Clear Sky Science · en
Antigen cross-presentation potentiating cancer vaccine adjuvant for T cell expansion and synergy with anti-PD-1
Turning the Immune System into a Precision Tumor Fighter
Cancer vaccines are often described as teaching the immune system to recognize and destroy tumors, but in practice they have rarely lived up to that promise. This study explores a new way to “supercharge” such vaccines so they can generate stronger, longer-lasting killer T cells and work better together with popular immunotherapy drugs like anti–PD‑1. For readers interested in how tomorrow’s cancer treatments may combine biologic drugs, vaccines, and immune engineering, this work offers a glimpse of what next‑generation, highly targeted immunotherapy could look like.
Why Cancer Vaccines Need Better Backup
Therapeutic cancer vaccines aim not to prevent disease, but to treat people who already have tumors by raising powerful, tumor‑specific immune responses. The key players are CD8 “killer” T cells, which can recognize and destroy cancer cells displaying telltale antigens. Unfortunately, tumor antigens are weakly stimulating because they mostly look like altered versions of the body’s own proteins. Continuous exposure during cancer development can also leave T cells exhausted or tolerant, blunting vaccine effects. To overcome this, vaccines must be paired with adjuvants—helper components that rouse the innate immune system, improve antigen presentation, and build a large, resilient pool of tumor‑specific T cells.
A Hybrid Immune Booster Built from Two Powerful Signals
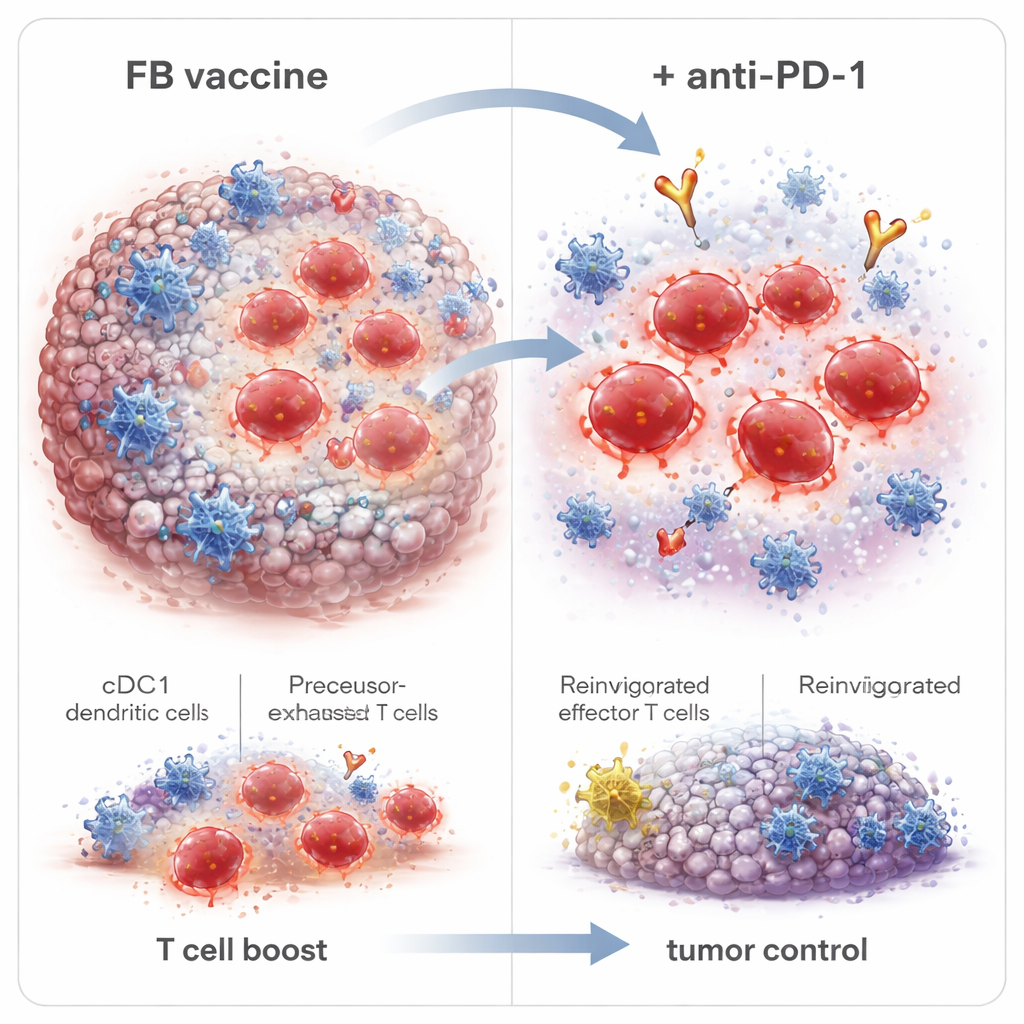
The researchers designed a hybrid adjuvant called Flt3L‑FlaB, or FB, by fusing two immune‑stimulating molecules into a single protein. One half, Flt3L, is a natural growth factor that expands a specialized group of dendritic cells known as cDC1. These cells excel at “cross‑presentation” – taking bits of tumor material and showing them to CD8 T cells in a way that triggers strong responses. The other half, FlaB, is a bacterial flagellin fragment that activates an innate sensor called TLR5 and acts as a potent danger signal. By linking them, the authors aimed to both expand cDC1 and switch them into a highly active state, improving how tumor antigens are shown to T cells and how long this stimulation lasts.
