Clear Sky Science · en
Contrasting effects of phase-oriented antioxidant localization on oxidative resistance and physical stability of double emulsions
Why this study matters for everyday foods
Many of today’s sauces, dressings, and “functional” foods rely on clever mixtures of oil and water to carry flavors and fragile nutrients. These mixtures, called emulsions, can separate or turn rancid over time, ruining taste and quality. This paper explores how placing natural antioxidants in different parts of a complex emulsion can either protect it from going bad or, unexpectedly, make it more likely to separate. The findings help explain how to design longer-lasting, healthier food products without relying only on synthetic additives.
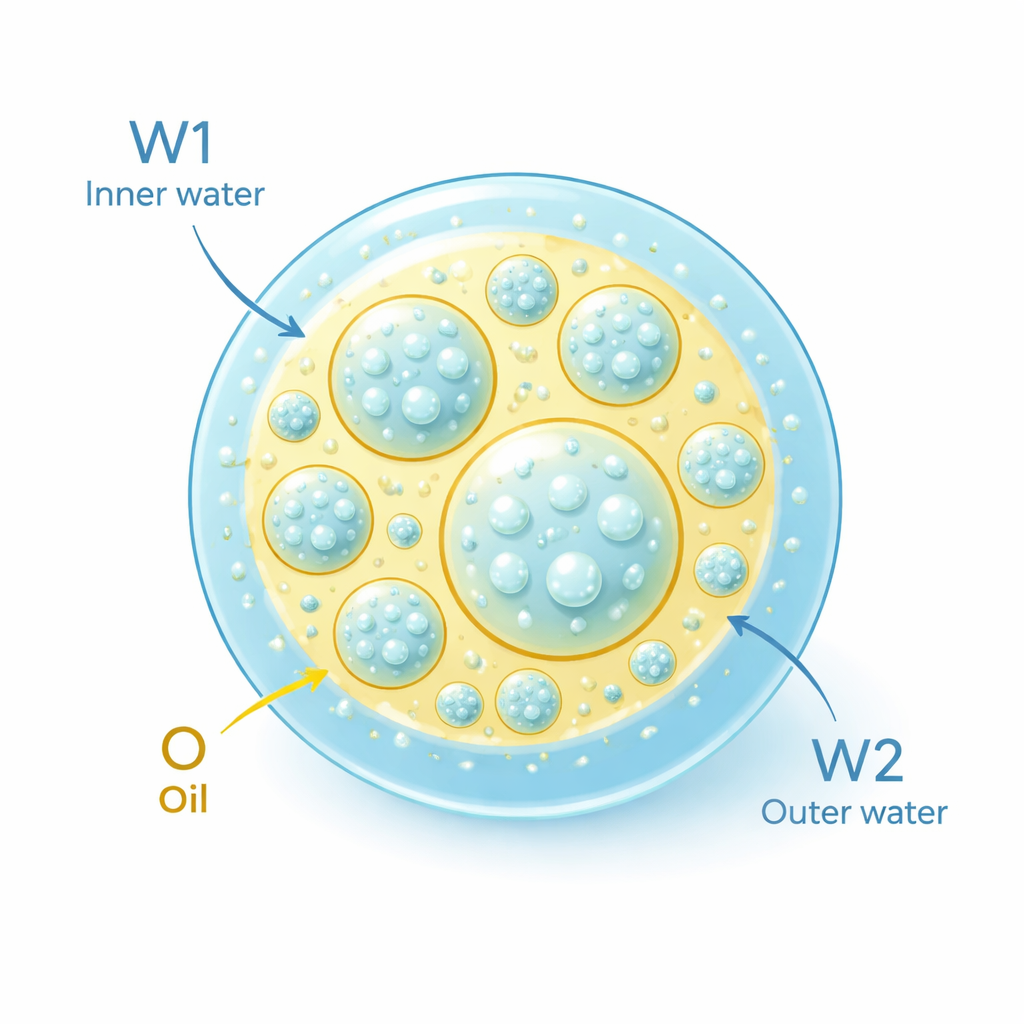
A droplet inside a droplet: building a double emulsion
The researchers worked with a special structure called a water-in-oil-in-water double emulsion. In simple terms, tiny droplets of water are trapped inside oil droplets, and those oil droplets are then suspended in another outer water layer. This design can hide bitter or unstable ingredients in the inner water pockets while still flowing like a thick sauce. However, such systems have an enormous contact area between oil and water, which makes the fats inside especially prone to oxidation (the same process that causes oils to go rancid) and to physical problems like creaming and layer separation.
Two natural defenders: one loves water, one loves oil
The team focused on two well-known natural antioxidants. Gallic acid is water-loving and is commonly found in many plant foods, while paprika oleoresin is oil-loving and rich in bright red and yellow carotenoids from peppers. Gallic acid was placed either in the inner water, the outer water, or both, while paprika oleoresin was mixed into the oil. The scientists then monitored how these choices affected droplet size, color, thickness (viscosity), electrical charge on the droplets, and signs of fat oxidation during four weeks of cold storage. They also grouped the samples with simple codes: NN (no gallic acid), GN (gallic acid only inside), NG (only outside), and GG (both inside and outside).
