Clear Sky Science · en
Point-of-use colorimetric detection of Escherichia coli in food matrices with DNAzyme crosslinked hydrogels
Why Safer Food Starts with Simple Tests
Most of us assume the food we buy is safe, yet invisible germs like harmful strains of E. coli can slip into milk, salads, meat, and ready‑to‑eat meals and cause serious illness. Today’s lab tests are accurate but slow, expensive, and rarely available where food is grown, processed, or sold. This paper describes a small, low‑cost sensor that changes color when it encounters E. coli, offering a way for farmers, factories, and even retailers to check for contamination on the spot using only their eyes.
A Hidden Threat in Everyday Groceries
Foodborne disease affects hundreds of millions of people each year worldwide, and dangerous E. coli strains are a major culprit. They have been found in ground beef, unpasteurized or poorly pasteurized milk, fresh vegetables, and especially leafy greens such as lettuce and spinach. Because recalls often happen only after people get sick, regular checks along the food chain—from farm to supermarket shelf—are essential. However, many existing detection methods, like bacterial culturing or PCR, require trained staff, specialized machines, and laboratory conditions, making them poorly suited for routine, on‑site use.
Why Food Is Hard to Test
Detecting bacteria in food is much trickier than in clean water. Real foods contain fats, proteins, sugars, and particles that can clog sensors, blur optical signals, or stick to the very molecules meant to recognize the germs. Enzymes found naturally in foods can even break down DNA‑based tests, and harmless background bacteria can confuse sensors that lack good specificity. To cope with these issues, many current methods add extra preparation steps to separate bacteria from the rest of the sample, which adds cost, time, and complexity—precisely what is undesirable for quick point‑of‑use checks.