Putting the New Adjuvant to the Test in Tumor Models
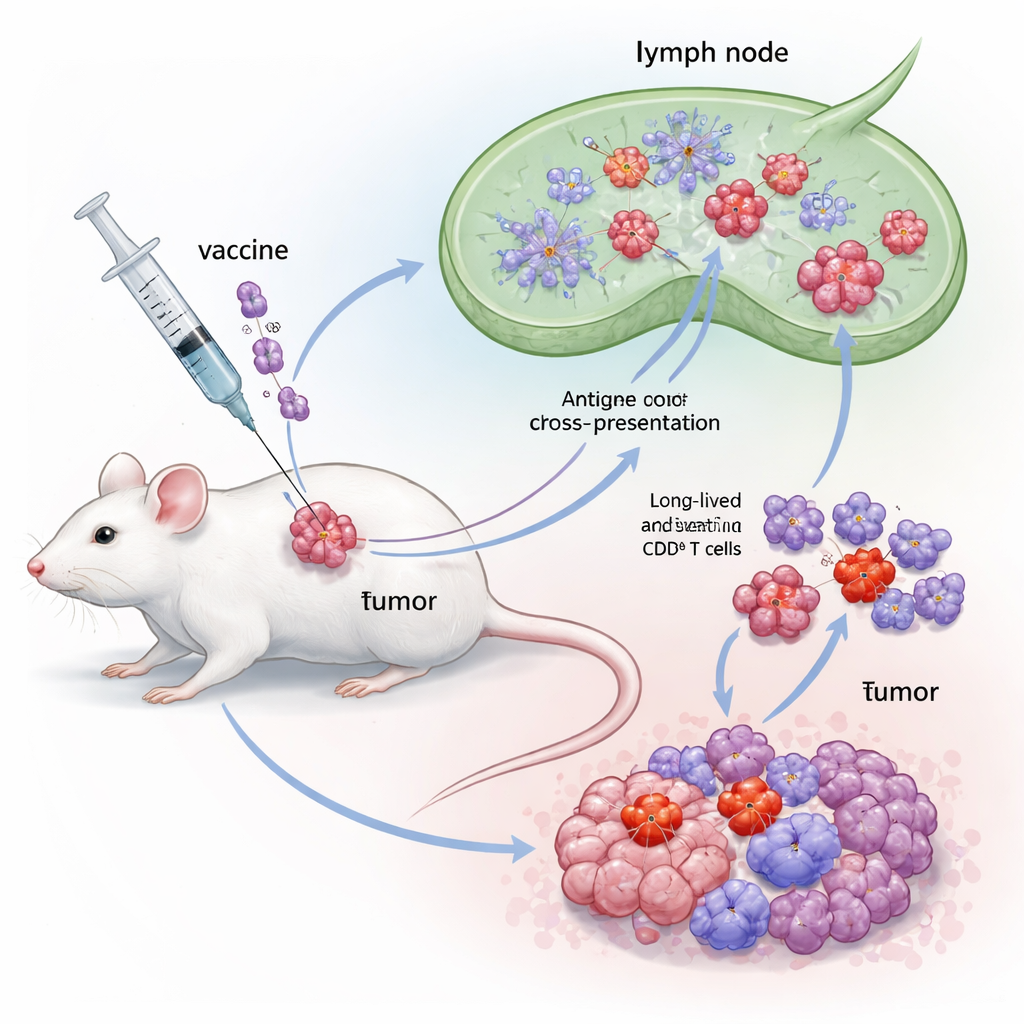
To test FB, the team used a well‑established mouse tumor model based on cells engineered to carry cancer‑causing proteins from human papillomavirus (HPV). Mice first received a small tumor implant and then a therapeutic vaccine containing an HPV E7 antigen, combined either with Flt3L alone, FlaB alone, or the new FB fusion. All vaccines were given near the tumor. While each single adjuvant slowed tumor growth somewhat, the FB‑based vaccine stood out: it delayed tumor progression far more, significantly extended survival, and left a notable fraction of mice completely tumor‑free. Immune analyses showed that FB vaccination generated unusually strong E7‑specific CD8 responses in blood, lymph nodes, and spleen, with high levels of interferon‑gamma and robust T‑cell proliferation—evidence that the fusion did more than simply add the effects of its two parts.
Building Lasting Immune Memory and a Better Partner for Anti–PD‑1
Digging deeper, the scientists found that FB changed not just how many CD8 T cells appeared, but what kinds. It boosted short‑lived effector cells that attack tumors immediately, as well as memory precursor and stem‑like memory cells that can persist and respond again in the future. Importantly, FB also expanded a group called precursor‑exhausted T cells (Tpex) within tumors—cells that sit at the boundary between exhaustion and full activity and are particularly responsive to anti–PD‑1 drugs. When tumor‑free survivors were challenged with higher doses of cancer cells long after vaccination, most resisted regrowth, showing durable immune memory. Blocking immune‑cell exit from lymph nodes erased FB’s benefits, indicating that tumor‑killing T cells were primed in those nodes by cDC1 and then migrated into the tumor.
