Clear Sky Science · en
Novel copper-ion coordinated andrographolide-loaded hydrogel activates Rac1/JNK1 axis for enhancing diabetic wound healing
Why stubborn wounds matter
For many people with diabetes, a small blister on the foot can spiral into a chronic wound that refuses to close, sometimes ending in amputation. These hard-to-heal sores are fueled by infection, runaway inflammation, and poor blood supply. The paper behind this summary describes a new gel-like dressing that aims to tackle all three problems at once, using a plant compound and a trace metal to coax damaged skin back toward healthy repair.
A smart bandage built from gel
The researchers created a soft, water-rich material known as a hydrogel, designed to sit directly on a wound. It is made from gelatin (a collagen-derived protein) and a modified form of chitosan, a sugar-based polymer. Copper ions act like tiny connectors, linking the components together into a flexible network while also binding a plant molecule called andrographolide, known for its anti-inflammatory, antimicrobial, and blood-sugar-lowering effects. This copper coordination greatly improves the drug’s water solubility and allows it to be held evenly within the gel rather than clumping as crystals, which is crucial for steady release into the wound.
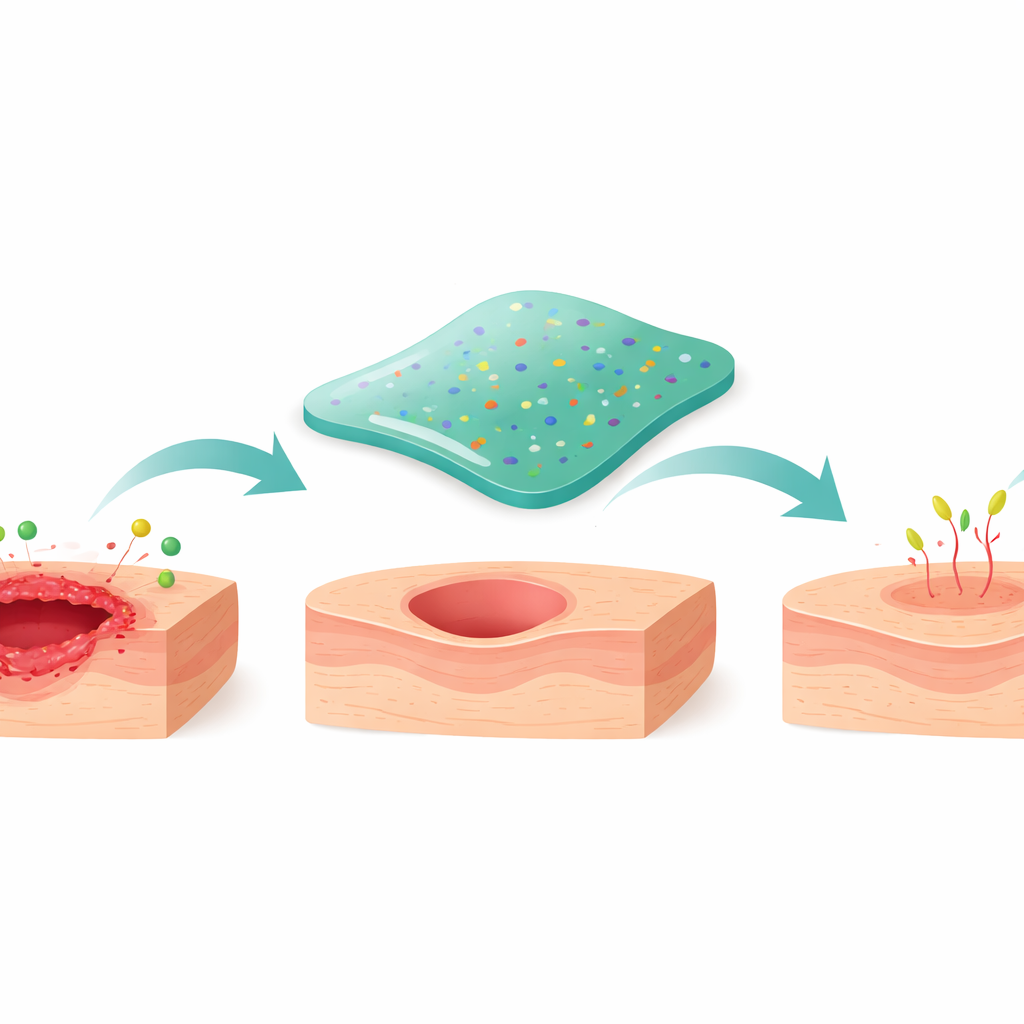
Built to stay put and self-repair
A wound dressing must cling to wet, moving skin, soak up fluid, and still remain intact. Detailed physical tests showed that the optimized version of the gel (called ASFH-L) behaves like a soft solid: it can deform to match the wound surface yet resists flowing away. Under the microscope, it displays an interconnected pore structure large enough for nutrients and cells to move through, but not so loose that it falls apart. When stretched or briefly damaged, its copper-based links re-form, giving the hydrogel a “self-healing” ability that lets a cut piece fuse back together within minutes. On moist pig skin, ASFH-L stayed attached through repeated bending and twisting under water, suggesting it could protect real wounds without constant re-application.