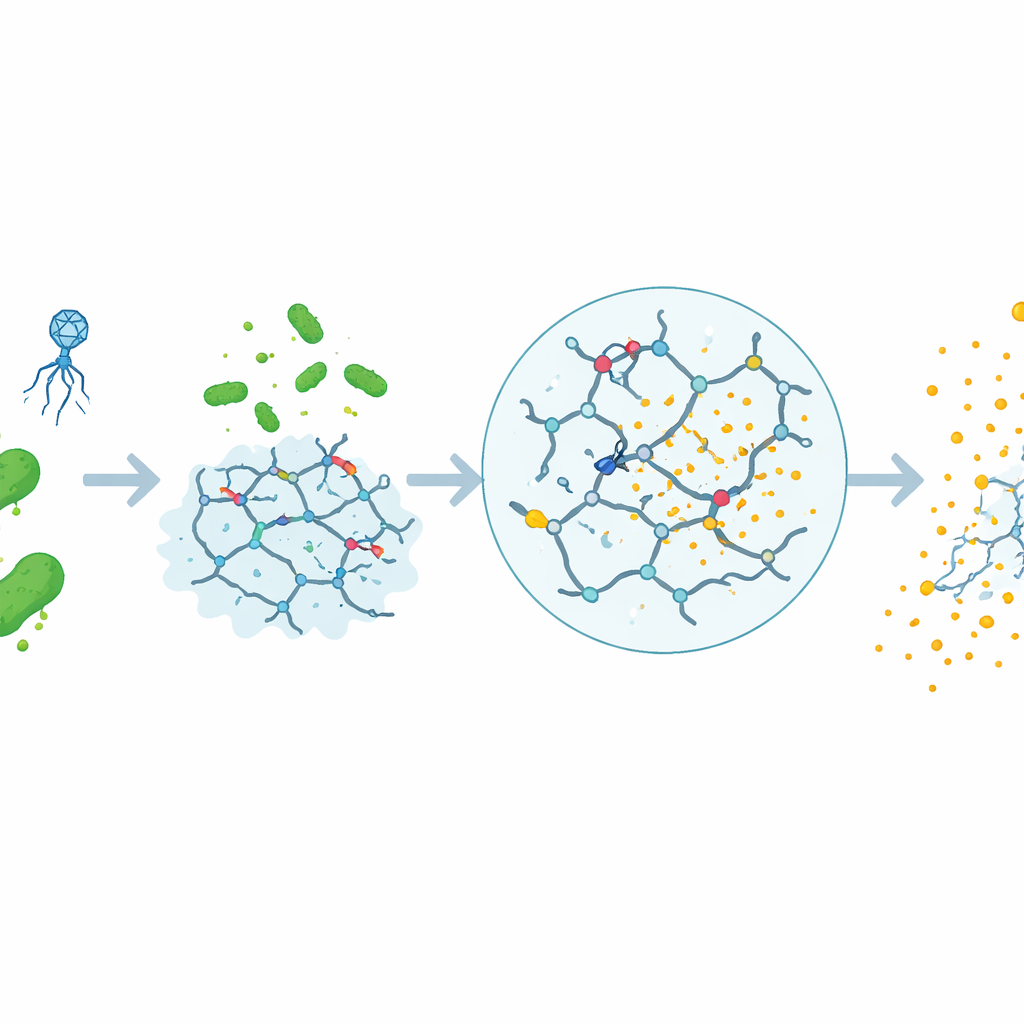
A Smart Gel That Lets Gold Particles Go
The authors previously created a tiny hydrogel—a soft, water‑rich solid—linked together by special DNA strands called DNAzymes. These DNAzymes are designed to recognize a protein released by many E. coli strains. Within the gel, red‑colored gold nanoparticles are trapped, giving it a vivid hue. To run a test, the gel is mixed with a food sample, simple nutrients, and a virus (a bacteriophage) that infects E. coli. If E. coli is present, the phage causes the bacteria to burst, releasing more of the target protein. This protein activates the DNAzymes, which cut the DNA links holding the gel together. As the gel falls apart, the gold nanoparticles are freed and spread through the liquid, changing how the sample looks to the naked eye. If no E. coli is present, the gel remains intact and the color stays concentrated.
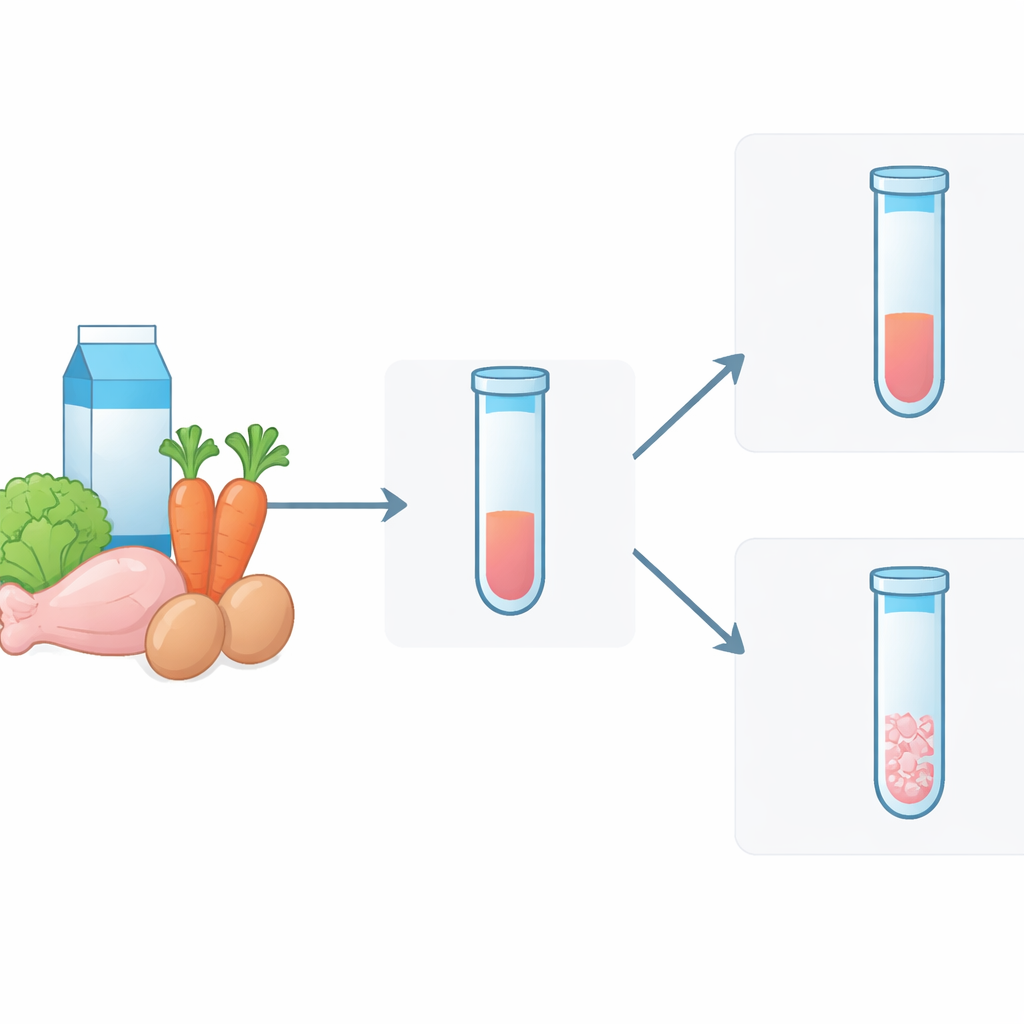
From Milk to Salad: Putting the Sensor to Work
In this study, the team tested whether their color‑changing gel could handle real foods rather than just clean laboratory samples. They contaminated skim milk, liquid from grocery‑store rotisserie chicken, brine from packaged hard‑boiled eggs, and juice from baby‑cut carrots with known amounts of harmless model E. coli. Because some samples were too thick, they diluted them slightly, then incubated everything with the gel and phage at body temperature. After about 18 hours, the difference was visible by eye: contaminated samples showed a clearly degraded gel and dispersed color, while clean controls stayed intact. The sensor could detect contamination around 104–105 bacterial cells per milliliter in milk, similar to or better than many simple field tests.
Tackling the Leafy Greens Problem
