Clear Sky Science · en
Universal broad-spectrum mucosal vaccine design for human coronaviruses inspired by artificial antibodies
Why a Better Coronavirus Vaccine Still Matters
Even as COVID-19 shifts toward a seasonal infection, waves of new variants continue to appear, and several other human coronaviruses still cause serious disease. Current shots do a good job of preventing severe illness but are less effective at blocking infection and transmission, especially in the nose and throat where these viruses first take hold. This study describes a new kind of intranasal vaccine, inspired by engineered antibodies, that aims to give long-lasting, broad protection across many coronaviruses at the body’s mucosal front line.
Building a Smarter Vaccine Scaffold
The researchers started from a familiar molecule: human IgG1, the main antibody type used in many approved antibody drugs. They redesigned its heavy chain so it could serve as a flexible “scaffold” for presenting coronavirus spike protein receptor-binding domains (RBDs)—the parts the virus uses to grab our cells. By removing the usual light chain and adding specific mutations, they reduced binding to immune receptors linked to side effects such as cytokine storms, while strengthening binding to the neonatal Fc receptor (FcRn). FcRn is abundant on mucosal surfaces and helps carry antibodies across the barrier and keep them around longer. Onto this optimized backbone, the team attached one, two, three, or four different RBDs from SARS, MERS, seasonal coronaviruses, and multiple SARS‑CoV‑2 variants, including Omicron lineages, creating mono-, bi-, tri-, and tetravalent vaccine candidates.
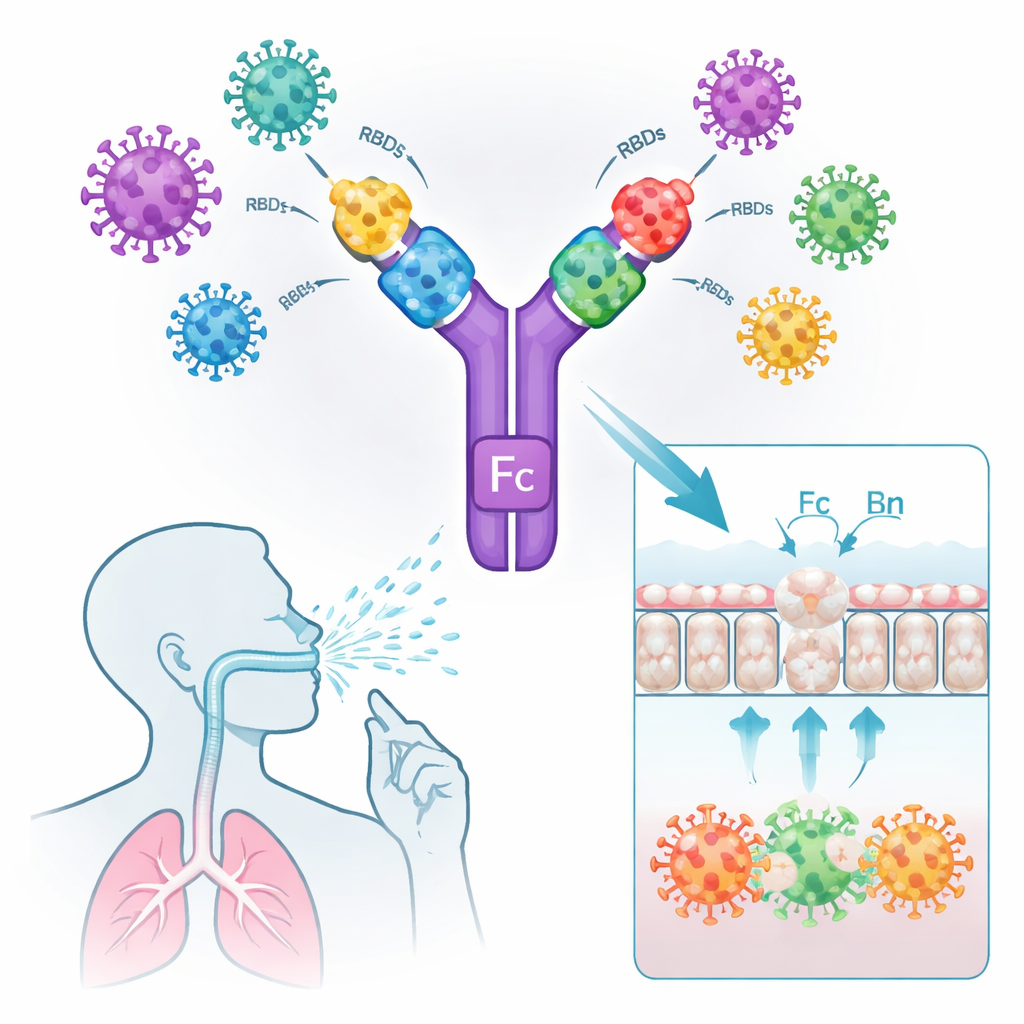
Crossing the Mucosal Barrier and Waking Up Local Defenses
To work as a nasal spray, the vaccine antigens must reach and linger at the airway lining and be efficiently noticed by immune cells. In cell culture systems, the Fc-bearing constructs bound strongly both to ACE2 (the SARS‑CoV‑2 entry receptor) and to human or mouse FcRn, and were actively transported across a layer of mucosal cells. A similar RBD dimer lacking the Fc portion failed to cross. When fluorescent versions of the vaccines were delivered into the noses of mice, the Fc-containing antigens remained in the lung mucosa for hours, while the bare RBD dimer quickly disappeared. Microscopy showed that the Fc-modified vaccines attracted and activated antigen-presenting cells just below the mucosal surface, as indicated by higher levels of the immune marker CD80 and colocalization with FcRn.
