Clear Sky Science · en
Lycopene mitigates T-2 toxin-induced hepatic ferroptosis by targeting the Nrf2/mitophagy axis in mice
Why a Mold Toxin in Everyday Grains Matters
Many staple foods, including wheat and corn, can be contaminated by a mold toxin called T-2 toxin. This invisible contaminant survives cooking and processing, and long-term exposure can quietly damage vital organs, especially the liver. The study summarized here asks two urgent questions: how exactly does T-2 toxin harm the liver, and can a natural food compound—lycopene, the red pigment in tomatoes and other fruits—help protect against this damage?
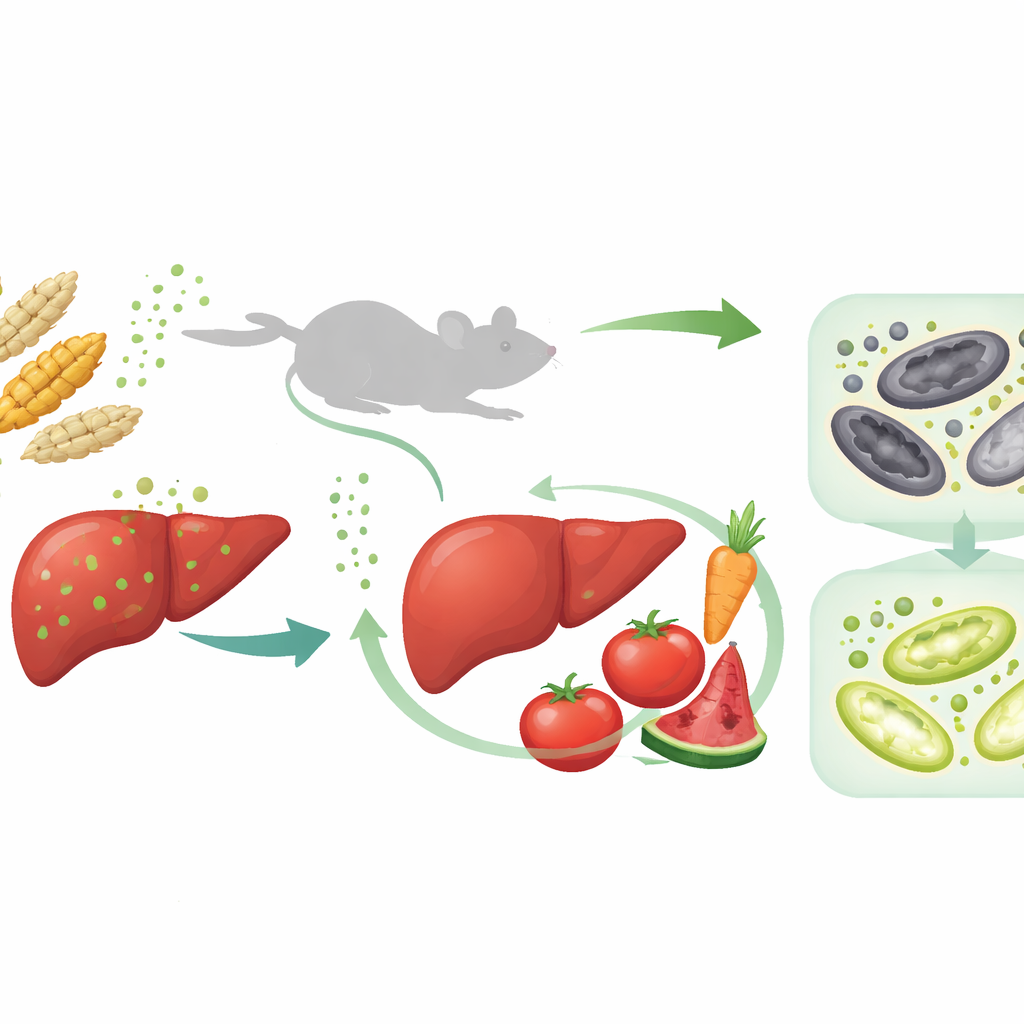
How the Toxin Harms the Liver
The researchers first fed mice graded doses of T-2 toxin for four weeks to mimic ongoing dietary exposure. As toxin levels rose, the animals gained less weight, their livers became enlarged and pale yellow, and blood tests showed rising levels of liver enzymes that signal injury. Under the microscope, liver tissue looked swollen and disorganized, with vacuoles inside cells and signs of cell death. A large-scale protein survey of liver samples revealed that many of the most altered pathways were tied to iron handling, fat metabolism, and a specific form of cell death called ferroptosis, which is driven by iron and runaway oxidation of fats.
