Clear Sky Science · en
Metabolomics aided by machine learning decodes adaptive remodeling of Bacillus biofilms in response to pasteurization stress
Why milk drinkers should care
Pasteurized milk is designed to be safe and long‑lasting, yet some hardy bacteria can ride through the heat treatment and quietly build slimy communities—called biofilms—inside processing equipment. This study asks an unsettling question with very practical consequences: can pasteurization sometimes make those biofilms worse, and if so, what hidden chemical changes inside the bacteria are to blame?
Stubborn microbes in modern dairy plants
Milk is rich in nutrients and supports a booming industry of chilled, low‑temperature dairy products. But it also provides a home for Bacillus, a group of bacteria that form tough spores and cling to metal surfaces. These microbes build biofilms—protective layers of cells and glue‑like material—that resist cleaning, slow heat transfer, and raise the risk of spoilage or even foodborne illness. The authors collected 14 Bacillus strains from raw milk on Chinese farms and tested how well they formed biofilms before and after a simulated pasteurization step at 75 °C for 15 seconds. Surprisingly, while many strains formed weaker biofilms after heating, several actually became stronger, stickier builders.
Heat that helps some biofilms, hurts others
To mimic real dairy equipment, the researchers grew selected strains on 304 stainless steel coupons bathed in sterile milk. They then stained and measured the total gunk—bacteria plus milk residues—left on the metal. Two strains, one of Bacillus cereus (BC01) and one of Bacillus subtilis (BS01), shifted from weak to strong adhesion after pasteurization, while closely related cousins (BC02 and BS02) showed the opposite trend. Electron microscope images revealed how the biofilm architecture changed: in heat‑boosted strains, the usual thin fibrous mesh of extracellular polymeric substances fused into thick, blocky aggregates that trapped more cells and milk proteins, forming a tougher, more extensive coating. In heat‑weakened strains, the matrix became sparse and fragmentary.
When surface stickiness breaks the rules
Conventional wisdom holds that the more water‑repelling (hydrophobic) a spore is, the better it sticks and the more easily a biofilm forms. The team tested spore hydrophobicity using an oil‑water system and found the opposite of what textbooks predict. After pasteurization, the strains that gained biofilm strength actually showed lower spore hydrophobicity, while those that lost biofilm capacity became more hydrophobic. Even within biofilms, spores of strong formers were less hydrophobic than their free‑floating counterparts. This contradiction pointed to a deeper driver: heat‑induced shifts in metabolism and gene activity that could overpower simple physical properties like surface stickiness.
Chemical rewiring under heat stress
Using untargeted metabolomics—a broad survey of small molecules inside cells—combined with machine‑learning analysis, the authors mapped how heat reshaped the chemistry of each strain’s biofilm. All four strains showed large changes in hundreds of metabolites, especially in transport systems and amino‑acid pathways, but the details differed sharply. In BC01, heating appeared to activate an enzyme called glutaminase, draining the pool of the milk nutrient L‑glutamine and the amino acid histidine. This shift both supplied building blocks for the biofilm matrix and lifted natural brakes on biofilm formation. Levels of xanthosine, a compound that normally nudges bacteria away from biofilms, also dropped, likely favoring a stable, attached lifestyle. In BS01, heat reduced arginine and several D‑amino acids, dopamine, and arachidonic acid—molecules known from other studies to destabilize biofilms or block their formation. Lower amounts of these inhibitors, together with altered energy metabolism, helped tip the balance toward more robust biofilms. By contrast, BC02 and BS02 suffered shortages of key polysaccharide precursors and energy‑cycle components, and in BS02, anti‑biofilm metabolites such as D‑tryptophan and D‑arabinose increased, collectively undermining biofilm growth.