Stronger Together: Vaccines Plus Checkpoint Blockade
Because anti–PD‑1 therapy works best when there is already a pool of responsive T cells, the team combined FB‑adjuvanted vaccination with an anti–PD‑1 antibody. In the HPV tumor model, adding anti–PD‑1 on top of FB vaccination further slowed tumor growth, increased the number of mice that fully cleared their cancers, and provided complete protection against an extremely high‑dose tumor rechallenge. Fine‑tuning the timing mattered: starting anti–PD‑1 after the second rather than the first vaccine dose modestly improved complete tumor eradication, consistent with waiting until Tpex cells had expanded. The FB adjuvant also improved responses to peptide‑based vaccines, including tumor‑specific neoantigens in a colon cancer model, suggesting that this strategy could apply broadly beyond HPV‑related disease.
What This Could Mean for Future Cancer Care
For non‑specialists, the key message is that this work refines how to “teach” the immune system to fight cancer. By combining a dendritic‑cell growth factor with a strong danger signal in a single, stable protein, the FB adjuvant creates more of the right antigen‑presenting cells, helps them show tumor antigens more effectively, and shapes a population of killer T cells that are both powerful now and poised to respond to later checkpoint therapy. While these results are in mice and many steps remain before use in people, the study outlines a clear roadmap: smarter adjuvants that orchestrate cross‑presentation and T‑cell stemness could make therapeutic cancer vaccines a much stronger partner for drugs like anti–PD‑1, potentially leading to deeper, longer‑lasting tumor control in patients.
Citation: Dang, G.C., Loeurng, V., Pa, P. et al. Antigen cross-presentation potentiating cancer vaccine adjuvant for T cell expansion and synergy with anti-PD-1. npj Vaccines 11, 56 (2026). https://doi.org/10.1038/s41541-026-01376-1
Keywords: cancer vaccines, immunotherapy, dendritic cells, PD-1 blockade, HPV-related tumors