Clear Sky Science · en
Food-grade Lacticaseibacillus paracasei postbiotics suppress oral Streptococcus mutans biofilm formation and cariogenicity
Fighting Cavities with Food-Based Helpers
Tooth decay is one of the most common health problems in the world, driven largely by sugar-loving bacteria that drill away at our enamel. This study explores an emerging idea: instead of relying only on harsh mouthwashes or live “good bacteria” (probiotics), we might protect our teeth using safe, stable substances made by friendly food microbes. The researchers show that postbiotics from a food-grade bacterium, Lacticaseibacillus paracasei, can weaken a major cavity-causing germ and its sticky dental plaque, pointing toward new kinds of oral-care foods and rinses.
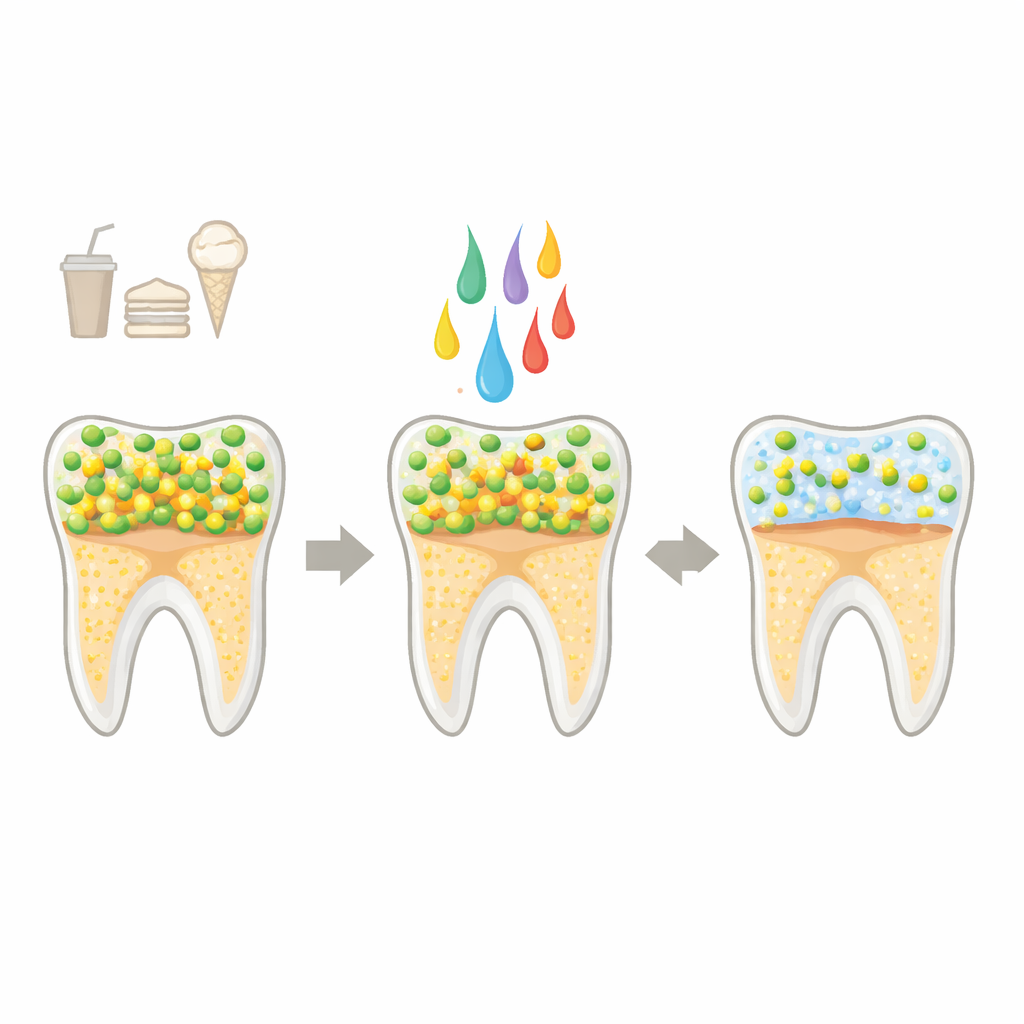
The Cavity Culprit and a New Kind of Ally
Tooth decay begins when our normal mouth community falls out of balance. Frequent sugar intake feeds acid-producing bacteria such as Streptococcus mutans, which build thick, glue-like layers (biofilms) on teeth. These biofilms trap acids against the enamel, dissolving minerals and eventually causing cavities. Brushing, flossing, and chemical rinses help, but they can be hard to use perfectly every day and may cause side effects. Probiotic products with live bacteria are promising but raise questions about stability, storage, and safety. Postbiotics—non-living preparations made from beneficial bacteria or their secreted components—offer a middle road: they can be food-grade, long-lasting, and easier to handle, while still nudging harmful microbes in a healthier direction.