Boosting Antibodies and T Cells Where They Are Needed
The team then compared immune responses after intramuscular injection versus intranasal delivery in mice. Across several formulations, the Fc-based vaccines triggered very high levels of IgG antibodies in the blood against both the original SARS‑CoV‑2 strain and Omicron XBB.1.5. Intranasal dosing, especially when combined with a modern STING-agonist adjuvant (CF501), stood out for generating strong IgA responses not only in the blood but also in nasal secretions and lung washings—precisely where mucosal protection is most important. The Fc-bearing constructs also drove more powerful cellular responses than bare RBD dimers, expanding helper T cells, killer T cells, and long-lived tissue-resident memory T cells in the lungs. These cell types are thought to be crucial for clearing respiratory viruses and preventing spread to sensitive organs such as the brain.
Broad Protection Against Many Variants and Species
A central test was whether a bivalent vaccine combining RBDs from the prototype virus and Omicron XBB.1.5 (called XBB.1.5 + PT‑Fc) could protect animals from multiple viral challenges. In human ACE2–expressing mice given two low intranasal doses, this candidate induced strong neutralizing antibodies and completely shielded animals from lethal infection with both the original virus and XBB.1.16: vaccinated mice maintained weight, survived, and had no detectable virus in brain, lungs, or nasal tissues, while control animals became sick and died. The same vaccine blocked disease and death when mice were challenged with other major variants including Beta, Delta, and several Omicron sublineages. In hamsters, which are well suited to transmission studies, vaccinated animals had dramatically lower viral loads after direct challenge, and, in co-housing experiments, were largely protected from airborne transmission. Antibody levels and memory T cells in both mice and hamsters remained high for at least 48 weeks, and animals were still fully protected from lethal challenges nearly a year after vaccination.
