
Putting the Cocktail to the Test in Pigs
The team vaccinated young pigs three times with this adenovirus cocktail, either alone or mixed with a commercial booster substance known as Quil-A. A separate control group received only an adenovirus that made a green fluorescent protein, not viral fragments. After the vaccinations, all pigs were housed with a pair of “spreader” pigs deliberately infected with a highly lethal strain called Georgia 2007/1. This setup mimicked natural barnyard transmission instead of a high-dose laboratory injection. In the group that received the vaccine without Quil-A, five out of six pigs survived the challenge, showed only mild or no signs of illness, and continued to gain weight. In contrast, every pig that received the Quil-A–boosted versions of the vaccine, as well as all control animals, developed severe disease and had to be euthanized.
What Protected the Survivors
Detailed examinations after death revealed stark differences between survivors and non-survivors. Pigs that died had enlarged, dark spleens; swollen, blood-filled lymph nodes; and widespread tissue damage typical of acute African swine fever. The survivors that received the non-adjuvanted vaccine showed almost none of these changes, and laboratory tests detected no trace of viral genetic material or live virus in their organs by the end of the study. Surprisingly, although vaccinated pigs produced strong antibodies that recognized infected cells, these antibodies did not neutralize the virus in cell culture. Instead, the best clue to protection came from killer T cells. Survivors had T cells loaded with destructive molecules such as granzyme B and perforin that responded strongly to specific African swine fever protein fragments. These cells are well suited to hunt down and destroy infected cells, suggesting that cellular rather than antibody immunity was the main line of defense.
Why the Helper Additive Backfired
One of the most unexpected findings was that pigs receiving the vaccine mixed with Quil-A fared worse, even though their antibody levels looked similar to those of the survivors. The authors suggest that this adjuvant, while useful in many other veterinary vaccines, may interfere with the live viral carriers used here—possibly by damaging the particles or skewing the immune response in an unhelpful way. As a result, these pigs did not build the robust T cell responses needed for protection and developed severe disease similar to unvaccinated animals.
What This Means for Future Pig Vaccines
This study shows that a carefully designed cocktail of live viral carriers encoding many African swine fever proteins can protect most pigs from a realistic, lethal exposure to a major field strain, without leaving them chronically infected. The work points toward a future vaccine that emphasizes strong killer T cell responses rather than virus-blocking antibodies and highlights that not all adjuvants are helpful when live vectors are involved. Although many questions remain—such as which of the 43 components are truly essential and whether the approach works against other strains—this live-vectored antigen cocktail offers a promising blueprint for safer, more effective control of African swine fever.
Citation: Kumar, R., Kim, T., Zajac, M.D. et al. Live-vectored antigen cocktail confers protection against African swine fever virus (ASFV) Georgia 2007/1 challenge. npj Vaccines 11, 66 (2026). https://doi.org/10.1038/s41541-026-01399-8
Keywords: African swine fever, pig vaccines, adenovirus vector, T cell immunity, livestock disease