Clear Sky Science · en
Cost-effectiveness of maternal vaccine and/or monoclonal antibody strategies against respiratory syncytial virus in Belgian infants
Why this matters for parents and health planners
Every winter, a common virus called RSV (respiratory syncytial virus) sends thousands of young Belgian children to hospital. New tools now promise to shield babies during their most fragile first months of life: a vaccine given to pregnant women and long‑acting antibodies given directly to infants. This study asks a simple but crucial question for families and taxpayers alike: given their benefits and their high prices, which of these options gives the best health results for the money spent in Belgium?
The hidden weight of a winter virus
Before any new protection is added, RSV already causes a heavy burden in Belgian children under five. Following one year’s birth cohort over five years, the researchers estimate about 116,000 RSV episodes, including many mild infections at home and tens of thousands of doctor visits. Most serious problems occur in the very youngest: babies under three months face the highest chances of being hospitalised or needing intensive care. Each year, RSV is linked to around 8,600 regular hospital stays, more than 400 intensive care admissions, and about five deaths, costing the health system roughly €43 million and leading to nearly 1,000 lost years of healthy life when both illness and premature death are considered.
New ways to protect the smallest babies
Belgium has recently gained access to two new ways of preventing severe RSV in infants. A maternal vaccine is given during late pregnancy so that protective antibodies pass through the placenta to the baby, covering the first few months after birth. A long‑acting monoclonal antibody, nirsevimab, is given to the baby as a single injection and directly blocks the virus. The team compared several realistic programme designs: giving the maternal vaccine all year or only to pregnancies due in the RSV season, giving nirsevimab only to babies born in season or also to those born earlier through a large catch‑up round, and a combined approach that offers both tools in different circumstances. They assumed maternal vaccine would reach fewer families (40% coverage) than nirsevimab (90%), reflecting how pregnancy vaccines and infant shots are usually taken up in Belgium.
Health gains versus programme price tags
