Clear Sky Science · en
Refined phenotyping of vaccine responses reveals transcriptomic determinants of neutralizing antibody heterogeneity
Why some vaccines work better for certain people
When we roll up our sleeves for a COVID-19 shot, we might assume everyone’s immune system reacts in the same way. In reality, people vary widely in how quickly and strongly they build protective antibodies. This study followed adults who received inactivated SARS-CoV-2 vaccines and shows that vaccine responses fall into distinct patterns, each driven by different early immune signals written in our genes and blood cells. Understanding these patterns could one day help tailor vaccination schedules so that everyone gets the protection they need.
Three paths to protection after the same shot
The researchers tracked 73 healthy adults who received two doses of an inactivated COVID-19 vaccine. They measured neutralizing antibodies—those that can block the virus—before vaccination, one week after the second dose, and again about a month later. Instead of simply splitting people into “high” and “low” responders, they used data-driven methods to group people by how their antibody levels changed over time. Three clear patterns emerged: a “low-delayed” group with a slow, modest rise in antibodies; a “rapid-stabilizing” group whose antibodies spiked quickly then leveled off; and a “continuous-increase” group whose antibodies kept climbing to the highest levels. These patterns were not explained by age, sex, or body weight and only partly by which inactivated vaccine brand people received.
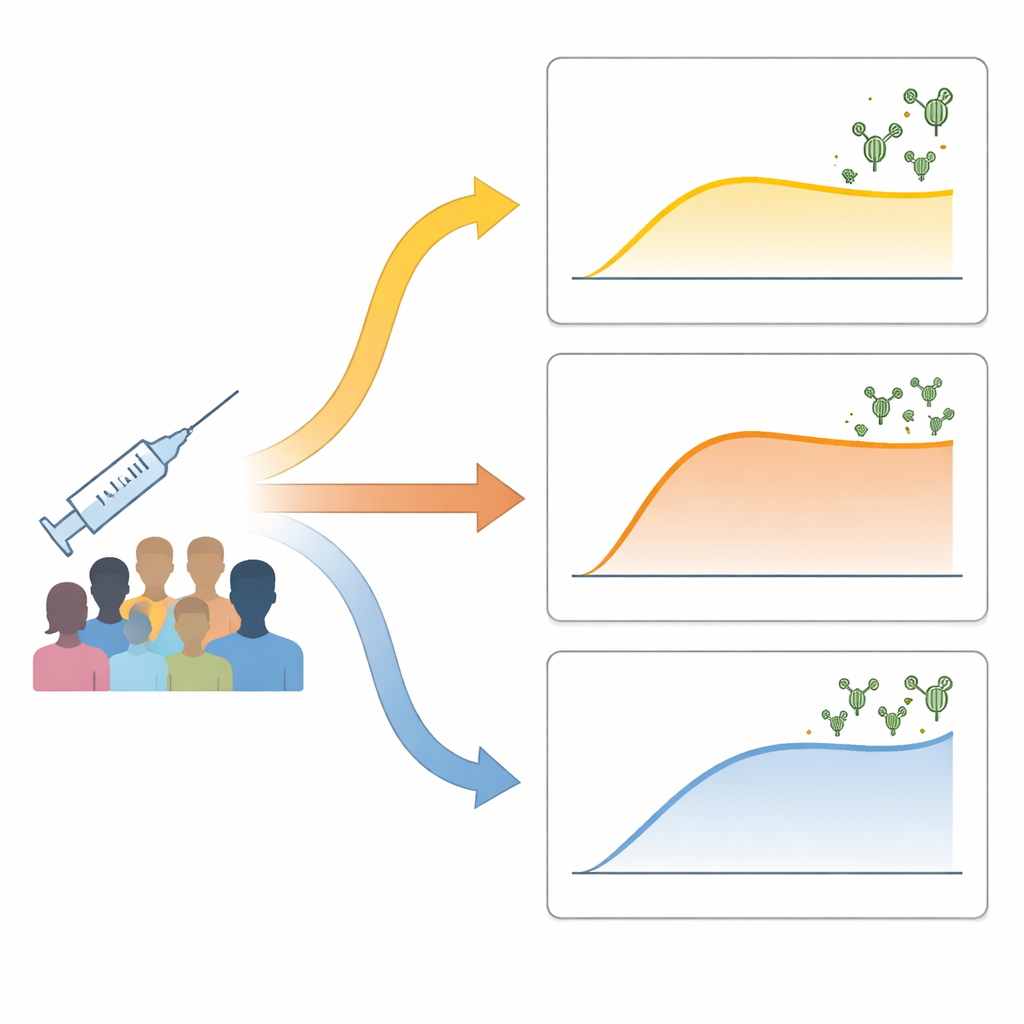
How early immune activity shapes the outcome
To learn what drives these different paths, the team analyzed the genetic activity of immune cells in the blood at the start and one week after the second shot. In the low-delayed group, very little changed: key immune pathways barely switched on, and the mix of immune cell types in the blood stayed largely the same. In contrast, the rapid-stabilizing and continuous-increase groups showed clear signs of early immune activation. In the strongest responders, there was coordinated activity in pathways that process viral fragments, present them to other immune cells, and support a type of helper T cell response that is known to boost antibody production. These people also tended to have more naïve B cells and helper T cells poised to respond, and later showed somewhat stronger virus-specific T cell activity.
Hidden layers of control in RNA and B cells
Beyond simple gene on–off changes, the scientists looked at more subtle layers of regulation embedded in RNA, the intermediate molecules between DNA and protein. In the top-responding group, immune genes involved in presenting viral pieces to the immune system showed different “splicing” patterns and shorter tail regions at the end of their RNA. These features are thought to make it easier for cells to produce certain proteins and maintain strong signaling. The team also reconstructed the repertoire of B cell receptors—the unique antennas on antibody-producing cells—from the same blood samples. While overall diversity looked similar across groups, each response pattern used slightly different combinations and shapes of these receptors, and the strongest responders favored receptor types previously linked to powerful antibodies against the coronavirus spike protein.
Spotting likely low responders before vaccination
Because people in the low-delayed group may be less protected after a standard vaccination schedule, the researchers asked whether it might be possible to flag them in advance. They focused on a small set of genes that differed modestly at baseline between the weakest and strongest responders. Using machine-learning methods, they built a model based on the activity of just eight genes measured before vaccination. This model could distinguish low-delayed responders with high accuracy in their dataset, with two genes involved in basic cell signaling and inflammation contributing most of the predictive power. Although this work needs to be tested in larger and more diverse groups, it suggests that a simple blood test could one day guide more personalized vaccine plans.
What this means for future vaccines
Overall, this study shows that people do not simply mount “strong” or “weak” antibody responses; they follow different time courses shaped by early, hard-to-see immune events. The most protected individuals turn on coordinated networks of genes and cells that help them recognize the vaccine quickly and build durable antibodies, while low responders show a muted early reaction. By mapping these internal differences and building tools to predict who will respond poorly, the work lays the groundwork for personalized vaccination—such as earlier boosters or alternative formulations for at-risk individuals—so that the promise of vaccines can be more evenly shared.
Citation: Wu, Q., Hu, H., Qin, L. et al. Refined phenotyping of vaccine responses reveals transcriptomic determinants of neutralizing antibody heterogeneity. npj Vaccines 11, 61 (2026). https://doi.org/10.1038/s41541-026-01386-z
Keywords: vaccine responses, neutralizing antibodies, COVID-19 vaccination, immune heterogeneity, personalized vaccines