Clear Sky Science · en
Overcoming vascular niche–mediated TKI resistance in acute myeloid leukemia through miR-126 inhibition
Why this matters for cancer patients
Acute myeloid leukemia (AML) is one of the most aggressive blood cancers, and even with modern targeted drugs many patients relapse. This study asks a deceptively simple question with big implications: is part of the problem not just the cancer cells themselves, but the "neighborhood" they live in inside the bone marrow? By building a detailed computer model grounded in lab data, the authors explore how tiny blood vessels can shield leukemia stem cells from drugs—and how blocking a single small RNA signal might dismantle that protection.
The hidden safe house in bone marrow
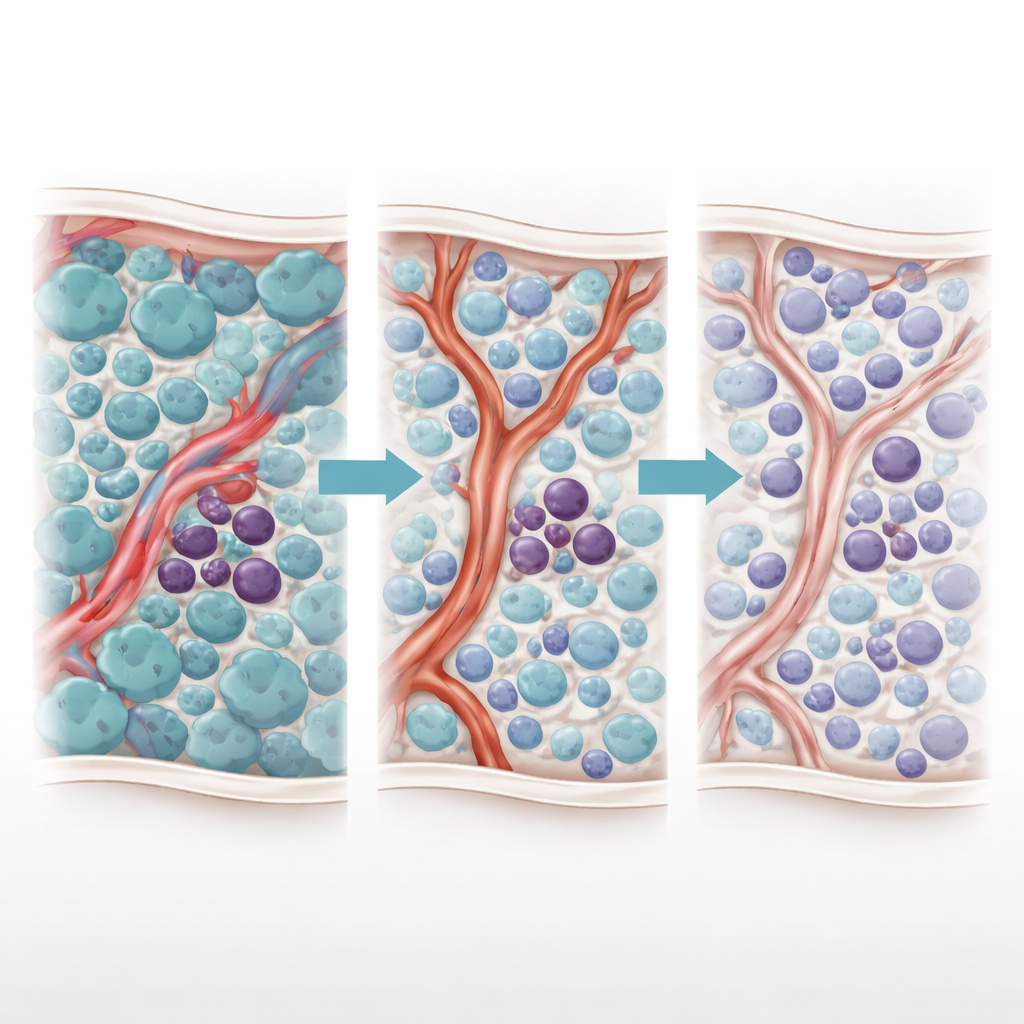
AML arises in the bone marrow, where malignant blast cells crowd out normal blood production. Within this swarm hides a smaller population of leukemia stem cells that can self-renew and survive treatment, seeding relapse. These stem cells do not float freely; they tuck into specialized pockets around small arteries and capillaries, known collectively as the vascular niche. The study focuses on patients whose leukemia cells carry FLT3-ITD mutations, which are treated with tyrosine kinase inhibitors (TKIs). Although these drugs can sharply shrink tumor burden, many patients still relapse, suggesting that some cells are being hidden or protected.