Leafy greens are frequent sources of large E. coli outbreaks, so the authors explored several realistic ways to test them. They first collected water droplets from misted iceberg lettuce and then the rinse water from a mixed salad washed in a spinner, spiking both with bacteria. In each case, the gel correctly signaled when E. coli was present, even at relatively low levels. To mimic how public health labs might process real contaminated produce, the researchers next directly spiked salad leaves, physically mashed them with water in a “stomaching” step, and measured how many bacteria were recovered. After diluting these particle‑rich liquids, the sensor still produced a clear visual response at both moderate and high contamination levels, while ignoring other naturally occurring bacteria that were present.
What This Could Mean for Your Dinner Plate
By showing that a DNAzyme‑crosslinked hydrogel can reliably spot E. coli in many common foods, this work points to a future where simple, inexpensive, and equipment‑free tests could be built directly into food packages or used throughout the supply chain. Although the current sensor takes about 18 hours to show a result and would need further refinements to improve speed and sensitivity, it already matches traditional culture‑based testing times while avoiding complex lab tools. With additional development and adaptation to other bacteria, similar gels could help catch contamination earlier and more often, reducing the risk that dangerous microbes ever reach your plate.
Citation: Mann, H., Prasad, A., Uthayasekaram, R. et al. Point-of-use colorimetric detection of Escherichia coli in food matrices with DNAzyme crosslinked hydrogels. npj Sci Food 10, 92 (2026). https://doi.org/10.1038/s41538-026-00745-3
Keywords: food safety, E. coli detection, colorimetric sensor, DNAzyme hydrogel, point-of-use testing