Clear Sky Science · en
Development of a severe rat refeeding syndrome model and mathematical modeling of the associated hypophosphatemia
Why Eating Again Can Be Dangerous
When people who have been severely undernourished finally receive food or intravenous nutrition, doctors must proceed carefully. A sudden rush of calories can trigger “refeeding syndrome,” a complication that disturbs the body’s salts and can harm the heart, muscles, and lungs. One key player is phosphorus, a mineral needed for energy in every cell. This study used rats and computer modeling to uncover why phosphorus in the blood can crash so dramatically during refeeding, and how we might better prevent this dangerous drop.
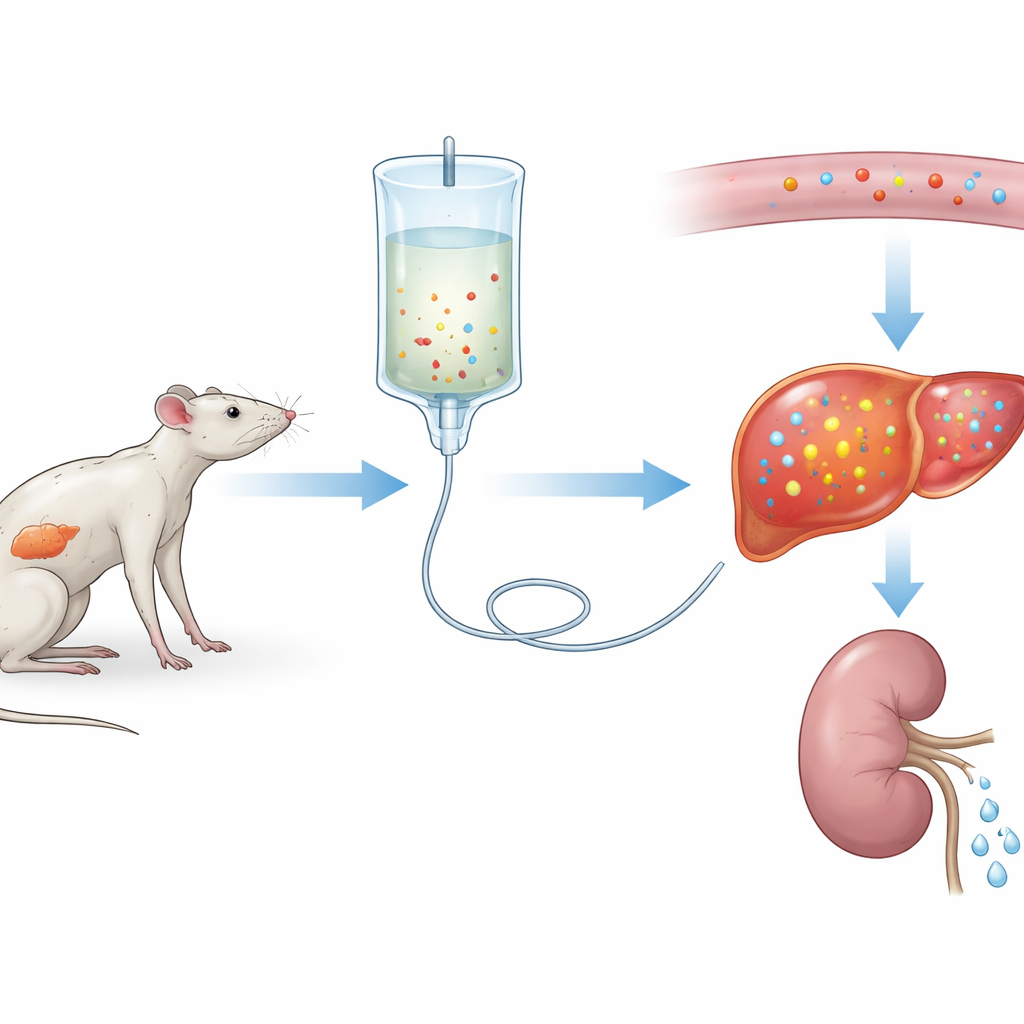
A Closer Look at Refeeding Trouble
The researchers first created a severe version of refeeding syndrome in rats. For three weeks, one group ate a normal diet while another received very little protein, mimicking long-term undernutrition. Both groups were then given the same intravenous nutrition for three days. Rats on the low-protein diet showed a steep fall in blood phosphorus—about a 75% drop—along with shifts in other salts and signs of organ injury, especially in the liver and muscles. These changes resembled serious refeeding syndrome in patients, suggesting the team had built a faithful experimental model.
