Clear Sky Science · en
Adipose-derived dual cell therapy enhances arteriogenesis and limb preservation through vascular integration in critical limb ischemia
Saving Legs by Growing New Blood Vessels
When arteries in the legs slowly clog, people can develop crippling pain, stubborn ulcers, and even face amputation. This severe stage, called critical limb ischemia, is notoriously hard to treat because many patients cannot undergo bypass surgery or other standard procedures. The study summarized here explores a new strategy: using two types of stem-like cells taken from a person’s own fat to help the body grow sturdy new blood vessels that can rescue starved limb tissue.
A Common Disease with Limited Options
Peripheral artery disease occurs when arteries in the legs narrow, choking off blood flow. In its worst form, critical limb ischemia, people may suffer constant pain, blackened toes, and a high risk of losing part of the limb. Current therapies such as cholesterol-lowering drugs, angioplasty, or surgical bypass can help, but many patients either do not respond or are too sick for these procedures. Earlier attempts to use single stem cell types have mostly encouraged fragile, tiny vessels that do not provide enough flow to reliably save the limb. Researchers therefore set out to build a more complete “replacement plumbing system” by combining two cooperating cell types.
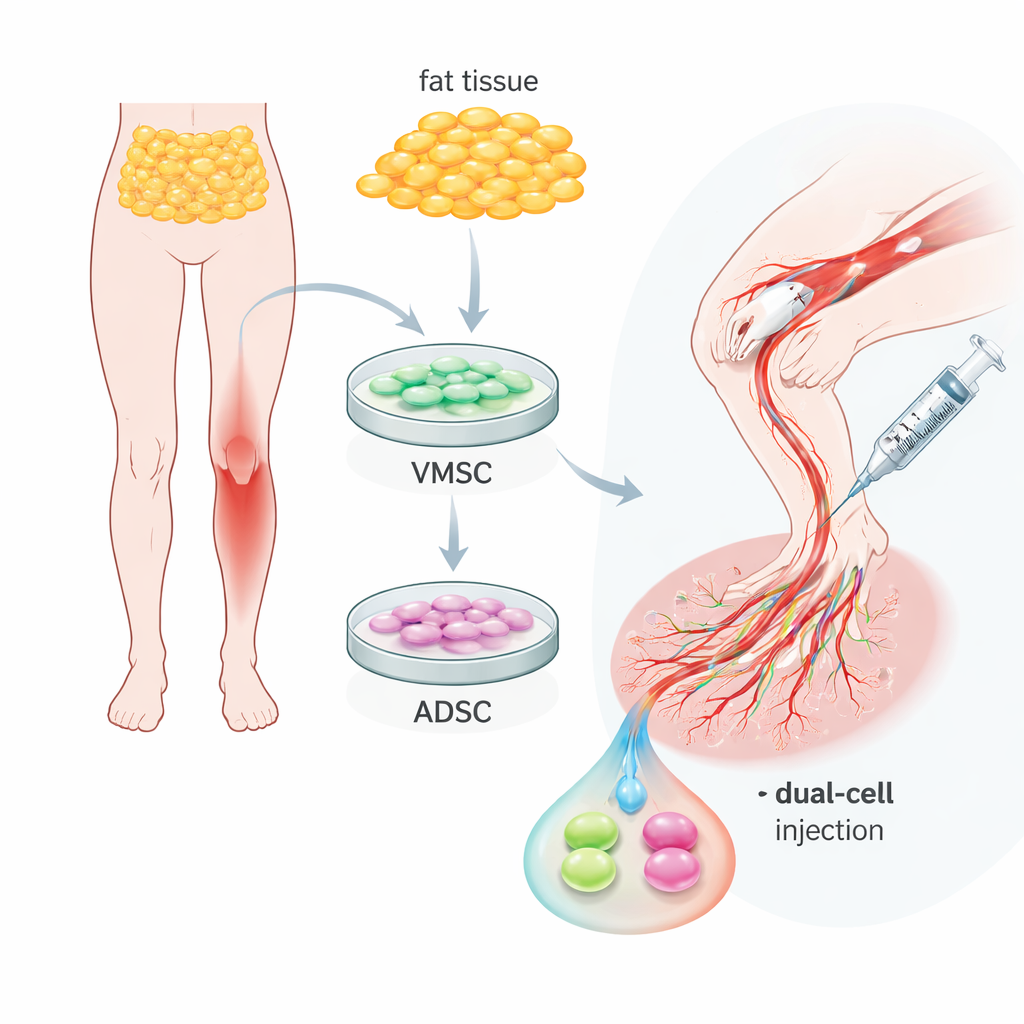
Two Helpful Cell Types in Ordinary Body Fat
The team focused on adipose tissue—ordinary body fat—which can be collected with relatively minor procedures. From the same sample, they isolated two related but distinct cell populations. One group, called vascular multipotent stem cells, behaved much like early blood vessel lining cells: they carried surface markers typical of endothelial cells and could organize into capillary-like tubes in lab dishes. The second group, adipose-derived stem cells, looked more like supporting cells that wrap around vessels, carrying markers seen in smooth muscle and perivascular cells. Both types could multiply well and turn into fat, bone, or cartilage cells, confirming their flexibility, but their natural roles around blood vessels were clearly different and complementary.
Working Together Under Stress
To mimic the harsh, inflamed environment of a diseased limb, the researchers exposed the cells to a strong inflammatory signal. Both cell types survived and adjusted their behavior by releasing mixtures of growth factors and enzymes that help cells move and reshape surrounding tissue. When grown alone on a gel that encourages vessel formation, the endothelial-like cells could form basic tubes, while the support cells mostly clumped. However, when mixed in a two-to-one ratio, the tubes became denser, more continuous, and more resistant to inflammatory stress. This suggested that, together, the two cell types can form not only more vessels, but more mature and stable networks.