The broadest nirsevimab plan — protecting babies born in the RSV season and catching up those born just before it — prevented by far the most illness, avoiding almost 20,000 infections and gaining over 200 additional healthy life years in infants, while saving around €19 million in treatment costs. Seasonal maternal vaccination was less powerful, preventing just over 3,000 cases and gaining 47 healthy life years at the assumed 40% uptake. However, prevention programmes themselves are expensive. At today’s Belgian list prices (about €186 per maternal vaccine dose and almost €778 per nirsevimab dose), seasonal nirsevimab with catch‑up would cost roughly €76 million to deliver, compared with about €5 million for the seasonal maternal vaccine.
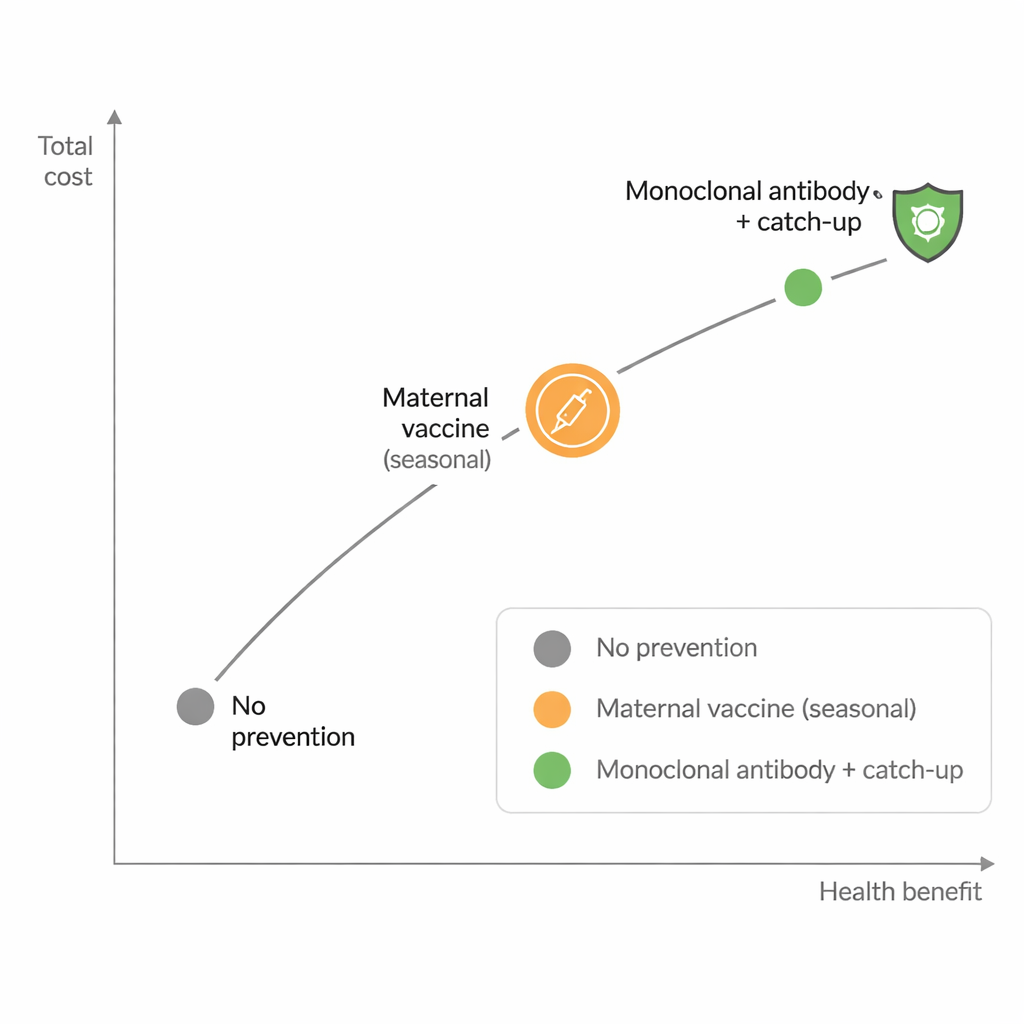
When is an option worth paying for?
To judge value for money, the authors used standard health‑economic methods that compare extra cost with extra health benefit, expressed as cost per additional healthy life year gained. Looking from the health payer’s point of view and using current list prices, only the seasonal maternal vaccination strategy stayed under common informal Belgian thresholds (below €50,000 per healthy life year). Nirsevimab strategies, despite being more effective, were far too costly per unit of health gained. When the researchers explored a scenario where both products would cost €200 per dose, the picture changed: seasonal nirsevimab, especially with catch‑up, became highly attractive and could even be close to cost‑saving, because the extra programme cost was offset by fewer hospitalisations. A two‑way price analysis showed exactly which combinations of maternal vaccine and nirsevimab prices would make each strategy the preferred choice at different willingness‑to‑pay levels.
What this means for future RSV protection
For now, the study suggests that, at public list prices, a targeted seasonal maternal vaccination programme is the only clearly cost‑effective option for Belgium, even though nirsevimab prevents more disease. If nirsevimab’s real purchase price is much lower than the list price, or if strong long‑term benefits such as reduced wheezing and asthma are confirmed, broad infant antibody programmes could become good value as well. The analysis also shows that results are very sensitive to how many RSV hospitalisations actually occur and how well each product prevents them. In practical terms, Belgium can safely say that protecting infants against RSV is worthwhile, but making the most of these new tools will depend on tough price negotiations, careful programme design, and ongoing monitoring of how often babies are hospitalised and how well the interventions work in real life.
Citation: Li, X., Willem, L., Roberfroid, D. et al. Cost-effectiveness of maternal vaccine and/or monoclonal antibody strategies against respiratory syncytial virus in Belgian infants. npj Vaccines 11, 52 (2026). https://doi.org/10.1038/s41541-026-01372-5
Keywords: respiratory syncytial virus, infant immunisation, maternal vaccination, monoclonal antibodies, health economics