Clear Sky Science · en
Polysaccharides and the colon mucus barrier: a review of biophysical interactions and functional impacts
Why the gut’s slippery lining matters
Deep inside your large intestine lies a slimy, invisible shield: a mucus layer that keeps trillions of microbes at arm’s length from your own cells. This review article explores how complex sugars called polysaccharides—from foods, seaweeds, fungi, and medical products—interact with that mucus barrier. Understanding this dialogue between diet and mucus helps explain why certain fibers are good for gut health, how some treatments can protect against colitis and other bowel diseases, and how smarter drug-delivery systems might one day slip medicines precisely where they are needed most.
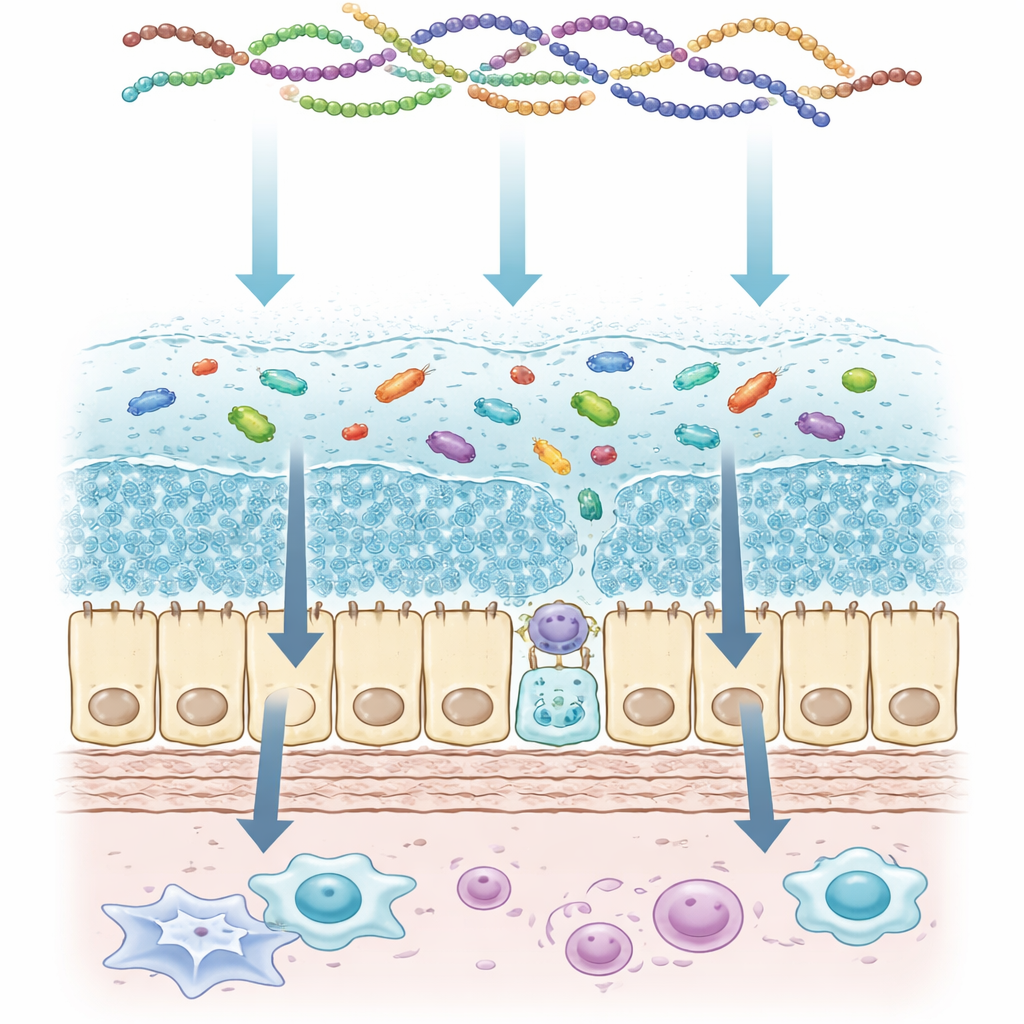
The colon’s two-layer safety blanket
The authors begin by describing the architecture of colon mucus as a two-story structure. Closest to the intestinal wall is a dense inner layer that is almost free of bacteria and acts like a tight shield. On top sits a looser outer layer that serves as a controlled habitat for friendly microbes. Specialized cells called goblet cells continuously produce and release large mucus molecules, which rapidly unfold and gel when they reach the intestinal surface. These gel-forming molecules are heavily decorated with sugar branches, which give mucus its thickness, water-holding ability, and negative charge. When this structure breaks down—through inflammation, infection, or genetic defects—bacteria press closer to the wall, immune alarms fire, and chronic gut disorders can emerge.