Testing Dual-Cell Therapy in Diseased Mouse Limbs
The scientists then moved to a mouse model of critical limb ischemia created by cutting off a main leg artery. Into the starved leg muscle, they injected the mixed human cells. Over four weeks, untreated mice commonly lost toes or entire feet, and their muscles showed scarring and heavy inflammation. In contrast, animals that received the dual-cell treatment largely kept their limbs. Blood-flow scans showed that circulation in treated legs rebounded to about 70 percent of normal, compared with less than 20 percent in controls. Microscopic and three-dimensional imaging revealed why: treated muscles contained many more medium and large vessels—arteriole-like branches—with strong “sleeves” of supporting cells, rather than just a tangle of tiny, unstable capillaries.
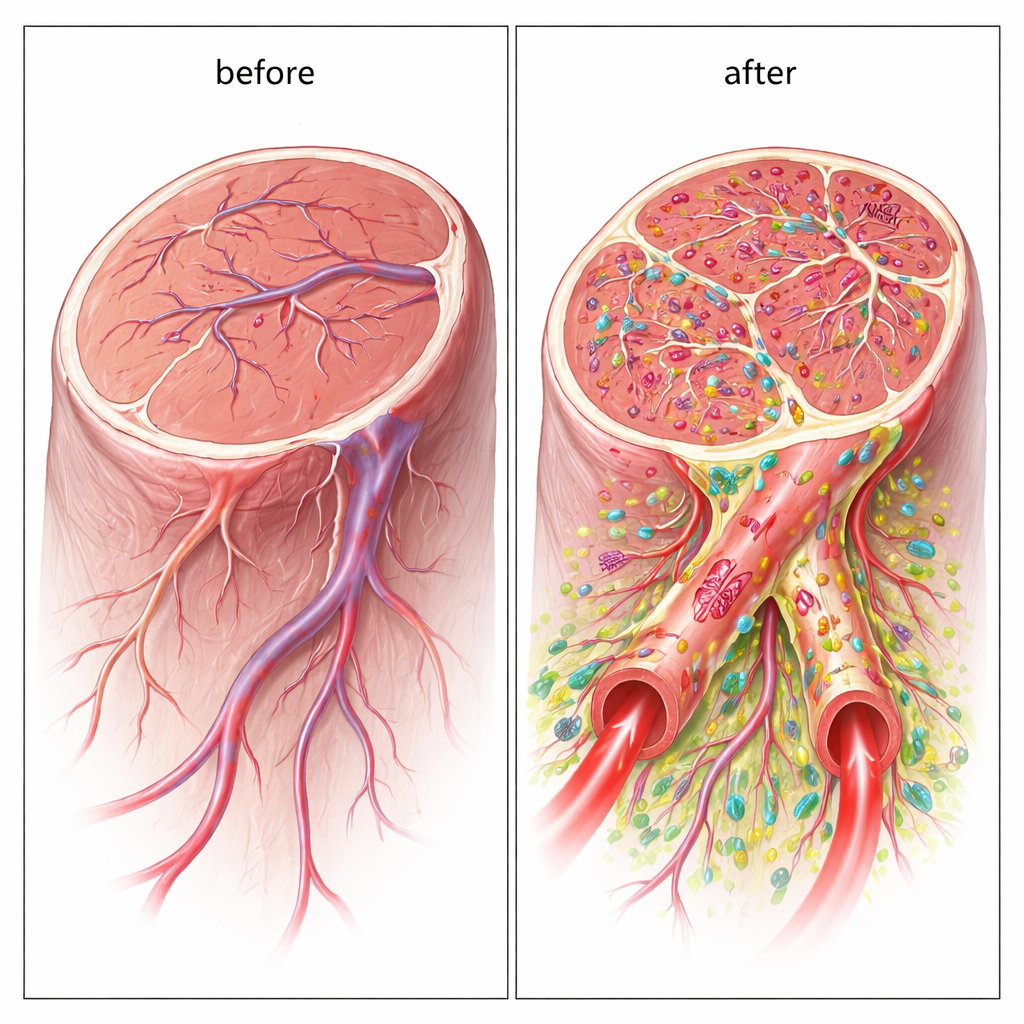
Hybrid Vessels Built from Donor Cells
Importantly, the new vessels were not just encouraged by the cells; they actually contained the transplanted human cells as part of their walls. Using human-specific markers, the researchers found that the endothelial-like cells helped form the inner lining, while the support cells wrapped around the outside. Many of these hybrid vessels were wide enough to function as proper conduits for blood, and fluorescent tracers confirmed that blood flowed through them. This direct building of large, perfused vessels sets the dual-cell approach apart from most previous stem cell therapies, which mainly rely on chemical signals rather than long-lasting structural integration.
What This Could Mean for Patients
To a non-specialist, the message is that scientists may be learning how to “re-pipe” dying limbs using a person’s own fat-derived cells. By pairing two cell types—one that forms the inner tube and one that builds the outer support—the therapy generates strong, well-lined arteries and arterioles rather than fragile sprouts. In mice, this was enough to restore blood flow and prevent limb loss. While more work is needed to prove long-term safety and effectiveness in people, the study points toward a future in which a minimally invasive fat harvest could provide customized cell mixtures to rebuild circulation in patients at high risk of amputation.
Citation: Kim, D.Y., Hwang, D.Y., Park, G. et al. Adipose-derived dual cell therapy enhances arteriogenesis and limb preservation through vascular integration in critical limb ischemia. npj Regen Med 11, 13 (2026). https://doi.org/10.1038/s41536-026-00458-x
Keywords: critical limb ischemia, stem cell therapy, adipose-derived cells, neovascularization, peripheral artery disease