Fighting germs and helping skin cells move
Chronic diabetic wounds are often crowded with bacteria. In lab tests against common culprits such as Staphylococcus aureus and Escherichia coli, all versions of the hydrogel slowed bacterial growth, and those carrying more andrographolide produced larger kill zones. At the same time, the extracts from ASFH-L were gentle to human skin cells, keeping them viable and even promoting their growth over a 24-hour period at suitable doses. When skin cells were cultured in high sugar conditions that usually blunt their ability to move, adding the hydrogel extracts helped them migrate into scratch gaps more quickly. This motility boost is important, because the closing of a wound depends on skin cells crawling to cover the exposed area.
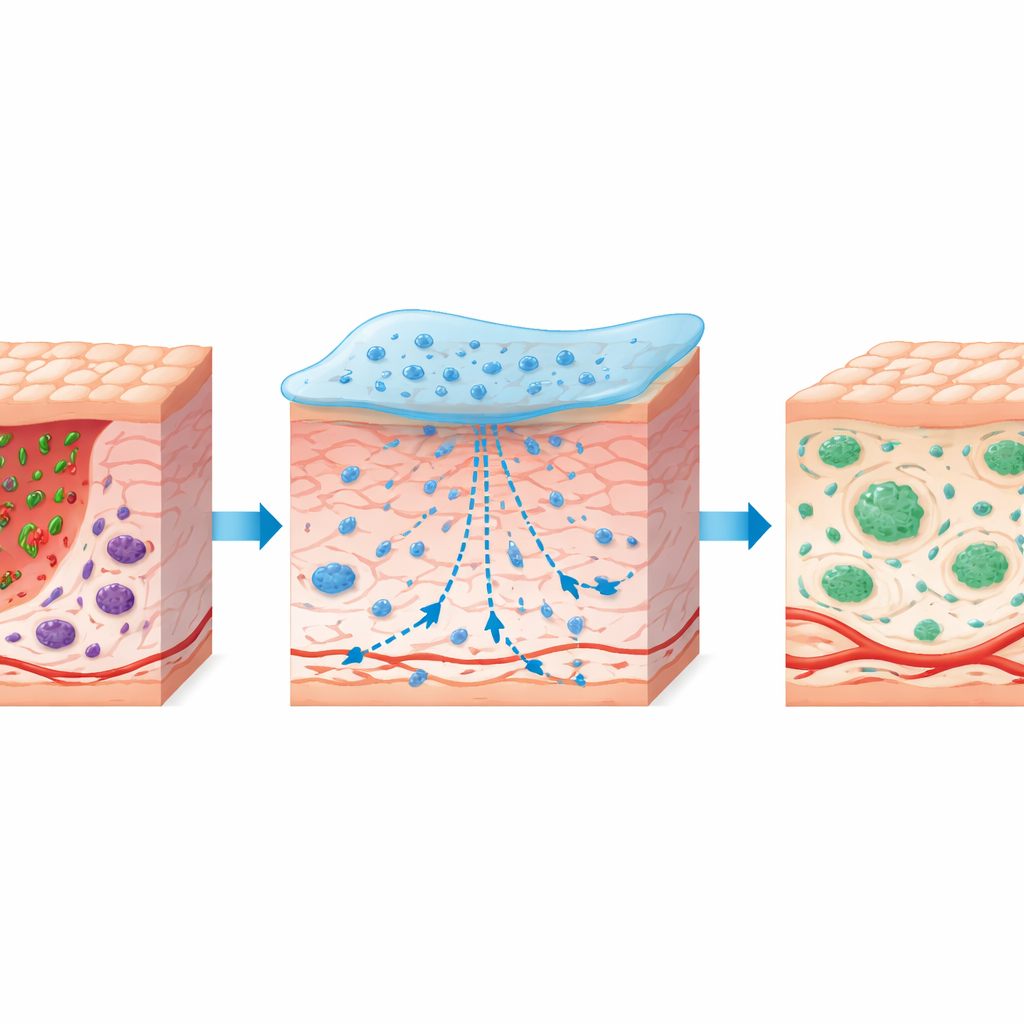
Guiding the body from inflammation to repair
In diabetic mice with circular back wounds, the ASFH-L hydrogel sped up closure compared with untreated controls and with a similar gel lacking the plant drug. Treated wounds showed thicker new outer skin, more orderly collagen fibers, and even the regrowth of hair follicles. Blood flow imaging and tissue staining revealed denser networks of small blood vessels and more supporting muscle-like cells around them, suggesting better nutrient delivery. At the immune level, the dressing altered the behavior of macrophages—front-line defender cells that can either stoke inflammation (M1 type) or calm it and support rebuilding (M2 type. ASFH-L reduced the pro-inflammatory M1 population and expanded the pro-healing M2 group, while lowering harmful signals such as IL‑1β and TNF‑α and increasing the soothing messenger IL‑10. This coordinated shift mirrors the natural transition from an early “cleanup” phase toward true tissue repair, which is often stalled in diabetic wounds.