Different sugars, different ways of sticking
Polysaccharides themselves are long chains of simple sugars, but their behavior in mucus depends on details like size, branching, and electrical charge. Neutral chains such as resistant starches and inulin mainly weave into the mucus mesh through hydrogen bonds and physical entanglement. Negatively charged chains from pectins, alginates, and seaweed gums can weakly associate or repel the already negative mucus, depending on local ions and the fine pattern of charges on both partners. Positively charged chains, especially chitosan from shellfish and fungi, are strongly attracted to mucus and can form tight complexes with it. The review explains that these interactions are not governed by a single force but by a mix of electrostatic attraction, hydrogen bonding, hydrophobic contacts, and subtle van der Waals forces that together determine how deeply a polysaccharide penetrates the gel and how firmly it adheres.
Model gut walls on chips, dishes, and slices
To untangle these complex interactions, researchers rely on a toolbox of experimental models. Flat cell cultures can be enriched with mucus-secreting cells to examine how specific polysaccharides change mucus production, thickness, or leakiness. Three-dimensional organoids grown from stem cells build miniature intestinal tubes that secrete their own mucus, while microfluidic “gut-on-a-chip” platforms add realistic flow, stretching, and live bacteria. Ex vivo setups keep freshly isolated intestinal tissues alive for short periods, preserving natural mucus layers and immune cells. Each model has trade-offs: simple dishes are easy but artificial, chips and organoids are more realistic but technically demanding. By comparing results across these systems, scientists can distinguish direct physical effects of polysaccharides on mucus from indirect effects driven by microbes and the immune system.
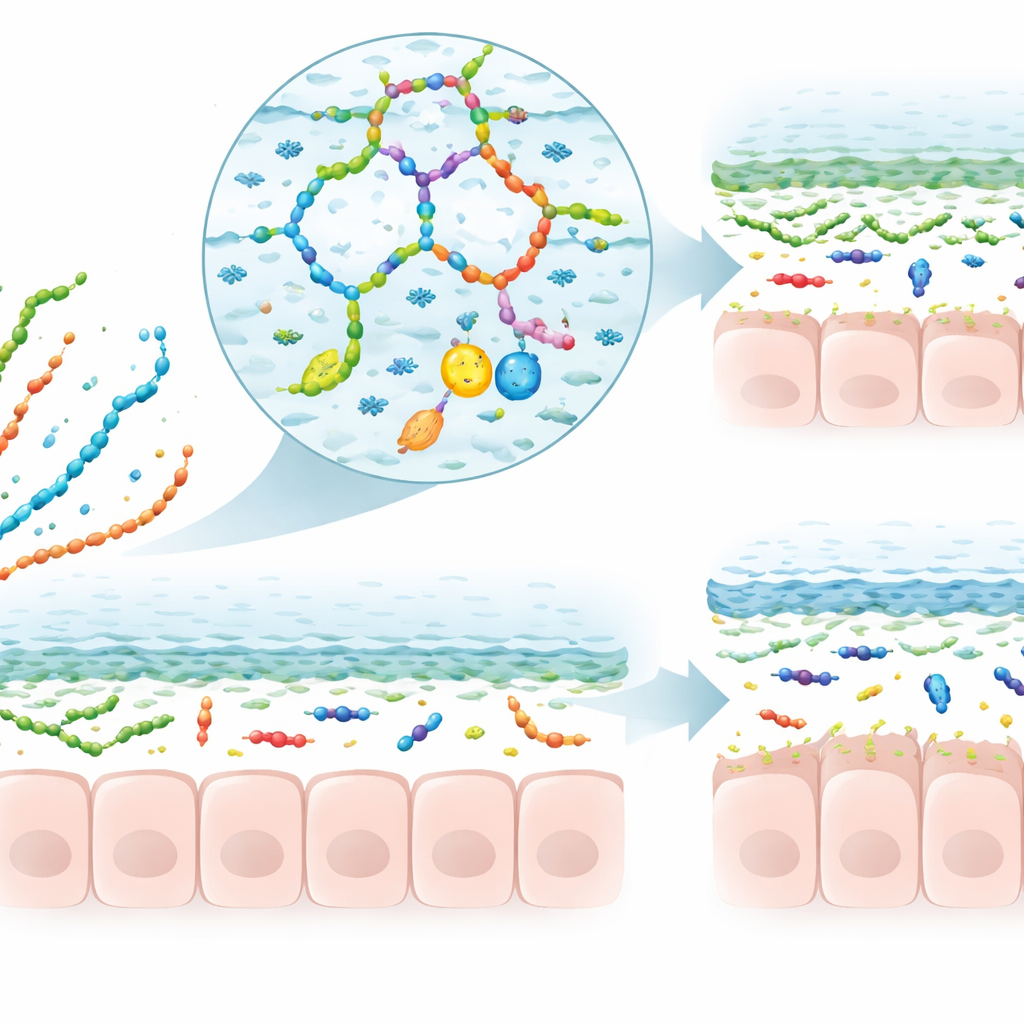
Shaping mucus, microbes, and the immune response
