Clear Sky Science · en
Optimal control theory as a method for designing multidrug adaptive therapy regimens
Why taming cancer, not wiping it out, can work better
Cancer treatment usually aims to kill as many tumor cells as possible, fast. Yet this aggressive approach can backfire: it often wipes out drug‑sensitive cells and unintentionally clears the way for drug‑resistant cells to take over. This article explores a counter‑intuitive idea—using mathematics and two cancer drugs together to keep sensitive and resistant cells competing with each other, so the tumor stays controllable for much longer.
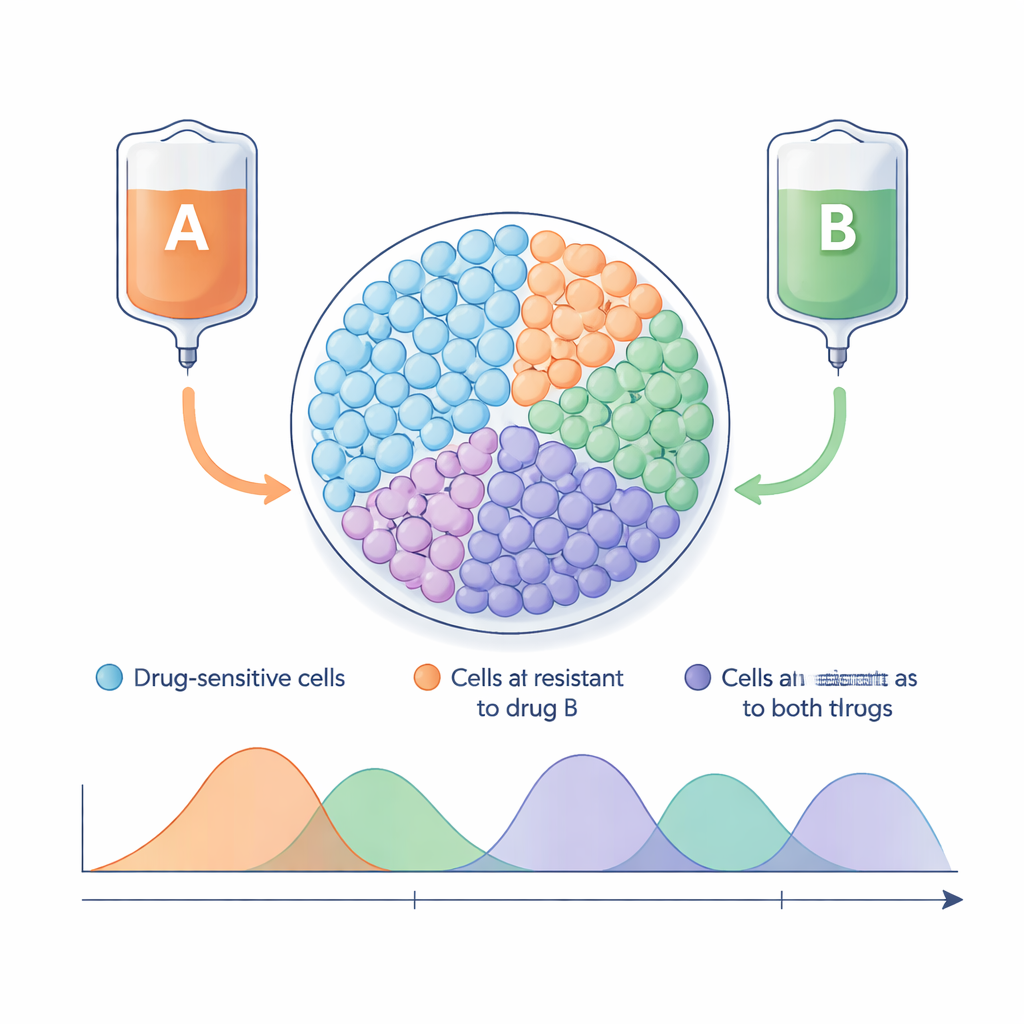
Rethinking how tumors evolve under treatment
Tumors are not uniform masses of identical cells. Instead, they are mixed communities that include cells easily killed by treatment and others that already resist one or more drugs. Standard chemotherapy regimens focus on maximum cell killing and rarely consider how this diversity lets the tumor evolve under drug pressure. When a powerful treatment removes sensitive cells, resistant ones can grow almost unhindered, leading to relapse. Adaptive therapy flips this logic: instead of aiming for complete eradication, it aims to keep a stable population of sensitive cells alive so they can crowd out resistant competitors, holding the overall tumor at a size the patient can tolerate.
Using math to design smarter multidrug schedules
The researchers built a mathematical model of a tumor made of four cell types: cells sensitive to both drugs, cells resistant only to drug A, cells resistant only to drug B, and cells resistant to both. The model assumes that these cells share limited space and resources, so they grow more slowly as the total tumor approaches a maximum size. On top of this, two drugs can be turned up or down over time. The team then applied optimal control theory, a branch of mathematics used to find the best way to steer dynamical systems, to answer a clinical-style question: how should we vary the doses of the two drugs over time to keep the tumor below a chosen size threshold for as long as possible?
