Clear Sky Science · en
Functional antibody signatures following therapeutic immunization in Simian and Human immunodeficiency virus infection
Why this research matters
For people living with HIV, today’s medicines can keep the virus in check, but they rarely allow treatment to stop. This study explores whether therapeutic vaccines can train the immune system—especially antibodies—to help control the virus on their own, bringing us closer to a long‑term remission or even a functional cure without lifelong daily pills.
A new look at HIV vaccines
The researchers tested HIV "mosaic" vaccines originally designed to prevent infection, repurposing them for people and monkeys who were already infected but had their virus suppressed by standard drugs. These vaccines use harmless carrier viruses (Ad26 and MVA) to deliver carefully designed HIV proteins meant to cover many viral strains. The team focused not just on whether the vaccines raised antibody levels, but on what those antibodies could actually do once they latched onto HIV‑infected cells.
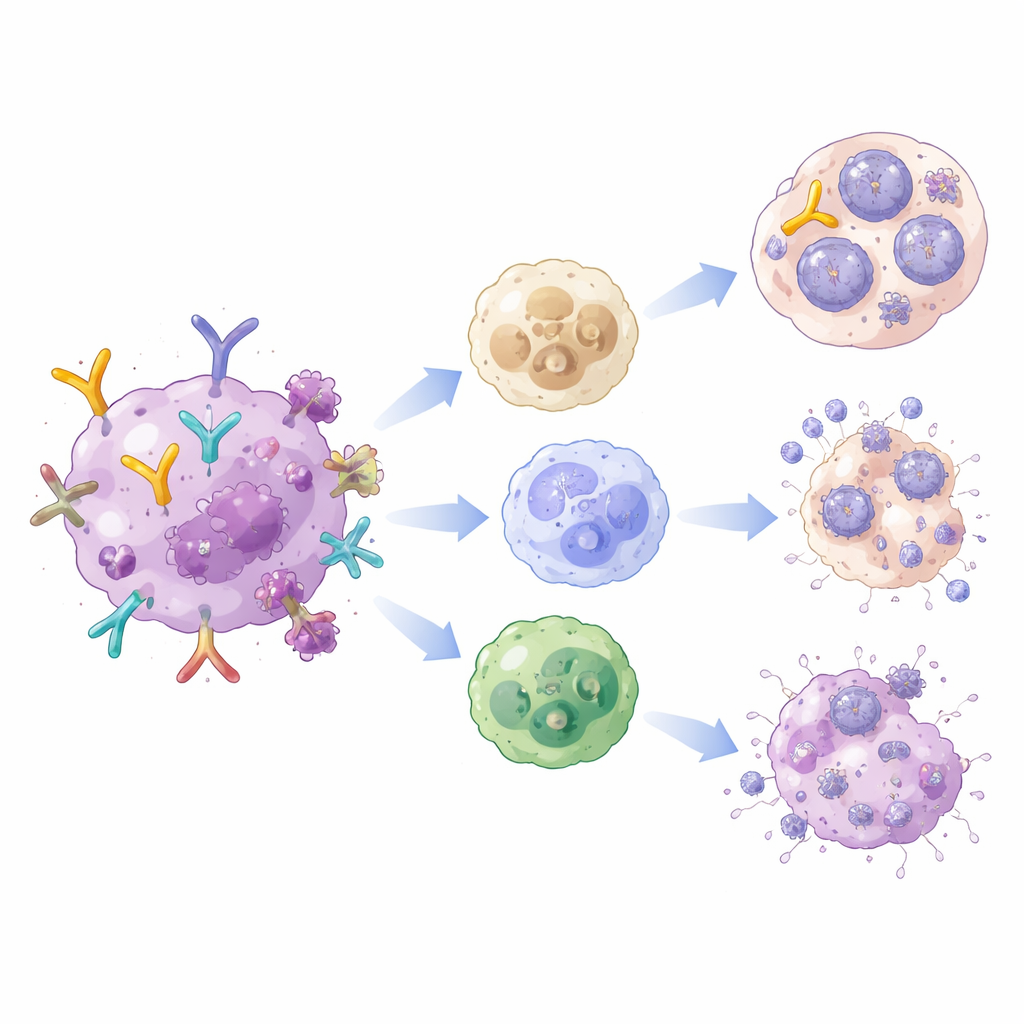
Antibodies that do more than block
Antibodies are often thought of as simple blockers, sticking to viruses and preventing them from entering cells. But their "tail" region can also call in backup from other parts of the immune system. In vaccinated rhesus macaques infected with a hybrid simian–human immunodeficiency virus, antibody levels against HIV’s outer coat protein rose sharply after vaccination. These antibodies bound better to special receptors on immune cells and triggered a suite of helpful actions: marking targets for destruction, drawing in scavenger cells that engulf viral particles, and activating natural killer cells that can kill infected cells.
Short‑lived power, lasting impact
The most powerful antibody responses in monkeys appeared after booster shots and then gradually faded over months. Even so, vaccinated animals kept higher levels of functional antibody activity than unvaccinated controls. When drug treatment was later paused, these antibody features did not reliably predict if or when the virus would first come roaring back. However, animals whose antibodies showed stronger functional signatures tended to settle at lower long‑term viral levels after the initial rebound, suggesting that such responses can help keep the virus more contained once it returns.
Partnering vaccines with engineered antibodies
Some monkeys also received PGT121, a laboratory‑made broadly neutralizing antibody. Animals given both the vaccine and PGT121 were most likely to maintain strong control of the virus after stopping standard drugs. Measurements taken while PGT121 was present showed that better engagement of antibody tail receptors was linked with a longer delay before the virus resurfaced. This pattern hints that combining therapeutic vaccination with potent engineered antibodies could shrink or weaken the viral reservoir in ways that pay off later, even after these extra antibodies are gone.
What this means for people with HIV
The team ran a parallel trial in people living with HIV whose virus was already suppressed by therapy. Here, vaccine regimens that paired the Ad26 shots with an added HIV protein boost most clearly increased functional antibodies, while Ad26 and MVA alone had more modest effects. Because the human study did not interrupt treatment, it could not show whether these responses translate into better viral control. Still, across both monkeys and people, the message is similar: the right therapeutic vaccines can awaken antibodies that not only recognize HIV, but also recruit the body’s own cells to attack infected targets. Harnessing these multi‑purpose antibody responses—especially in combination with other immune‑based tools—may be an important part of future strategies aiming for durable HIV control with less dependence on daily antiretroviral drugs.
Citation: Ilan, S., Bartsch, Y., Jung, W. et al. Functional antibody signatures following therapeutic immunization in Simian and Human immunodeficiency virus infection. npj Vaccines 11, 62 (2026). https://doi.org/10.1038/s41541-026-01390-3
Keywords: HIV therapeutic vaccines, functional antibodies, Fc effector functions, viral rebound control, broadly neutralizing antibodies