Clear Sky Science · en
Transcriptomic comparison of human intestinal organoids and Caco-2 cells in modeling nutrient absorption: insights from infant formula and breast milk
Feeding Babies and Testing Their Food
Parents and scientists alike want to know how closely infant formula can match the benefits of breast milk, especially for a baby’s growing gut. This study looks under the hood of both breast milk and several commercial formulas, not in real infants, but in advanced lab-grown mini-intestines. By comparing how these feeding options interact with realistic gut models, the researchers explore which lab system best mimics a baby’s intestine and how different formula processing methods might influence growth, immunity, and long‑term safety.
Mini-Guts in a Dish
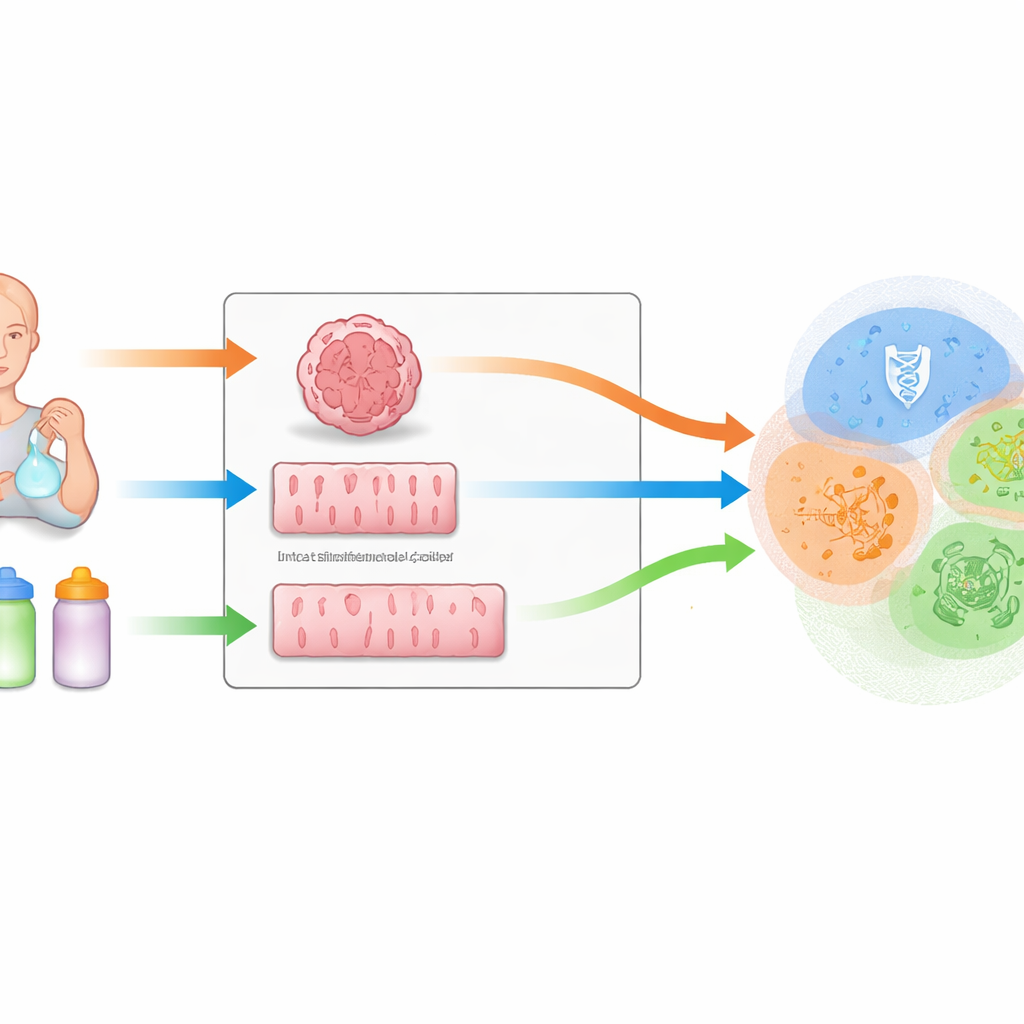
To study infant nutrition safely and in detail, the team used two lab models of the human intestine. One is a long-standing workhorse: Caco-2 cells, a flat sheet of identical gut-like cells. The other is newer and more lifelike: three-dimensional human small intestinal organoids, often called “mini-guts.” These organoids are grown from stem cells and contain a mix of cell types normally found in the intestine, including cells that absorb nutrients, secrete mucus, and support immune functions. Careful staining and microscopy confirmed that the organoids formed organized, hollow structures with diverse, actively dividing cells, making them a promising stand‑in for the infant gut.
