Clear Sky Science · en
Association between COVID-19 vaccine efficacy and epidemic force of infection
Why how well vaccines work can still surprise us
COVID-19 vaccines were hailed as game changers, but people quickly noticed that the same vaccine did not always seem to work equally well everywhere. Some trials and real‑world studies reported very high protection; others, run in different places or at different times, saw more infections in vaccinated people. This article asks a deceptively simple question: how much does the intensity of an outbreak in a community—the “infection pressure” around you—shape how effective a vaccine appears to be?
Measuring the "pressure" of an outbreak
To tackle this question, the researchers focused on the force of infection, a term that captures how often susceptible people in a community become infected. Instead of relying only on what happened inside vaccine trials, they linked three large phase 3 COVID-19 vaccine studies—Moderna’s mRNA-1273 (COVE), AstraZeneca’s ChAdOx1 nCoV-19 (AZD1222), and Janssen’s Ad26.COV2.S (ENSEMBLE)—to independent surveillance estimates of how widely SARS‑CoV‑2 was spreading in each trial location over time. These surveillance data, drawn from hospitalizations, deaths, and antibody surveys, provided a day‑by‑day picture of local infection levels from the start of the pandemic through late 2021. 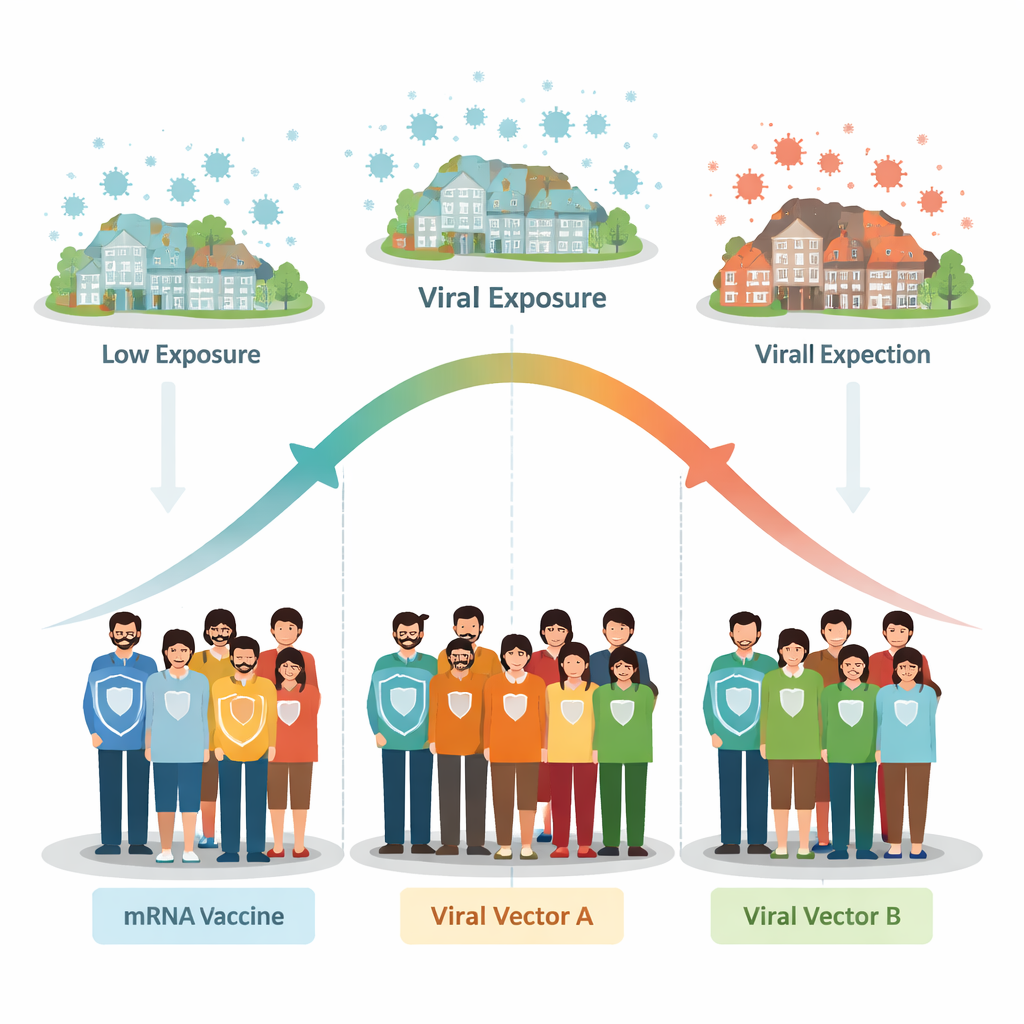
What the three vaccine trials revealed
The picture that emerged was not a single simple rule, but a pattern that shifted with vaccine type and exposure level. In the U.S. subgroup of the AstraZeneca trial, higher infection pressure was linked to higher measured vaccine efficacy: as exposure increased within the generally low‑to‑moderate U.S. setting for that trial, infection risk rose more steeply in the placebo group than in the vaccinated group. In contrast, when the researchers looked at the combined U.S. and non‑U.S. data from the Janssen trial, they saw the opposite tendency: places with higher infection levels tended to show lower vaccine efficacy, especially in regions outside the United States where community spread was intense. For the Moderna trial, the overall AstraZeneca trial, and the U.S. portion of the Janssen trial, the data did not show a clear link in either direction—vaccine performance looked relatively stable across the range of infection pressures that those particular participants experienced.