What Happens to Phosphorus During Refeeding
To understand how phosphorus moves in the body over time, the scientists repeatedly sampled blood and urine from the same rats. They found that blood phosphorus levels fell in both normal and protein-starved animals after feeding began, but the drop was much deeper and longer-lasting in the undernourished group. Surprisingly, the kidneys actually reduced phosphorus loss in the urine right after refeeding, so the mineral was not being thrown away. Instead, phosphorus was being pulled from the bloodstream into tissues, particularly the liver, where its stores had been depleted by weeks of poor diet. Computer calculations confirmed that the underfed rats started with much lower phosphorus inside their cells and then soaked it up aggressively once nutrients arrived.
Insulin Is Not the Whole Story
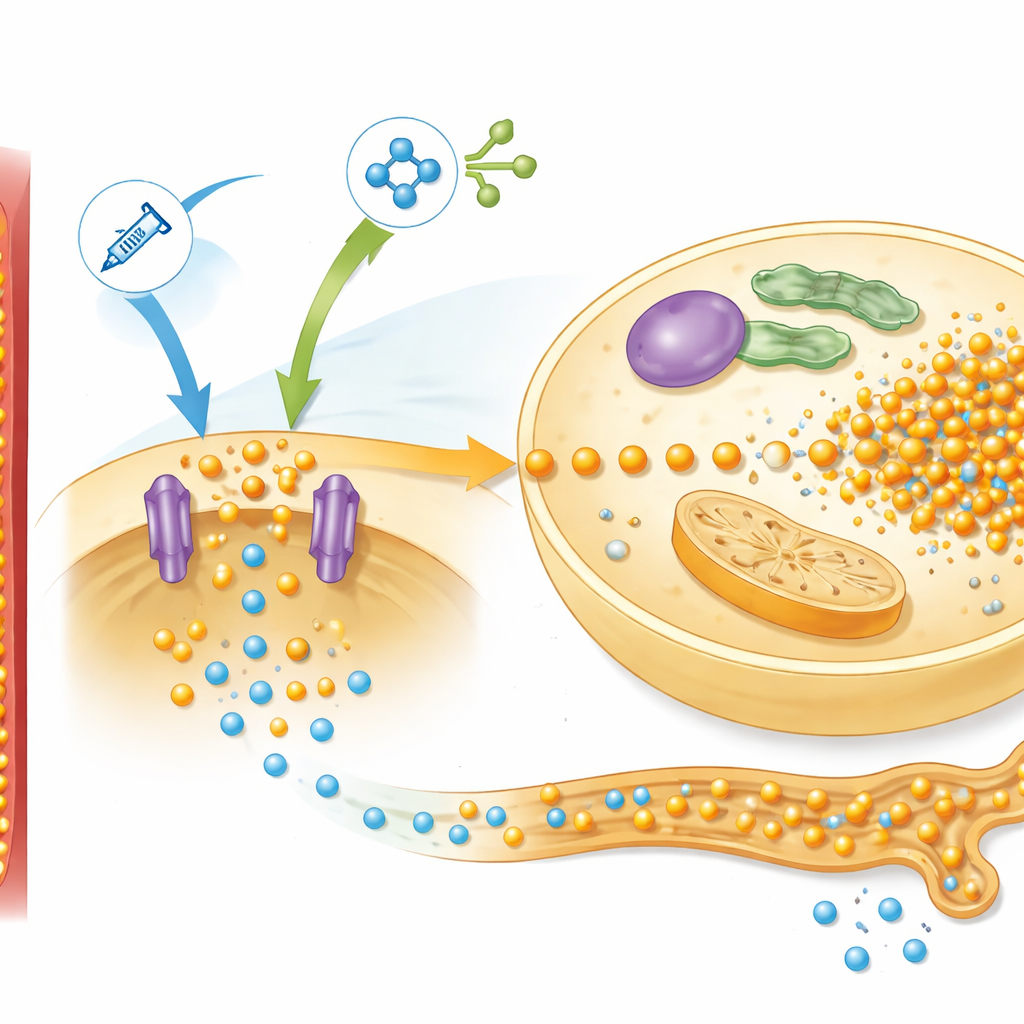
Because refeeding raises blood sugar and the hormone insulin, doctors have long blamed insulin for driving phosphorus into cells. The team tested this idea by dampening insulin release with the hormone somatostatin. As expected, blood sugar rose, but the plunge in blood phosphorus barely improved. At the same time, levels of free amino acids—the building blocks of proteins—went up. In a separate experiment, the scientists removed amino acids from the infusion. Now, phosphorus levels stayed much more stable, and the severe crash was prevented, even though insulin levels were similar. These results point to a combined effect: both insulin and amino acids, not insulin alone, spur cells to grab extra phosphorus during refeeding.
A Hidden Control Network in Liver and Kidney
Digging deeper, the researchers examined liver proteins involved in nutrient sensing, focusing on a pathway known as mTOR, which responds to insulin and amino acids. In the low-protein rats, refeeding strongly activated this pathway and increased levels of a phosphate transporter called Pit2, which helps liver cells pull in phosphorus. The pattern matched the behavior predicted by their mathematical model, which bundled this network into a single control signal they called “simTOR.” The model also required a separate feedback factor to explain how the kidneys adjust phosphorus loss in urine. Measurements of a bone-derived hormone, FGF-23, fit this role well: its levels plunged when feeding began, limiting phosphorus excretion, then diverged between normal and undernourished rats in a way that mirrored the simulated feedback signal.