Putting Breast Milk and Formula Through Digestion
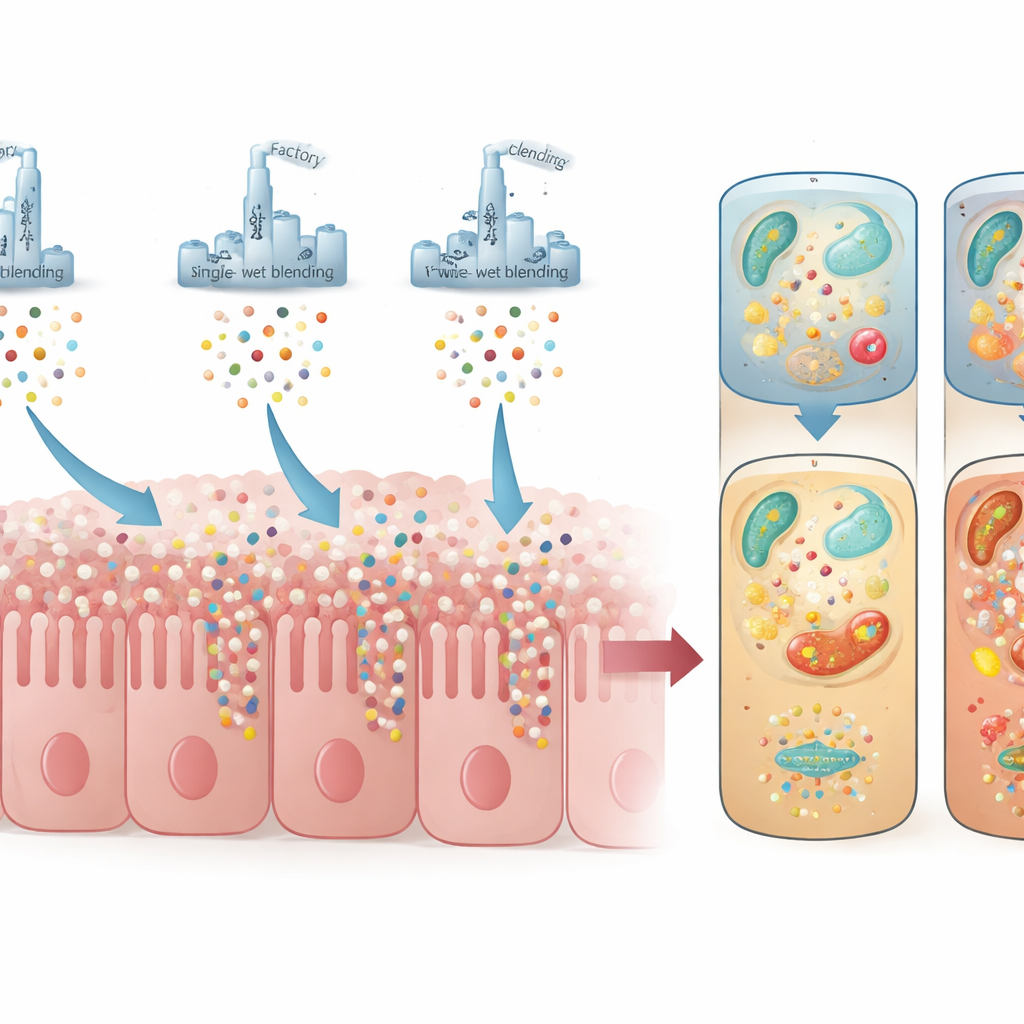
The researchers then simulated what happens to milk inside a baby’s stomach and small intestine. They took breast milk from seven healthy donors and three brands of stage‑1 infant formula, each produced with a different industrial method: dry blending (IFA), single-step wet blending (IFB), and two-step wet blending (IFC). All samples were run through a lab digestion system that mimics infant gastric and intestinal conditions. The resulting digested fluids were applied to either organoid or Caco‑2 cell layers, and the team measured how thousands of genes in these cells switched on or off using RNA sequencing.