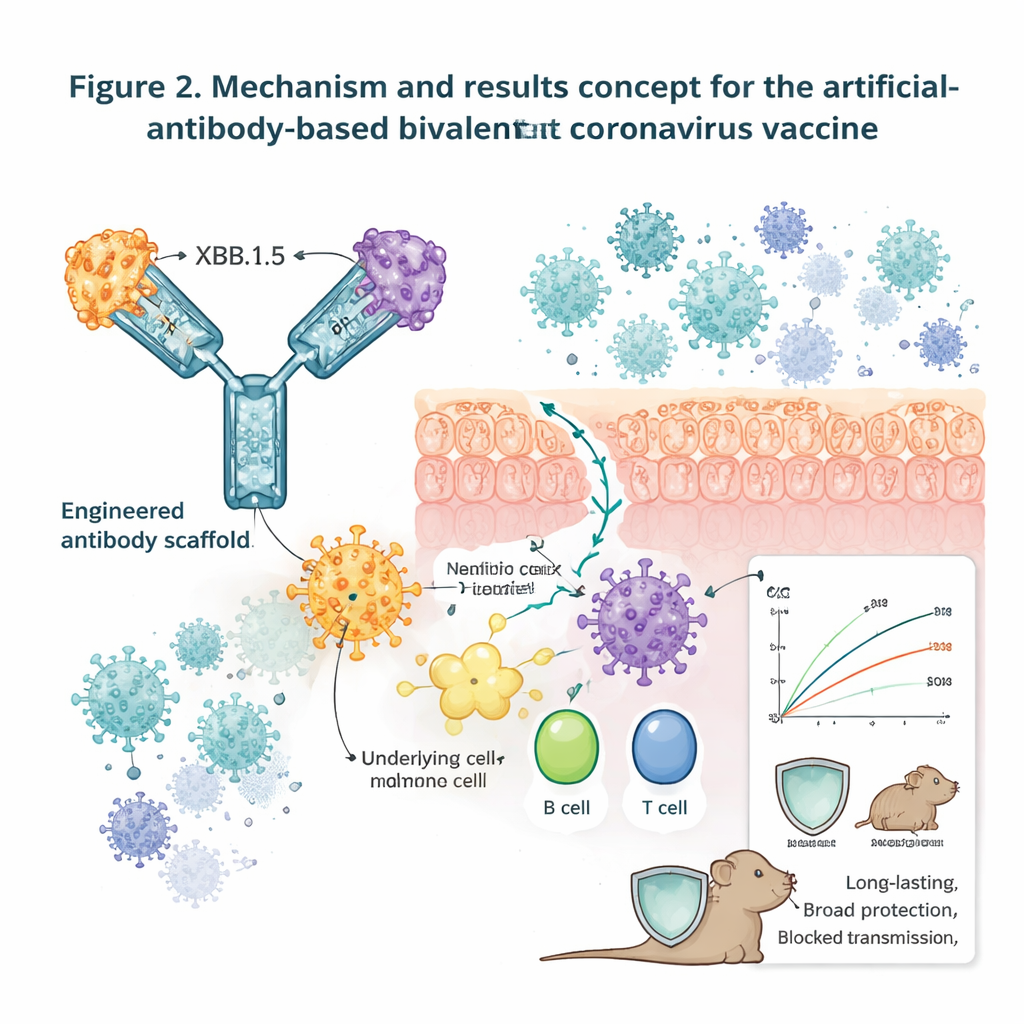
Toward a Universal Coronavirus Nasal Spray
Finally, the researchers pushed the platform toward a truly universal design. They mixed several bivalent constructs covering SARS‑CoV‑2, SARS, MERS, and multiple seasonal coronaviruses, and also built tetravalent molecules carrying four distinct RBDs or spike fragments on a single scaffold. These combinations raised antibodies that recognized a wide panel of human coronaviruses and, in mouse challenge experiments, reduced viral loads for several representatives, including an endemic virus (NL63). Although further testing in models closer to humans and eventually in clinical trials is needed, the work shows that an engineered antibody backbone can safely ferry diverse coronavirus antigens across the nasal barrier, stimulate strong local and systemic immunity, and provide broad, durable protection. For non-specialists, the message is that future “universal” coronavirus boosters may not be injections at all, but gentle nasal sprays built on the same antibody frameworks already proven safe in many medicines.
Citation: Wu, Y., Lu, J., Fang, L. et al. Universal broad-spectrum mucosal vaccine design for human coronaviruses inspired by artificial antibodies. npj Vaccines 11, 55 (2026). https://doi.org/10.1038/s41541-026-01375-2
Keywords: intranasal vaccine, broad-spectrum coronavirus, mucosal immunity, artificial antibody scaffold, SARS-CoV-2 variants