A molecular switch for healing
To understand how a plant compound in a gel could orchestrate these effects, the team used computer modeling, database mining, and biophysical measurements. They found that andrographolide can directly bind to a small signaling protein called Rac1 and, through it, activate a chain of molecules (JNK1, Jun, and Fos) that collectively regulate cell movement, growth, inflammation, and new vessel formation. Simulations showed stable, energetically favorable binding between andrographolide and Rac1, and surface-based experiments confirmed this interaction in real time. In the mouse wounds, genes and proteins along this Rac1/JNK1/Jun/Fos axis were more active in the hydrogel-treated group. Put simply, the dressing does not just cover the wound; it delivers a botanical ingredient that flips on a built-in repair switch inside cells.
What this could mean for patients
This work introduces a “smart” dressing that combines infection control, immune calming, and blood vessel growth into a single, self-healing hydrogel platform. By using copper ions to solubilize and slowly release andrographolide, the material turns a difficult-to-deliver plant compound into a practical local therapy. In diabetic animals, this approach speeds closure, organizes scar tissue, and rebalances the immune response, all while tapping into a defined molecular pathway. Although human trials are still needed, the concept points toward future bandages that do far more than cover a wound—they actively coach damaged tissue through each step of the healing journey.
Citation: Ye, P., Dai, Y., Zhang, Q. et al. Novel copper-ion coordinated andrographolide-loaded hydrogel activates Rac1/JNK1 axis for enhancing diabetic wound healing. npj Regen Med 11, 14 (2026). https://doi.org/10.1038/s41536-026-00457-y
Keywords: diabetic wound healing, hydrogel dressing, andrographolide, macrophage polarization, angiogenesis