Clear Sky Science · en
Asthma-mediated control of optic glioma growth via T cell-microglia interactions: A mathematical model
When Breathing Troubles Connect to Brain Tumors
Asthma and brain tumors seem worlds apart: one affects how we breathe, the other how we see, think, and move. Yet doctors have noticed a puzzling pattern—children with asthma appear less likely to develop certain brain tumors of the optic nerve. This article explores how a chronic lung condition might unexpectedly defend the brain, using a mathematical model to trace the hidden chemical conversations between immune cells and tumor cells.
A Slow-Growing Tumor with High Stakes
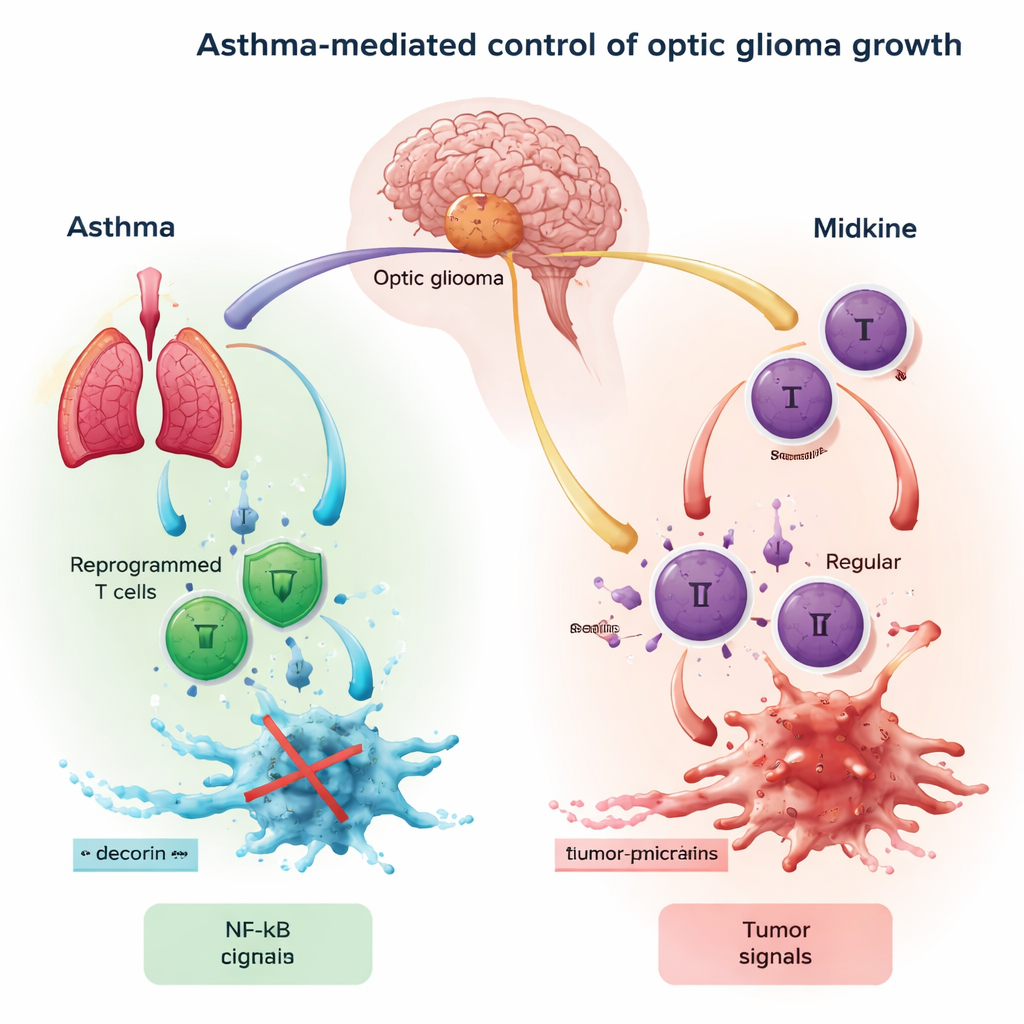
Optic gliomas are generally slow-growing tumors that form along the optic nerve, most often in children with a genetic condition called neurofibromatosis type 1 (NF1). Even though these tumors are considered “low grade,” they can still cause vision loss and hormonal problems. In NF1, a broken gene causes a signaling protein called RAS to become overactive. That overactivity turns on production of another molecule, midkine, which acts like a loudspeaker in the optic nerve, calling in immune cells and reshaping the local environment in ways that usually help the tumor rather than fight it.
