Clear Sky Science · en
Heterologous immunization modulates B-cell epitope competition between helper peptides and the MPER segment in MPER/liposome vaccines
Why this HIV vaccine study matters
Designing an effective HIV vaccine has been extraordinarily difficult, in part because the virus mutates rapidly and hides its most vulnerable parts from the immune system. This study looks at a clever way to coax the body to focus on one such hidden weak spot, called the MPER region, by packaging it into tiny fat bubbles known as liposomes and carefully varying the “helper” components between shots. The work, done in mice, does not yet produce a working vaccine, but it reveals practical rules for steering immune responses toward rare, protective antibodies and away from distracting targets.
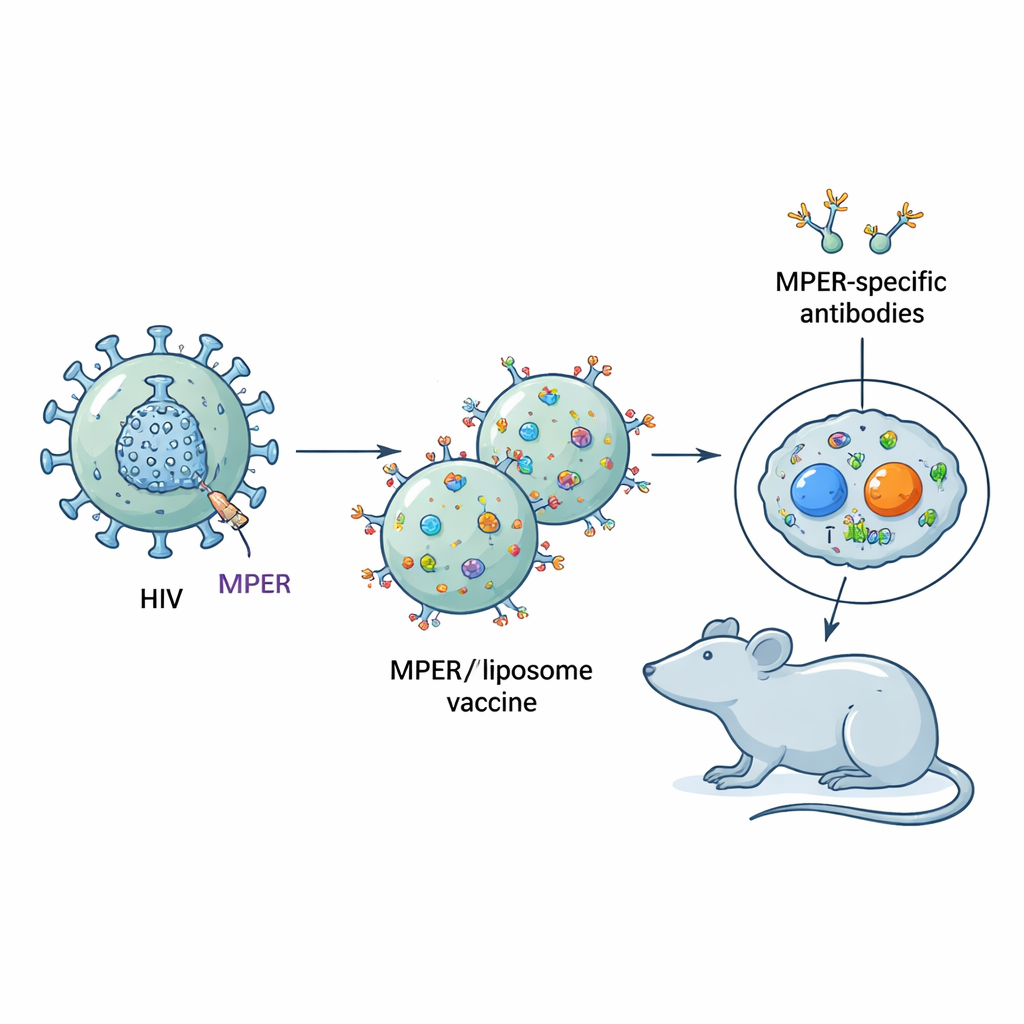
The virus’s hidden weak point
HIV is coated with a protein shell studded with spikes that the virus uses to enter cells. Many parts of these spikes mutate easily, helping HIV escape antibodies. In contrast, a short stretch near the base of one spike protein, called the membrane-proximal external region (MPER), is highly conserved across HIV strains and recognized by some of the most powerful neutralizing antibodies seen in a minority of people with long-term infection. Unfortunately, MPER sits right next to the viral membrane and is partly buried, so B cells—the cells that make antibodies—have trouble seeing it during infection or after standard vaccination.