A two-faced treatment effect
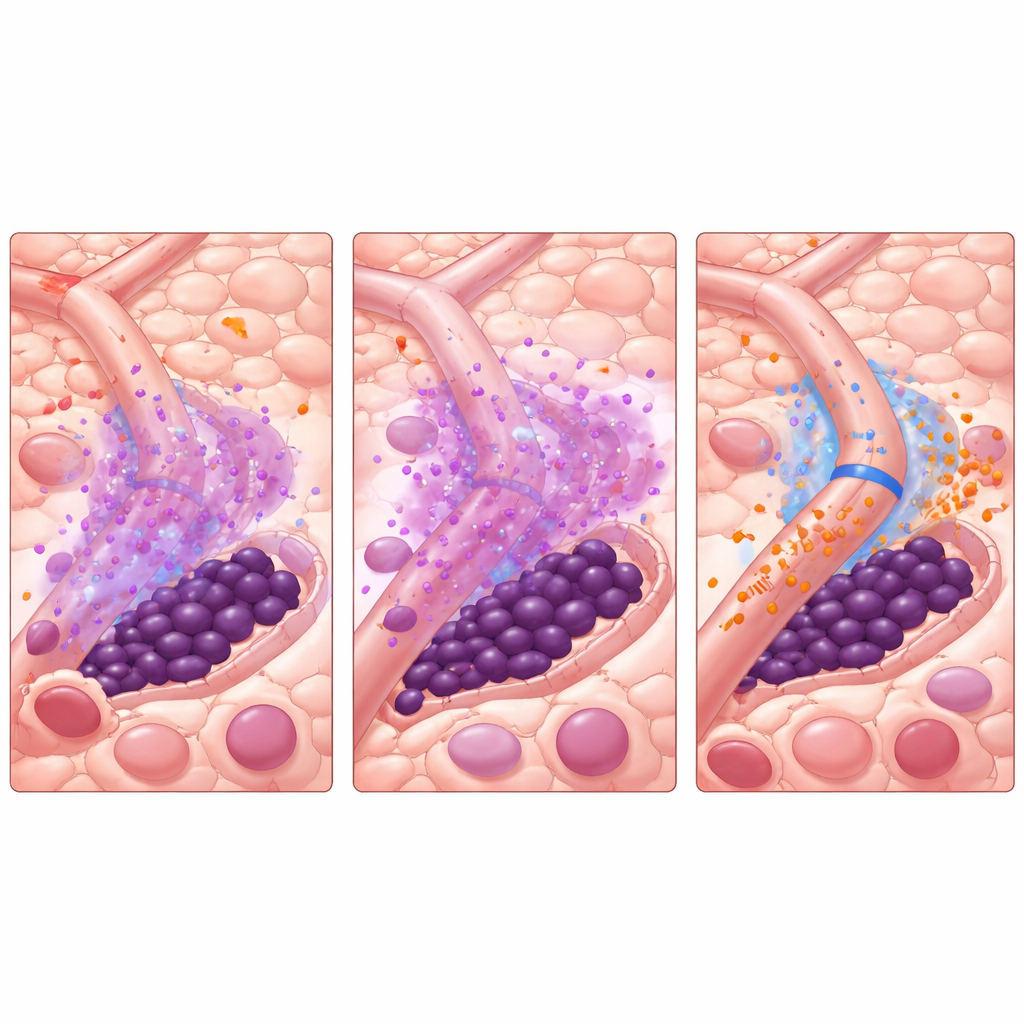
The authors previously uncovered a puzzling "Janus phenomenon"—a two-faced response to TKI therapy. As TKIs kill off rapidly dividing blast cells, levels of an inflammatory molecule, TNF-α, fall. This drop unexpectedly allows nearby blood vessel cells to ramp up production of a small regulatory RNA called miR-126. That miR-126 is passed from endothelial cells into leukemia stem cells, nudging them into a quiet, non-dividing state that makes them highly drug resistant. In other words, the very treatment that clears most cancer cells remodels the bone marrow in a way that shelters the most dangerous ones.
Building a virtual bone marrow
To dissect this dynamic, the team created an agent-based computational model of the AML bone marrow. Every key player—blast cells, leukemia stem cells, and endothelial cells lining blood vessels—is represented as an individual "agent" following biologically informed rules. Diffusing molecules such as TNF-α, miR-126, the TKI AC220, and a miR-126–blocking drug called miRisten spread through the simulated tissue. The model was calibrated with mouse imaging data that map real vascular patterns, including areas rich or poor in miR-126–producing vessels. In simulations, standard TKI treatment reliably reproduced the Janus effect: blasts declined, TNF-α dropped, miR-126 from vessels surged, stem cells retreated into protective niches, and disease later rebounded.
Turning off the shield with precise timing
Armed with this virtual system, the researchers tested ways to break the vicious cycle. Simply adding miRisten at the same time as the TKI helped but rarely cured the disease in the model. A more strategic approach was more powerful: short "pretreatment" with miRisten alone to dampen vascular miR-126, followed by either TKI alone or continued combination therapy. Across many simulated vascular layouts, even a few days of miRisten pretreatment before starting AC220 dramatically reduced or eliminated relapse while limiting total drug exposure. The model also revealed that how close leukemia stem cells sit to blood vessels strongly affects their fate—cells nestled right against vessels gain the most protection unless miR-126 is blocked.
What this could mean for future care
In plain terms, the study suggests that some leukemia stem cells survive not because TKIs are weak, but because the bone marrow’s small blood vessels temporarily switch into “protector” mode during treatment. By briefly turning off a single signal, miR-126, before giving the TKI, it may be possible to strip away this shield, force stem cells out of hiding, and make them killable by existing drugs. While this work is based on computer simulations anchored to lab data—not yet on clinical trials—it offers a concrete strategy: time a microenvironment-targeting drug just before and during standard therapy. If validated, this approach might not only improve outcomes for people with FLT3-mutated AML, but also inspire similar tactics in other blood and solid cancers where the tumor’s neighborhood helps it evade treatment.
Citation: Froid, M., Branciamore, S., Chen, Z. et al. Overcoming vascular niche–mediated TKI resistance in acute myeloid leukemia through miR-126 inhibition. npj Syst Biol Appl 12, 38 (2026). https://doi.org/10.1038/s41540-026-00675-6
Keywords: acute myeloid leukemia, leukemia stem cells, bone marrow microenvironment, tyrosine kinase inhibitor resistance, miR-126 inhibition