A Tumor-Feeding Conversation Between Nerve and Immune Cells
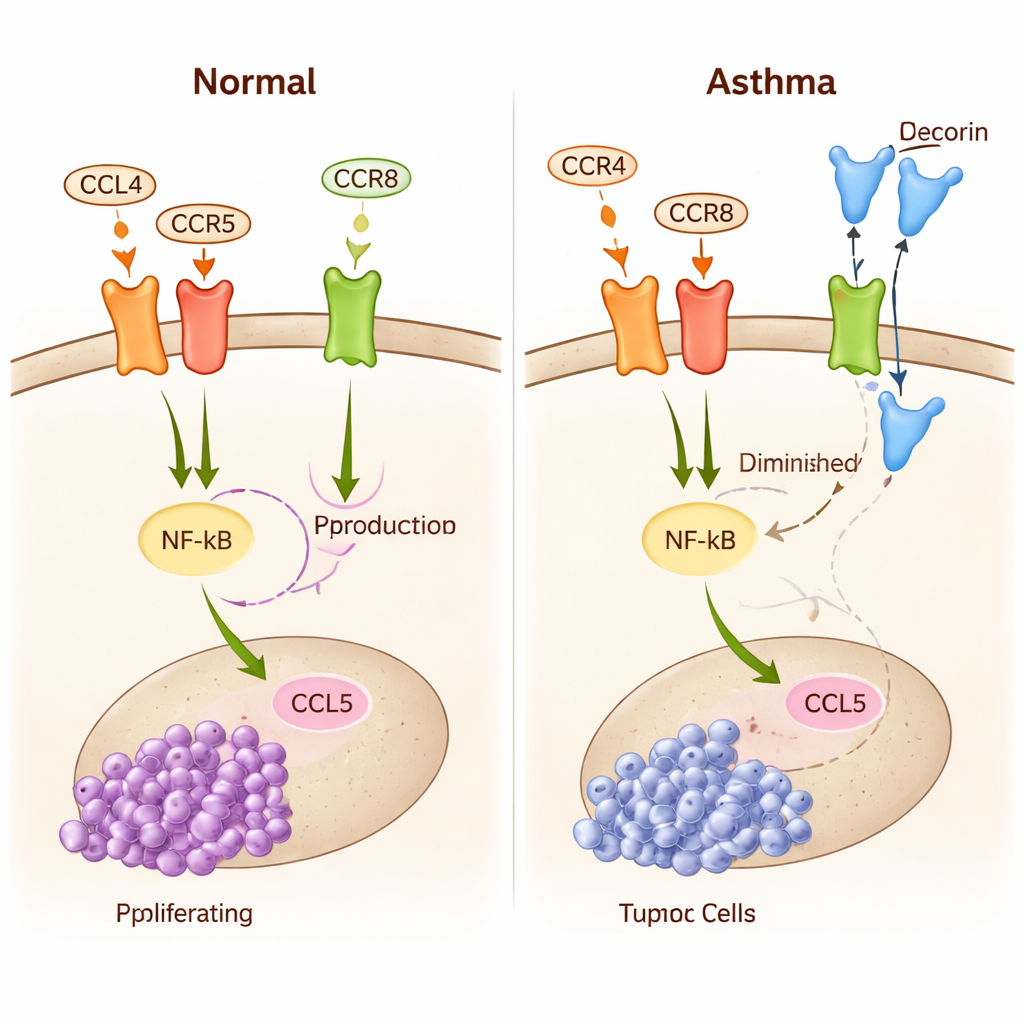
The authors focus on a chain of events that links the optic nerve, immune cells, and tumor growth. Midkine from the optic nerve first activates T cells, a type of white blood cell, prompting them to release a signal called CCL4. That signal then binds to receptors (CCR5 and CCR8) on microglia, the brain’s resident immune cells. When enough CCL4 latches onto these receptors, it flips on a master switch inside microglia known as NF-κB. Once activated, NF-κB drives production of another signal, CCL5, which encourages optic glioma cells to grow, migrate, and evade immune attack. In effect, the midkine–CCL4–NF-κB–CCL5 axis forms a circular “go” signal for tumor expansion.
How Asthma Reprograms T Cells into Tumor Restraints
Asthma is best known as a disease of chronic airway inflammation. But in people with asthma, some T cells become “reprogrammed” into a different state: they lose much of their direct cell-killing power and instead secrete molecules that reshape their surroundings. One of these molecules is decorin, a small protein with recognized anti-tumor properties. The key idea in this work is that asthma-primed T cells can travel from the lung to the brain and release decorin in the optic glioma’s neighborhood. Decorin competes with CCL4 for the CCR8 receptor on microglia, effectively blocking part of the tumor-feeding conversation and dialing down NF-κB and CCL5 inside the brain’s immune cells.
Using Mathematics to Follow an Invisible Network
Because this signaling web is too complex to grasp by intuition alone, the researchers built a detailed mathematical model using differential equations. The model tracks the levels of key molecules (midkine, CCL4, decorin, NF-κB, CCL5), the activity of receptors on microglia, and the growth of tumor cells over time. They used experimental data to calibrate how strongly each interaction occurs and then simulated many scenarios. The model shows how small changes in binding strength—how easily CCL4 or decorin attach to their receptors—can flip the microglia between two modes: a tumor-promoting state with high NF-κB and CCL5, and a tumor-suppressing state with strong decorin binding and weak CCL4 signals. A simple index based on the ratio of decorin-bound to CCL4-bound receptors accurately predicts whether the tumor tends to grow or stall.
Designing Smarter Immune-Based Treatments
Beyond explaining why asthma might protect against optic glioma, the model is used to explore treatment strategies. It suggests that therapies which boost decorin levels or weaken CCL4’s grip on its receptors could slow tumor growth. The authors digitally test approaches such as repeatedly infusing decorin-producing T cells or using the immune messenger IL-2 to expand these beneficial cells inside the body. Interestingly, the simulations reveal that not only the total dose but also the timing of such treatments matters: moderate, well-spaced dosing can keep microglia in the tumor-suppressing state as effectively as larger, less frequent doses, with potentially fewer side effects.
What This Means for Patients and Families
For non-specialists, the central message is that asthma’s chronic inflammation, usually seen as harmful, can sometimes reshape the immune system in ways that hinder certain brain tumors. By turning T cells into factories for decorin, asthma appears to tilt the balance in the optic nerve from a growth-friendly environment toward a more hostile one for cancer. While no one is suggesting asthma as a therapy, the mechanisms uncovered here could inspire new treatments that copy its protective aspects—using tailored T-cell therapies or drugs that mimic decorin’s blocking action—to safeguard children at risk for optic glioma.
Citation: Lee, D., Lawler, S. & Kim, Y. Asthma-mediated control of optic glioma growth via T cell-microglia interactions: A mathematical model. npj Syst Biol Appl 12, 26 (2026). https://doi.org/10.1038/s41540-026-00647-w
Keywords: optic glioma, asthma, decorin, T cells, mathematical modeling