How Postbiotics Weaken Harmful Mouth Bacteria
The team focused on the cell-free supernatant (CFS) of L. paracasei, essentially the broth left after removing the bacteria. They tested how this liquid affected S. mutans floating freely and growing in biofilms. In lab cultures, the CFS almost completely stopped S. mutans from multiplying. Under the electron microscope, treated cells looked crumpled and damaged, with rough surfaces and debris. Flow-based cell measurements confirmed that their membranes became leaky, their internal electrical balance collapsed, and far fewer cells remained metabolically active. The CFS also made the bacterial surface less water-repelling, which is important because stickier, more hydrophobic cells cling more easily to teeth and to each other when building plaque.
Breaking Down Sticky Plaque and Protecting Enamel
When S. mutans formed biofilms, the postbiotics still had a strong impact. The CFS reduced the overall mass of the biofilm, and a concentrated form cut it even more. The bacteria inside these films grew poorly, formed shorter chains, and produced less of the sugary glue (exopolysaccharides) that gives plaque its structure. Three-dimensional imaging showed thinner, patchier biofilms with fewer live, intact cells and a weakened matrix. To mimic real teeth, the researchers grew S. mutans on synthetic enamel disks coated with human saliva and then cycled them through “feeding” and “rinsing” with either CFS, a standard mouthwash (chlorhexidine), or a control solution. The CFS-treated biofilms were smoother and lighter, and—most importantly—released less free calcium from the enamel, a direct sign of reduced tooth demineralization, even though the surrounding liquid remained quite acidic.
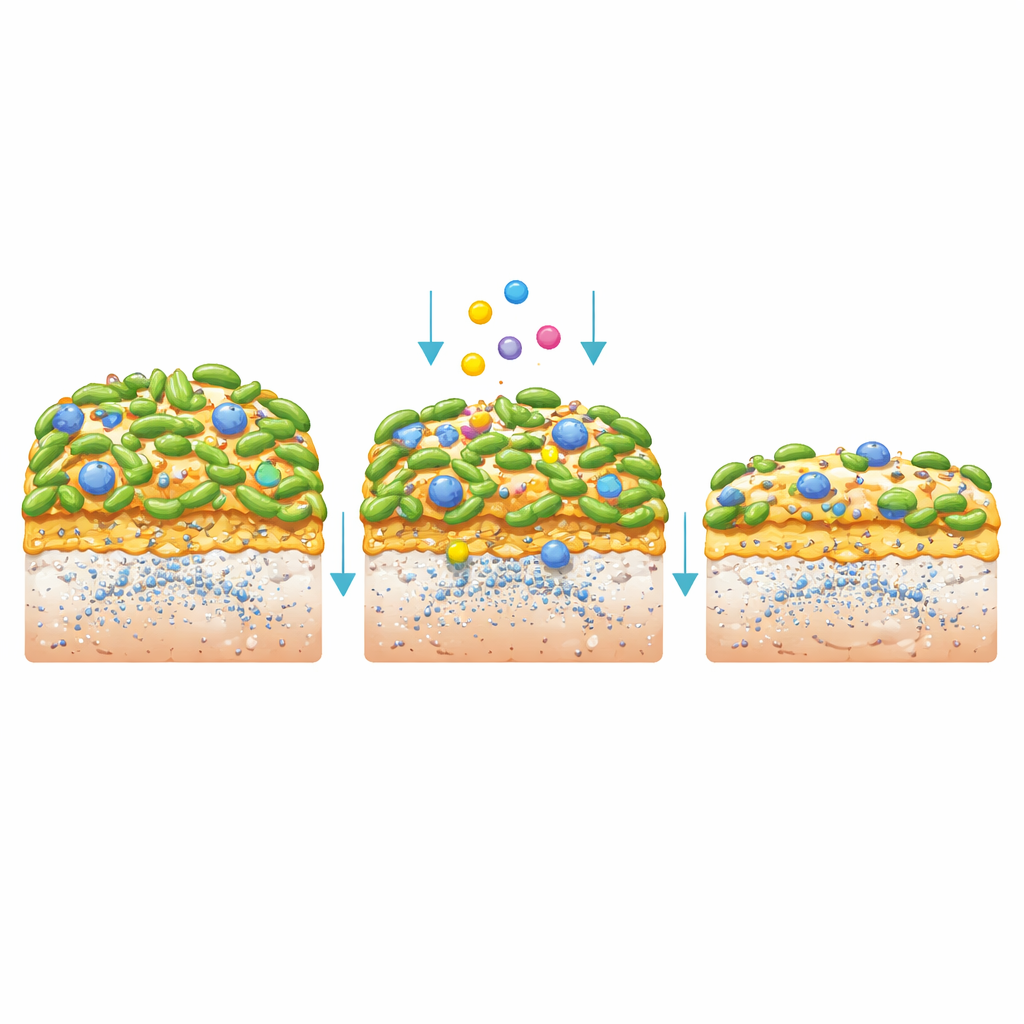
Digging into the Molecular Playbook