A possible "sweet spot" and "stress test" for vaccines
Putting the results together, the authors suggest that the relationship between vaccine efficacy and infection pressure may resemble an inverted U shape. At relatively low infection levels, small increases in exposure may reveal more of a vaccine’s benefit compared with placebo, making efficacy appear to rise. But at very high infection levels—where people may be exposed repeatedly or intensely—even good vaccines may be “stress‑tested,” leading to more breakthrough infections and lower apparent protection. 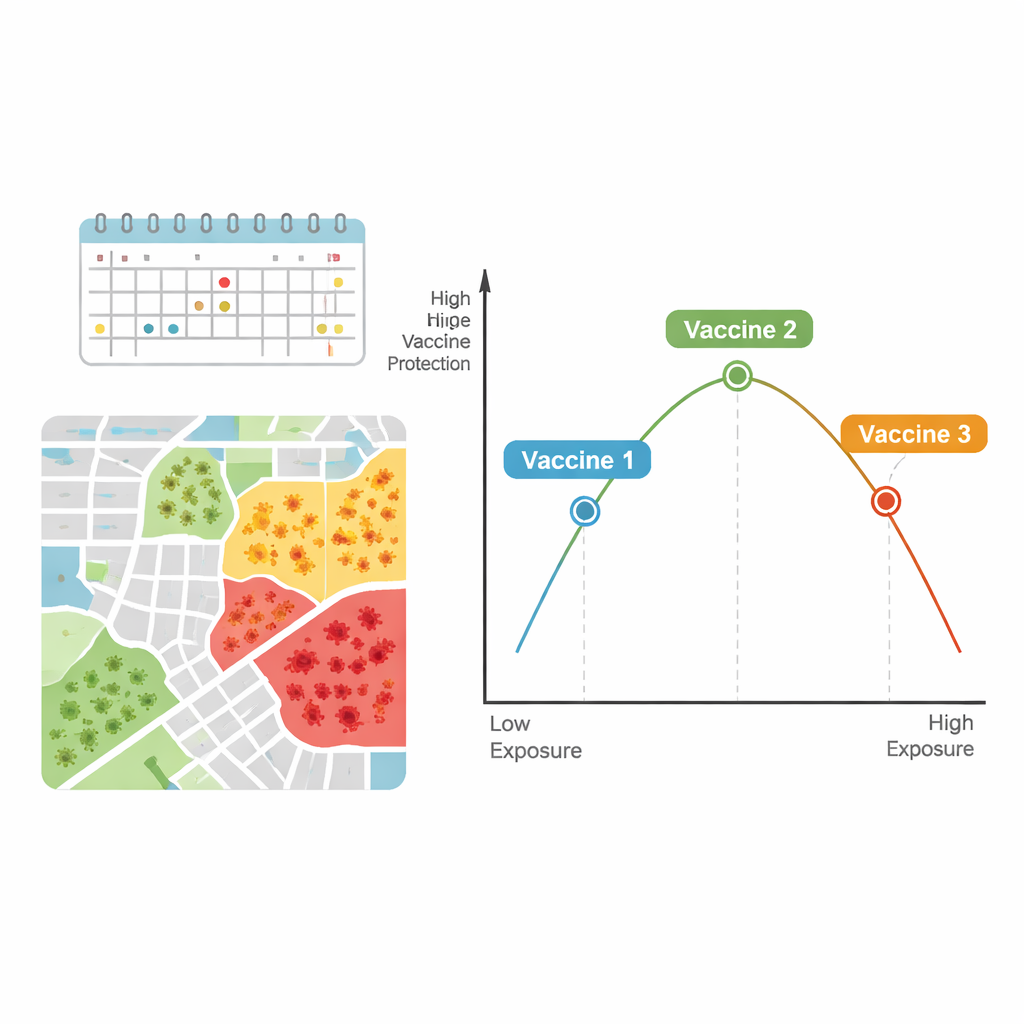
Limits and what they mean for real‑world decisions
The authors note that their work does not prove cause and effect, and that they were constrained by the range of infection levels that actually occurred during the trials and by not being able to fully separate variant effects or disease severity. Still, using person‑by‑person trial data linked to daily local infection estimates allowed them to move beyond earlier studies that treated each country or trial as a single, unchanging number. Their findings suggest that when we compare vaccines, or try to apply trial results to a new setting, we should pay close attention to how hard the virus is hitting that community, not just to the headline efficacy percentage. For trials conducted at a single site, ignoring local infection pressure may lead to misleading expectations about how a vaccine will perform elsewhere.
What this means for the next pandemic
In everyday terms, this article argues that how well a vaccine “works” is not just about the vaccine or the person receiving it; it is also about what is happening in the surrounding community. In moderate outbreaks, a vaccine’s strengths may be most clearly seen. In explosive outbreaks, even strong vaccines can look weaker simply because people are bombarded with more chances to get infected. Understanding this nuanced, possibly inverted U‑shaped relationship can help scientists design better trials, choose more informative trial sites, and help public health officials interpret vaccine data more wisely when deciding how to protect different regions in future epidemics and pandemics.
Citation: Xu, J., Halloran, M.E., Moore, M. et al. Association between COVID-19 vaccine efficacy and epidemic force of infection. npj Vaccines 11, 54 (2026). https://doi.org/10.1038/s41541-026-01374-3
Keywords: COVID-19 vaccines, vaccine efficacy, force of infection, epidemic dynamics, clinical trials