Letting cells compete beats hitting with maximum doses
The analysis revealed broad rules for how the drugs should be used. When the tumor is kept close to the allowed size limit, sensitive and partly resistant cells strongly compete with the fully resistant ones, slowing their expansion. The most successful regimens, called competition maintenance (CM) schedules, deliberately modulated doses so that total tumor size hovered near this threshold. Sometimes both drugs were used together at a fixed ratio and gradually increased; other times one drug was used adaptively first and the second added later. In contrast, regimens that relied heavily on keeping one or both drugs at their maximum allowed doses tended to perform worse, because they eliminated too much competition and allowed fully resistant cells to eventually dominate.
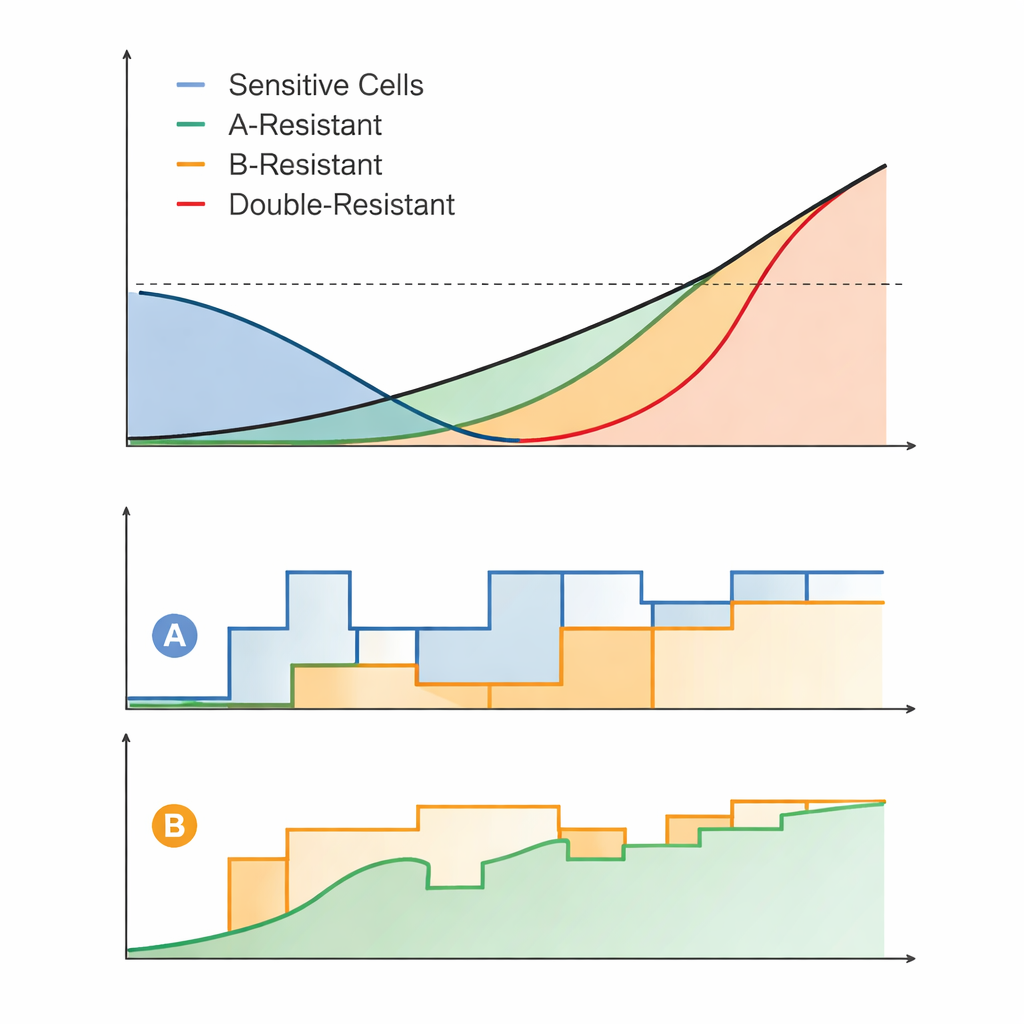
From ideal math to practical treatment plans
Perfectly adjusting doses continuously is unrealistic in the clinic or even in lab experiments, so the authors created "practical" versions of their CM regimens. In these simplified schedules, drug doses were only adjusted once per day and in coarse steps. Even with these constraints, the practical adaptive regimens usually controlled the tumor far longer than standard-of-care–like strategies, such as giving both drugs at a constant high dose or switching from one full‑dose drug to the other only after the tumor regrew. The advantage of competition‑based strategies was especially clear when the allowed tumor size threshold and maximum drug doses were not extremely low, conditions under which it is possible to sustain strong competition.
What this means for future cancer care
Overall, the study shows that, in a setting where drug resistance already exists, the key to prolonging control is not how cleverly we kill tumor cells, but how well we preserve competition between sensitive and resistant cells at a manageable tumor size. Optimal control theory provided a systematic way to narrow down and compare many possible two‑drug regimens, highlighting robust patterns rather than one fragile "perfect" schedule. While the work is based on a simplified lab-style model and will need experimental testing, it supports a growing view: for some advanced cancers, the best strategy may be to manage the disease as a chronic, evolving ecosystem rather than trying to annihilate it at all costs.
Citation: Widdershins, A., Hansen, E., Read, A. et al. Optimal control theory as a method for designing multidrug adaptive therapy regimens. npj Syst Biol Appl 12, 27 (2026). https://doi.org/10.1038/s41540-025-00613-y
Keywords: adaptive therapy, drug resistance, optimal control, cancer evolution, multidrug regimens