Using Math to Test Prevention Strategies
By tuning different knobs in their computer model, the authors could ask “what if” questions that would be difficult or risky to test directly in patients. The analysis highlighted three major influences on the depth and recovery of the phosphorus dip: the initial phosphorus inside cells, the strength of the mTOR-driven uptake system, and the rate of phosphorus intake from nutrition. Simulations suggested that simply adding a large amount of phosphate at the time of refeeding would be hard to balance safely. However, giving extra phosphorus before refeeding—allowing organs to quietly rebuild their internal stores—could blunt the later crash in blood levels. Reducing the amino acid load, especially those that strongly activate mTOR and insulin, also appeared protective in the animal experiments.
What This Means for Patients
For people at risk of refeeding syndrome—such as those with long-standing eating disorders or severe frailty—this work suggests that the danger comes from more than just a spike in blood sugar. Starved organs enter refeeding with empty phosphorus tanks, then, under the combined push of insulin and amino acids, rapidly absorb the mineral from the blood while the kidneys briefly hold on to it. The result is a sharp, temporary shortage in circulation that can damage vital tissues. The rat model and mathematical framework developed here point toward more targeted prevention: cautious early use of phosphorus supplements, careful control of amino acid–rich nutrition, and attention to hormones that govern kidney excretion. Together, these approaches may help clinicians feed vulnerable patients more safely.
Citation: Kato, H., Yamaoka, I. & Kubota, H. Development of a severe rat refeeding syndrome model and mathematical modeling of the associated hypophosphatemia. npj Syst Biol Appl 12, 34 (2026). https://doi.org/10.1038/s41540-026-00658-7
Keywords: refeeding syndrome, hypophosphatemia, phosphorus metabolism, mTOR signaling, parenteral nutrition