Mimicking MPER on tiny vaccine particles
The researchers built vaccines in which synthetic MPER fragments were anchored densely on the surface of liposomes, creating a membrane-like environment that better resembles how MPER appears on the virus. Inside or attached to these same liposomes, they also included short “helper” peptides, which are snippets of protein recognized by CD4 T cells. These helper T cells provide essential support to B cells in germinal centers, the immune system’s training grounds where antibody-producing cells compete, mutate, and improve their binding. A key twist is that some helper peptides, such as one called LACK, are themselves strong B-cell targets—meaning antibodies readily form against them and can steal attention away from MPER.
Competition between helpful and distracting targets
When mice received an initial shot containing MPER on liposomes together with the LACK helper peptide, their immune systems mounted a strong response against LACK but only a weaker, low-affinity response against MPER. Measurements of germinal center B cells and antibody binding showed that LACK-specific B cells had an intrinsic affinity advantage; they outcompeted MPER-specific B cells for T cell help and dominated the antibody response. This suggested that even in a vaccine meant to highlight MPER, off-target epitopes attached for technical reasons—like helper peptides—can become immunological “attention hogs” and undermine the desired response.
Using mix-and-match boosters to refocus the immune system
The team then tested “heterologous” prime–boost regimens, where later shots changed both the form of MPER and the identity of the helper peptide. After priming with an MPER/LACK formulation, some mouse groups received boosters that swapped in different MPER constructs and replaced LACK with alternative helper peptides (HIV30 and OVA) that were less dominant as B-cell targets. These mix-and-match boosters markedly expanded MPER-specific long-lived plasma cells in bone marrow and increased MPER antibody levels and binding strength, while reducing antibodies to LACK. Detailed mapping showed that these regimens shifted antibodies toward particular MPER residues and improved binding to a full-length HIV Env protein on cell surfaces—an important step toward recognizing the real virus, even though broad neutralization was not yet achieved.
