Clear Sky Science · en
Optimizing muscle satellite cell sources for cultured meat: anatomical origin influences cellular properties and quality attributes
Why Growing Meat from Cells Matters
Cultured meat—meat grown from animal cells in tanks instead of on farms—promises steaks and cutlets with far less land use, pollution, and animal suffering. But to turn this vision into supermarket products, scientists must first find the best cells to grow. This study asks a surprisingly simple question with big implications: if you take muscle stem cells from different parts of a pig’s body—neck, back, or leg—do they behave differently, and could that change the taste, texture, and nutrition of future lab-grown pork?
Muscle Cells that Repair—and Build—Meat
Inside every muscle are resident “satellite cells,” a type of stem cell that repairs damaged tissue and helps muscles grow. These same cells are the workhorses of cultured meat: they must multiply many times, then fuse into fibers that resemble real muscle. The researchers isolated these satellite cells from three commonly eaten pork muscles: a neck muscle used for posture, a back muscle that yields popular loin cuts, and a leg muscle that powers movement. Using cell-sorting technology, they purified the satellite cells and confirmed that all three sources produced similarly shaped, spindle-like cells under the microscope. However, the number of satellite cells they could harvest differed: neck muscles yielded the most, back somewhat fewer, and leg the least.
Genes Remember Where They Came From
Although all the cells were grown in the same lab conditions, their internal gene activity patterns still “remembered” their original body location. By sequencing RNA—the working copies of genes—the team showed that neck, back, and leg cells each formed distinct clusters based on which genes were turned on or off. A key part of this difference involved HOX genes, a family of genes that help lay out the body plan during embryonic development. Neck-derived cells expressed HOX genes linked to head and neck regions, while leg-derived cells favored HOX genes associated with hindlimbs. This suggests that positional identity from early development is carried forward into the stem cells used for cultured meat, potentially shaping how they grow and what kind of muscle they form.
Growth Speed Versus Muscle Quality
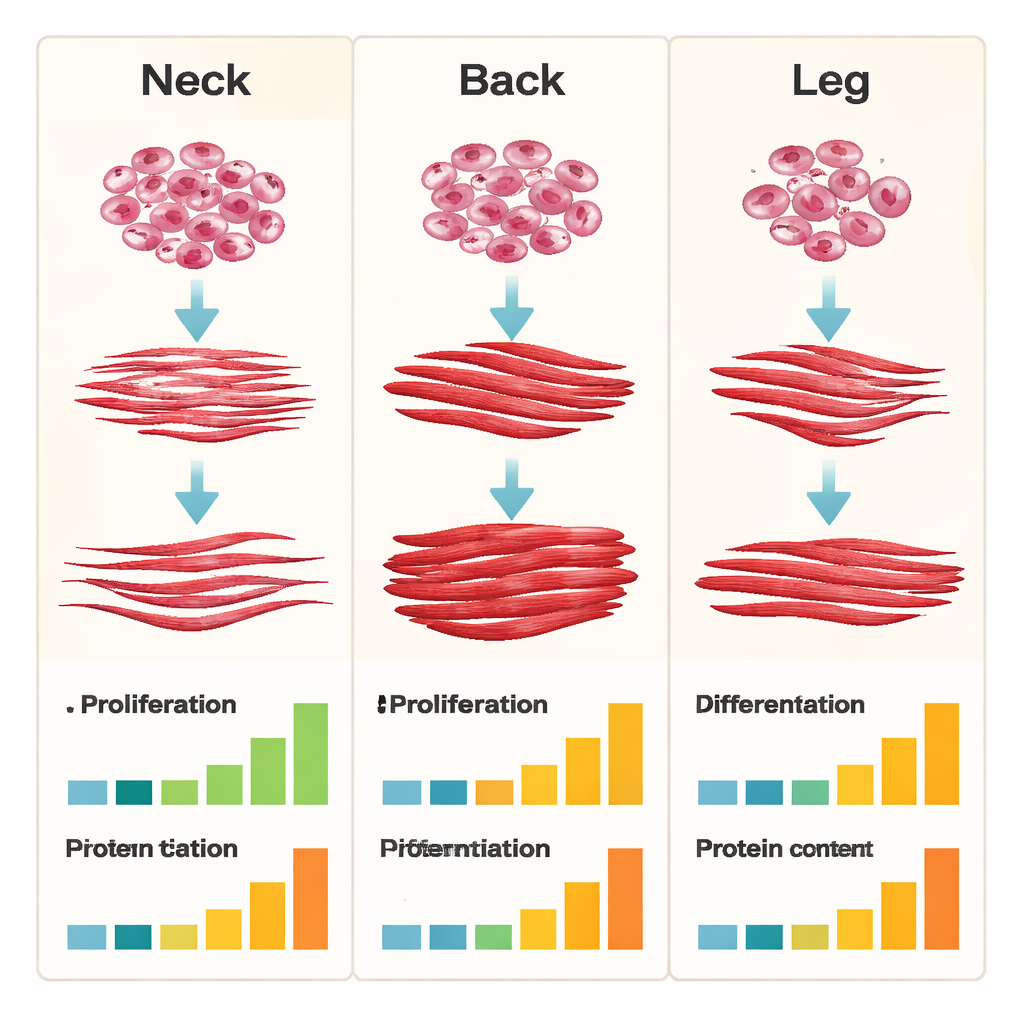
For large-scale meat production, two properties matter: how fast cells multiply, and how well they turn into mature muscle fibers. Neck-derived cells shone in proliferation. Over multiple weeks and passages in culture, they showed the highest levels of growth markers and the strongest overall expansion, making them attractive for quickly generating large numbers of cells. However, when it came time to differentiate—fuse into long, fiber-like structures—the back-derived cells stood out. They formed thicker, more extensively fused myotubes and maintained higher levels of key muscle differentiation genes over long-term culture. Leg-derived cells lagged in both stemness and differentiation; their ability to maintain core muscle stem cell markers and to form robust fibers declined the fastest.
Fiber Types, Protein Levels, and Eating Experience
Not all muscle fibers are the same. Slow-twitch fibers are more endurance-oriented, richer in fat and oxidative metabolism, and are often linked to tenderness and juiciness. Fast-twitch fibers specialize in quick power and tend to be leaner and higher in protein. The researchers found that neck-derived cells were biased toward slow-twitch fiber genes, while back-derived cells preferentially produced fast-twitch fiber markers. When they measured total protein content after differentiation, back-derived cultures had the highest protein levels, whereas neck-derived cultures had the lowest. These differences hint that the starting cell source could tune future cultured meat toward softer, possibly richer cuts (from slow-twitch–favoring cells) or leaner, higher-protein products (from fast-twitch–favoring cells).
Designing Better Cultured Pork
To a non-specialist, the main message is that “where you take the cells from” really matters for lab-grown meat. Neck muscle stem cells are excellent for quickly building up cell numbers, while back-derived cells are better at maturing into thick, protein-rich muscle fibers and keep their muscle identity over many rounds of growth. Leg-derived cells contribute less on both counts. The authors suggest that back-derived satellite cells offer the best overall balance for producing cultured pork, but that mixing sources might allow producers to customize texture, nutrition, and flavor. In other words, tomorrow’s cultured chops could be engineered from the cell up—starting with the right piece of the pig.
Citation: Lee, J., Park, J., La, H. et al. Optimizing muscle satellite cell sources for cultured meat: anatomical origin influences cellular properties and quality attributes. npj Sci Food 10, 56 (2026). https://doi.org/10.1038/s41538-026-00706-w
Keywords: cultured meat, muscle stem cells, pig satellite cells, meat texture, sustainable protein