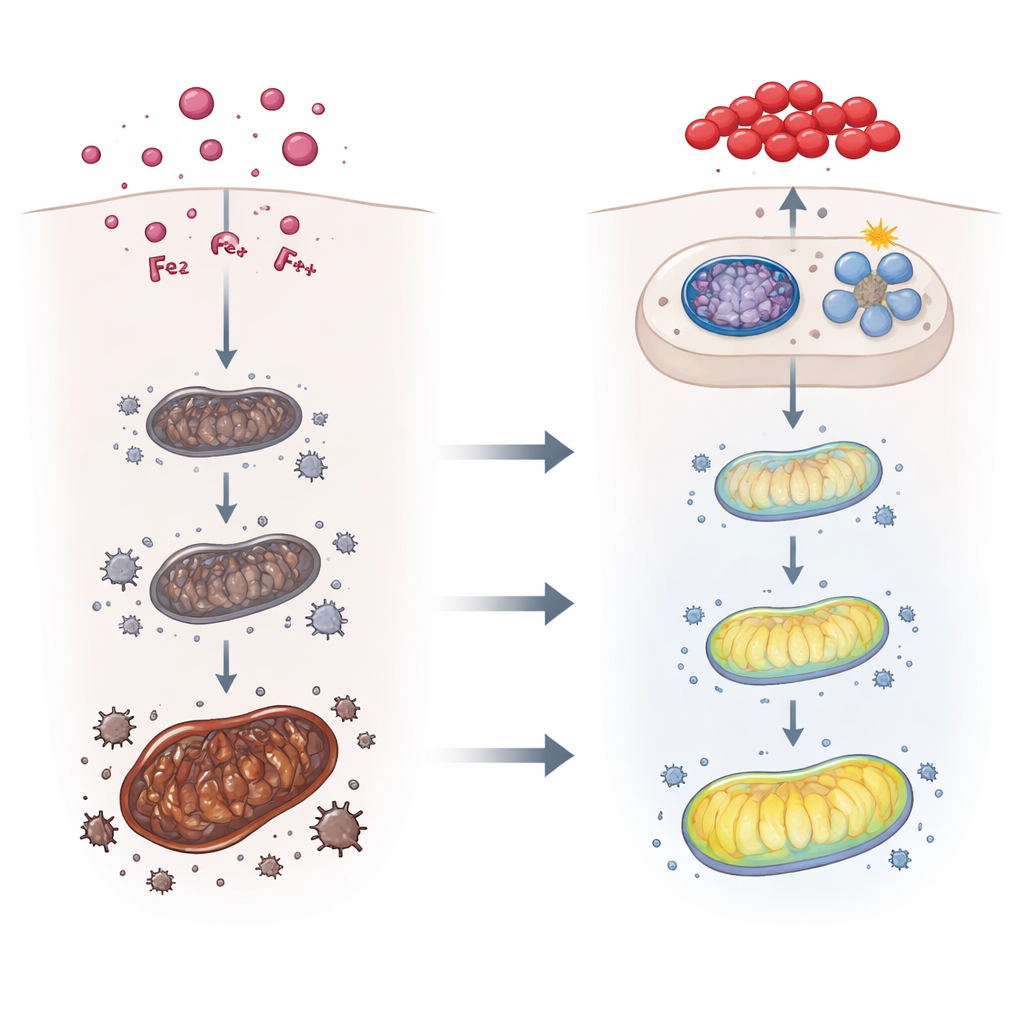
Iron Overload and a Rust-Like Cell Death
Diving deeper, the team measured classic markers of ferroptosis. In toxin-exposed livers, iron content climbed, while molecules that indicate fat damage increased and the activity of a key protective enzyme, GPx-4, fell. Proteins that normally store iron safely inside cells went up, but the main iron “exporter” on liver cells went down, suggesting that iron was getting trapped inside. When the scientists gave some mice a drug that specifically blocks ferroptosis, liver structure and function improved: swelling was reduced, injury scores fell, and chemical signs of oxidative damage eased. This firmly linked T-2 toxin’s liver toxicity to an iron-driven, rust-like form of cell death.
Mitochondria, Cell Cleanup, and a Defense Switch
The team then turned to mitochondria, the energy factories inside cells, which are both a source and a target of oxidative stress. In toxin-treated mice, mitochondrial energy output dropped while leakage of cytochrome c and overall reactive oxygen species rose, and electron microscopy showed shrunken, structurally damaged mitochondria. At the same time, the cell’s quality-control system—mitophagy, a selective “cleanup” process that removes defective mitochondria—was activated through a pathway involving proteins PINK1 and Parkin. When the researchers used mice lacking Parkin, and thus unable to mount normal mitophagy, T-2 toxin caused even worse iron buildup, stronger ferroptosis signals, and more severe liver damage. This showed that mitophagy acts as a protective brake, limiting toxin-induced injury.
A Built-In Shield Pathway and Lycopene’s Role
Because iron balance, oxidative stress, and mitophagy all seemed intertwined, the authors searched for an upstream controller and homed in on Nrf2, a master switch that turns on antioxidant and detox genes. T-2 toxin itself partially activated Nrf2, but when mice were given a known Nrf2-activating compound, liver iron export improved, ferroptosis markers declined, and mitophagy increased, all of which softened the toxin’s impact. The team then asked whether lycopene could serve as a natural Nrf2 booster. Computer modeling suggested that lycopene can physically bind both Nrf2 and its repressor protein Keap1 in ways that would favor Nrf2 activation. In live mice, lycopene treatment before and during toxin exposure improved body weight, restored liver appearance and blood tests, lowered iron accumulation and oxidative damage, and further activated Nrf2 and the PINK1–Parkin mitophagy pathway.
What This Means for Food Safety and Diet
In plain terms, the study shows that T-2 toxin injures the liver by trapping excess iron inside cells, wrecking mitochondria, and triggering ferroptosis. The body responds by switching on Nrf2 and mitophagy to clear damaged mitochondria and rebalance iron, but this defense can be overwhelmed. Lycopene appears to strengthen this internal shield, helping the liver export iron, clean up faulty mitochondria, and resist ferroptotic cell death. While more work is needed before translating these findings to humans, the research suggests that certain plant compounds, like lycopene, could one day become part of strategies to reduce the health impact of unavoidable foodborne toxins.
Citation: Yang, X., Song, W., Lu, Z. et al. Lycopene mitigates T-2 toxin-induced hepatic ferroptosis by targeting the Nrf2/mitophagy axis in mice. npj Sci Food 10, 94 (2026). https://doi.org/10.1038/s41538-026-00736-4
Keywords: T-2 toxin, lycopene, liver injury, ferroptosis, mitophagy