The review then examines what happens functionally when polysaccharides meet mucus. Some positively charged chains can temporarily loosen or reorganize the gel, affecting how easily particles and drugs move through it. Others form mixed networks with mucus that thicken or stabilize the layer. Many dietary polysaccharides are not digested by us but are instead fermented by gut bacteria, which turn them into short-chain fatty acids and other small molecules. These fermentation products signal to goblet cells to make more mucus, tune the sugar decorations on mucus, and support a community of microbes that tends to sit safely in the outer layer. In animal models of colitis, certain plant and mushroom polysaccharides increase goblet cell numbers, restore mucus thickness, and keep bacteria farther from the intestinal wall, often alongside lower inflammation and oxidative stress.
Designing foods and therapies that work with mucus
In closing, the authors argue that polysaccharides can be deliberately designed or selected to support the mucus barrier rather than accidentally damaging it. By linking specific structural traits—such as charge and molecular weight—to measurable outcomes like mucus thickness, viscosity, and penetrability, they propose guidelines for choosing polysaccharides that reinforce the barrier, carry drugs more effectively to the colon, or gently steer the microbiota towards health-promoting activities. For non-specialists, the central message is that the slimy film lining the colon is not just passive goo; it is an active interface where the chemistry of our diet, our resident microbes, and our immune defenses meet. Thoughtfully engineered polysaccharides could become key tools for keeping that interface intact and our guts in balance.
Citation: Cheong, KL., Biney, E., Wang, M. et al. Polysaccharides and the colon mucus barrier: a review of biophysical interactions and functional impacts. npj Sci Food 10, 98 (2026). https://doi.org/10.1038/s41538-026-00750-6
Keywords: colon mucus barrier, dietary polysaccharides, gut microbiota, mucoadhesive drug delivery, inflammatory bowel disease