To find out what in the CFS was doing the work, the scientists selectively disabled candidate components. Breaking down proteins, removing hydrogen peroxide, or adding extracted sugars from the broth hardly changed its antibacterial effect. But neutralizing its acidity sharply weakened its power, suggesting that organic acids were the key players. These acids were more than just simple low pH: similar acidity created with mineral acid alone did not suppress S. mutans nearly as well, pointing to specific organic acid mixtures acting together. The team then examined which bacterial genes and small molecules changed when S. mutans grew with CFS. They found reduced activity in genes tied to stickiness, glue production, stress resistance, and bacterial “conversation” systems (quorum sensing) that coordinate group behavior. Certain metabolites, such as creatine and phosphoenolpyruvate, shifted in ways that matched lower acid output and weaker virulence, helping explain why less enamel mineral was lost.
What This Could Mean for Everyday Oral Care
In simple terms, this work shows that safe, food-grade postbiotics from L. paracasei can punch holes in a key cavity-causing bacterium’s armor, thinning its plaque, weakening its defenses, and reducing the acid damage to tooth-like surfaces. Because these postbiotics are stable to heat, storage, and a range of conditions, they could be blended into lozenges, rinses, or functional foods as a gentle, long-lasting aid alongside brushing and flossing. While they do not replace good oral hygiene or regular dental visits, they point toward a future in which smart, food-derived ingredients quietly reshape the mouth’s microscopic community to keep teeth stronger for longer.
Citation: Luo, SC., Hu, PF., Wei, SM. et al. Food-grade Lacticaseibacillus paracasei postbiotics suppress oral Streptococcus mutans biofilm formation and cariogenicity. npj Sci Food 10, 89 (2026). https://doi.org/10.1038/s41538-026-00742-6
Keywords: dental caries, oral microbiome, postbiotics, Streptococcus mutans, functional foods