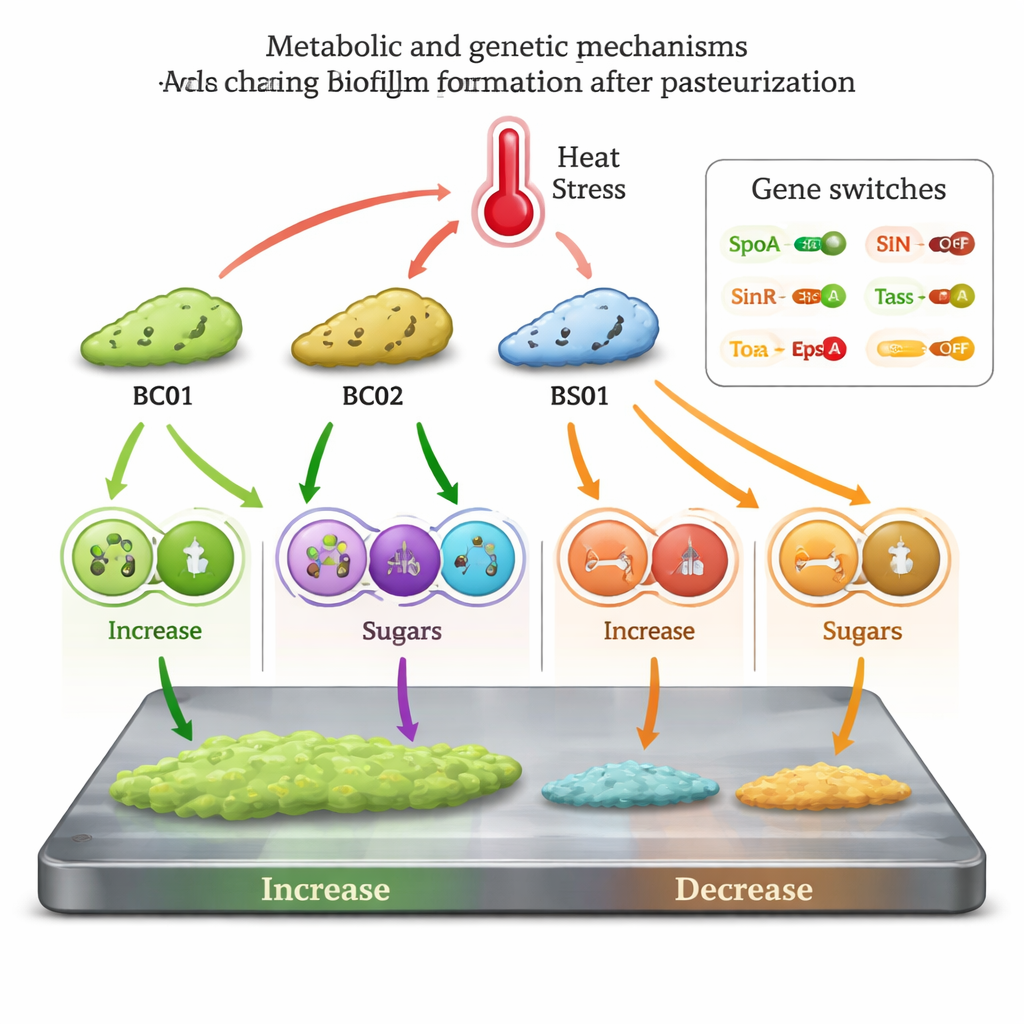
Genes that flip the biofilm switch
To link chemistry with behavior, the team measured key biofilm‑related genes. In the heat‑strengthened strains BC01 and BS01, master regulators that promote biofilm formation (Spo0A, TasA, and EpsA) were turned up, while SinR, a gene that normally keeps biofilm genes in check, was turned down. The opposite pattern appeared in BC02 and BS02. Together, the metabolic data and gene readouts support a model in which pasteurization acts as a stress signal that some Bacillus lineages exploit: they rewire their metabolism, shed certain natural anti‑biofilm molecules, and flip genetic switches to produce more matrix and thicker coatings, even as their overall activity slows.
What this means for safer milk
For consumers, the message is not that pasteurized milk is unsafe, but that the microbes which survive heat treatment can be more adaptable than expected. Rather than simply toughening spores, pasteurization can push some Bacillus strains into a “hunker down” mode where they build stronger biofilms inside pipes and tanks. By pinpointing specific metabolites and gene pathways that drive this shift, the study suggests new ways to fight back: instead of relying only on hotter or longer heating, processors might someday add benign metabolic blockers or tailor cleaning regimes that disrupt the chemical support system of these biofilms. In essence, understanding how heat reshapes bacterial chemistry opens the door to smarter strategies for keeping dairy products both nutritious and reliably clean.
Citation: Liang, L., Wang, P., Zhao, X. et al. Metabolomics aided by machine learning decodes adaptive remodeling of Bacillus biofilms in response to pasteurization stress. npj Sci Food 10, 62 (2026). https://doi.org/10.1038/s41538-026-00712-y
Keywords: pasteurized milk, Bacillus biofilms, food safety, dairy processing, bacterial metabolism