Which Lab Model Acts More Like a Real Gut?
When the scientists compared gene activity patterns, the organoid model clearly outperformed Caco‑2 cells. Organoids showed larger shifts away from untreated controls, cleanly grouped samples by feeding type, and produced many more genes that changed in response to breast milk and formulas. Crucially, gene pathways related to tissue growth, organ shaping, and other developmental processes were strongly activated in organoids but were much less consistent in Caco‑2 cells. At the same time, neither model showed strong activation of cell damage pathways at the doses used, suggesting the exposures themselves were not overtly toxic. Together, these findings indicate that mini-guts capture the fine details of how infant foods influence the intestine far better than traditional flat cell layers.
How Formulas Differ from Breast Milk and from Each Other
Using the organoid data, the team compared breast milk with the three formula brands. Many gene changes were shared, showing that formulas broadly support growth- and metabolism-related pathways. Still, important differences emerged. Breast milk had a distinct molecular signature and shared numerous beneficial pathways with one formula in particular, underscoring why it remains the nutritional gold standard. All formulas boosted genes involved in tissue development and cell junctions, which help maintain the gut barrier. Yet each brand had its own profile: IFA particularly enhanced fat-building processes and ion and organic acid transport; IFB strongly supported tiny hairlike structures on cells (cilia) and the surrounding support matrix; and IFC stood out for genes that dial down excessive inflammation and promote secretion, patterns that may be linked to its inclusion of human milk oligosaccharides.
Hidden Costs of Processing
Not all differences were positive. IFC, the product made by two-step wet blending, also triggered stronger responses related to toxic substances and oxidative stress—chemical wear and tear on cells. Follow‑up metabolite measurements confirmed higher levels of lipid breakdown products associated with oxidation in IFC compared with the other formulas. In organoids, IFC stimulated genes tied to stress and inflammatory signaling more than the dry‑blended or single‑step wet‑blended products. While the recipes of commercial powders are not identical, the data point to the more complex, heat‑intensive two-step process as a likely contributor to this added oxidative burden.
What This Means for Babies’ Food
In plain terms, this work shows that advanced mini-gut models are better than traditional flat cell cultures for revealing how infant foods shape the intestine at the molecular level. Breast milk still comes out on top, with a unique pattern of signals that support growth and immune balance. Modern formulas do activate many of the same beneficial pathways, but their manufacturing methods matter: gentler processing appears to limit the build‑up of oxidation-related byproducts, whereas more intensive, multi-step heating may carry added long‑term risks. The study suggests that organoids can guide the design of safer, more baby‑friendly formulas and encourage industry to refine processing techniques, all while reinforcing the value of breast milk whenever it is available.
Citation: Wang, X., Zhang, W., Yang, S. et al. Transcriptomic comparison of human intestinal organoids and Caco-2 cells in modeling nutrient absorption: insights from infant formula and breast milk. npj Sci Food 10, 101 (2026). https://doi.org/10.1038/s41538-025-00672-9
Keywords: infant formula, breast milk, intestinal organoids, nutrient absorption, oxidative stress