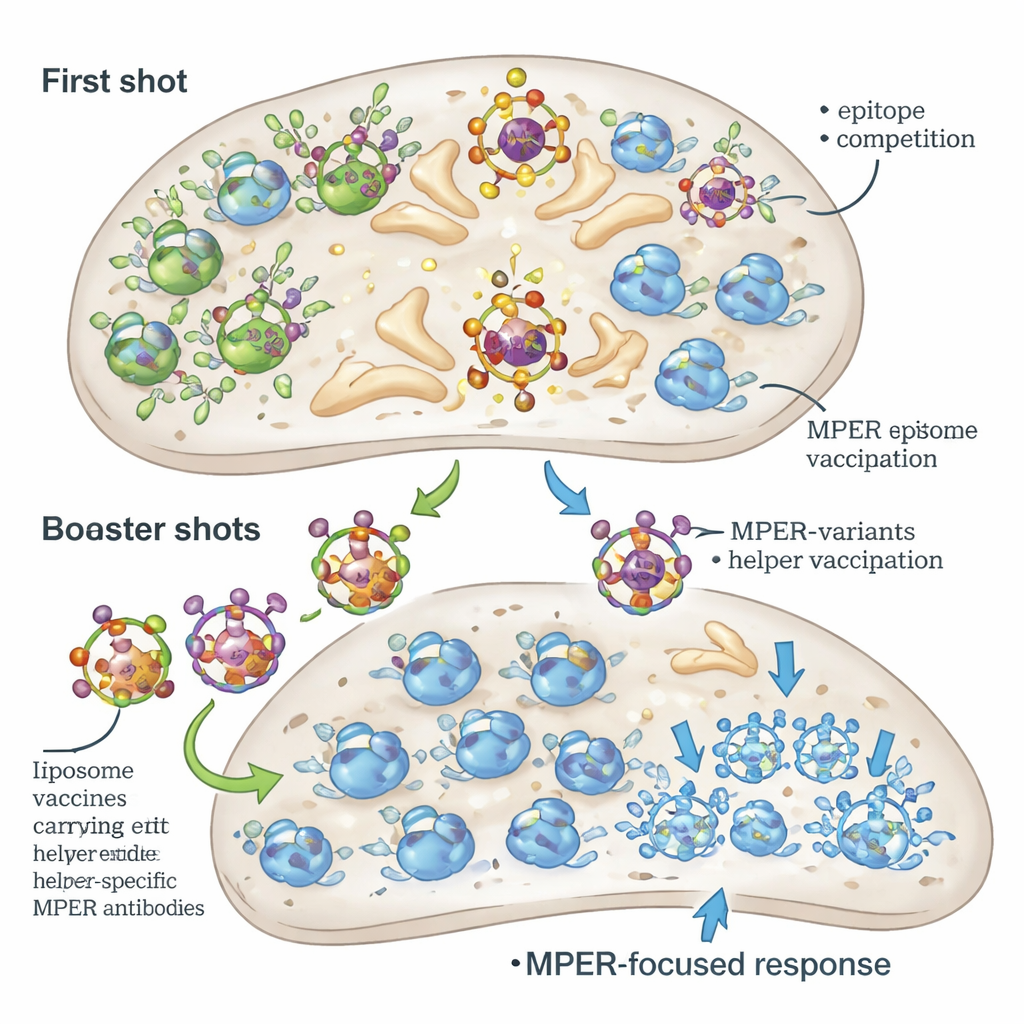
Balancing help and competition
Interestingly, the most helpful booster strategy was not simply the one that generated the largest overall T helper response. Boosts that continued to use LACK produced strong germinal center activity but still favored LACK-specific B cells over MPER. In contrast, boosters with alternative helper peptides provided somewhat weaker overall T cell help but narrowed the affinity gap between MPER-specific and helper-peptide–specific B cells, allowing MPER-targeted cells to compete more successfully. When helper peptides were anchored on the liposome surface rather than merely encapsulated, they provided robust T cell help without becoming overwhelmingly dominant B cell targets, further improving MPER antibody affinity.
What this means for future HIV vaccines
To a lay reader, the main message is that vaccines must train the immune system not just to recognize a viral weak spot, but to prioritize it over many other tempting targets. This study shows that by carefully choosing and swapping the helper components between shots—and by adjusting how antigens are displayed on nanoparticles—scientists can dial down distracting responses and amplify rare, desirable ones against the conserved MPER region. While the mouse antibodies produced here still do not neutralize HIV broadly, the work maps out concrete design principles for future HIV vaccines and other vaccines against fast-changing viruses: control epitope competition, vary boosters strategically, and pair smart immunogen design with equally smart immunization schedules.
Citation: Khan, R.A., Chen, J., Donius, L. et al. Heterologous immunization modulates B-cell epitope competition between helper peptides and the MPER segment in MPER/liposome vaccines. npj Vaccines 11, 51 (2026). https://doi.org/10.1038/s41541-026-01371-6
Keywords: HIV vaccine, MPER antibodies, liposome immunization, heterologous boosting, B cell epitope competition