Smaller droplets, brighter colors—and more separation
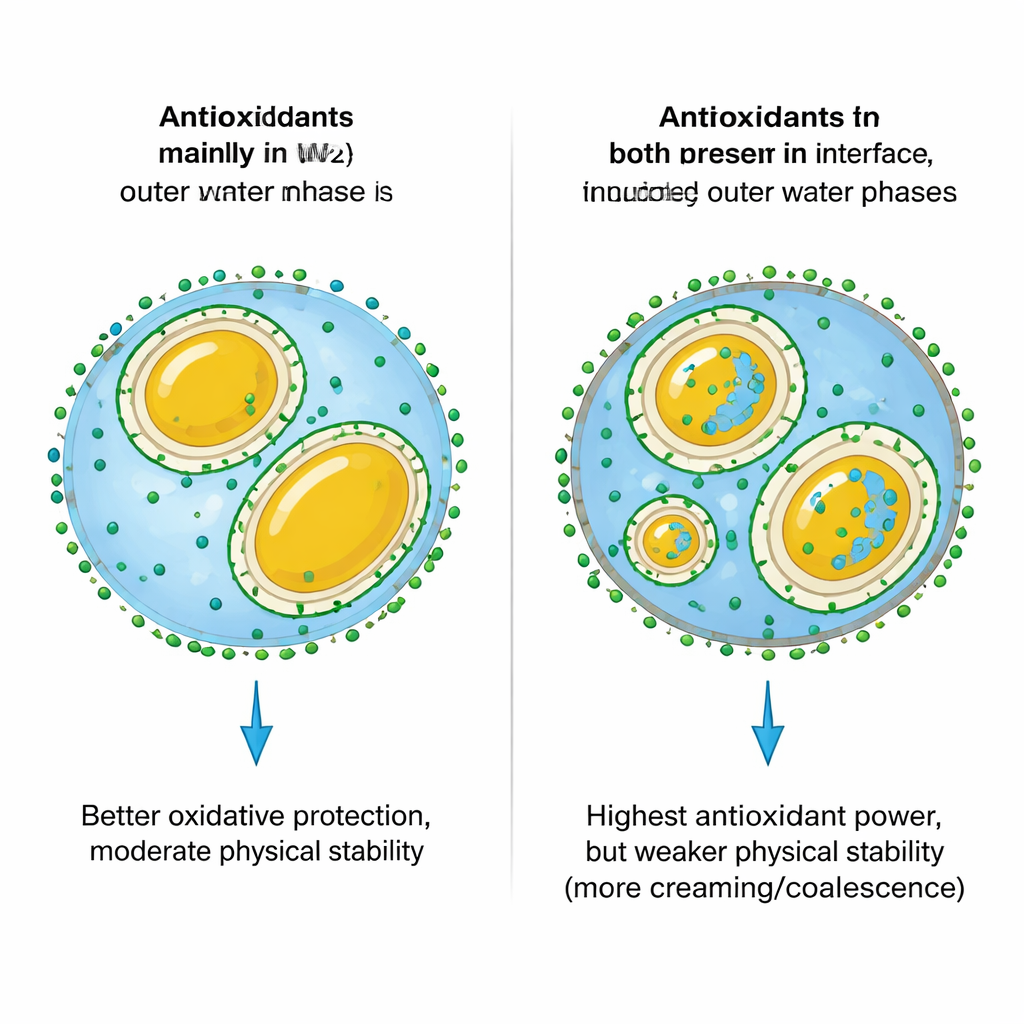
Adding paprika oleoresin and gallic acid changed how tightly the oil and water phases held together. Both ingredients lowered the “surface pull” at the oil–water boundary, allowing the mixer to break the oil into smaller droplets. This initially produced creamier-looking, whiter emulsions. But there was a trade-off: smaller droplets and lower viscosity also made it easier for droplets to move and reorganize. Over several weeks, samples with antioxidants, especially those containing paprika oleoresin and gallic acid in the outer water, showed more creaming (a visible top layer) and some oil rising to the surface. Measurements of droplet charge and flow behavior confirmed that certain antioxidant placements weakened the invisible forces that normally keep droplets apart and evenly suspended.
Where antioxidants sit changes how well they fight rancidity
To track oxidation, the researchers measured chemical markers of fat breakdown and used standard radical-scavenging tests. Putting gallic acid in the outer water phase (NG) was particularly effective at slowing rancidity, because this region is directly exposed to air and dissolved oxygen. In that position, gallic acid can intercept reactive molecules before they damage the oil. Adding paprika oleoresin to the oil further boosted antioxidant strength by placing protective pigments right where the fat resides. The strongest overall antioxidant capacity appeared when gallic acid was present in both inner and outer water phases (GG) together with paprika oleoresin. Yet, despite this chemical strength, GG samples sometimes showed worse practical oxidation results and poorer physical stability because the added acid lowered pH, reduced droplet charge, promoted layering, and may have nudged gallic acid toward pro-oxidant behavior under very acidic conditions.
Designing better foods means balancing strength and stability
To a non-specialist, the main message is that “more antioxidant” is not always “better,” and where you put it inside a food structure matters just as much as how much you add. Placing water-loving antioxidants mostly in the outer water layer offered a good compromise: strong protection against rancidity while preserving the emulsion’s thickness and resistance to separation. Spreading them across both inner and outer water layers created a chemically powerful shield but destabilized the physical structure. Overall, the study provides a roadmap for food scientists to place natural antioxidants strategically within layered droplets so that everyday products like salad dressings, creamy drinks, and fortified spreads stay tasty, colorful, and stable for longer without sacrificing texture.
Citation: Jeon, S., Jeong, J., Sumnu, G. et al. Contrasting effects of phase-oriented antioxidant localization on oxidative resistance and physical stability of double emulsions. npj Sci Food 10, 66 (2026). https://doi.org/10.1038/s41538-026-00716-8
Keywords: double emulsions, food antioxidants, emulsion stability, paprika oleoresin, gallic acid