Clear Sky Science · en
Inhibition of CDK8 rescues impaired ischemic fracture healing
Why blood flow matters when bones break
Most broken bones eventually knit themselves back together, but for millions of people each year, that process stalls. Poor blood flow around a fracture—known as ischemia—dramatically raises the risk that a break will heal slowly, badly, or not at all. This study explores why that happens at the cellular level and tests an experimental pill that appears to nudge the body away from scar-like repair and back toward true bone regeneration.
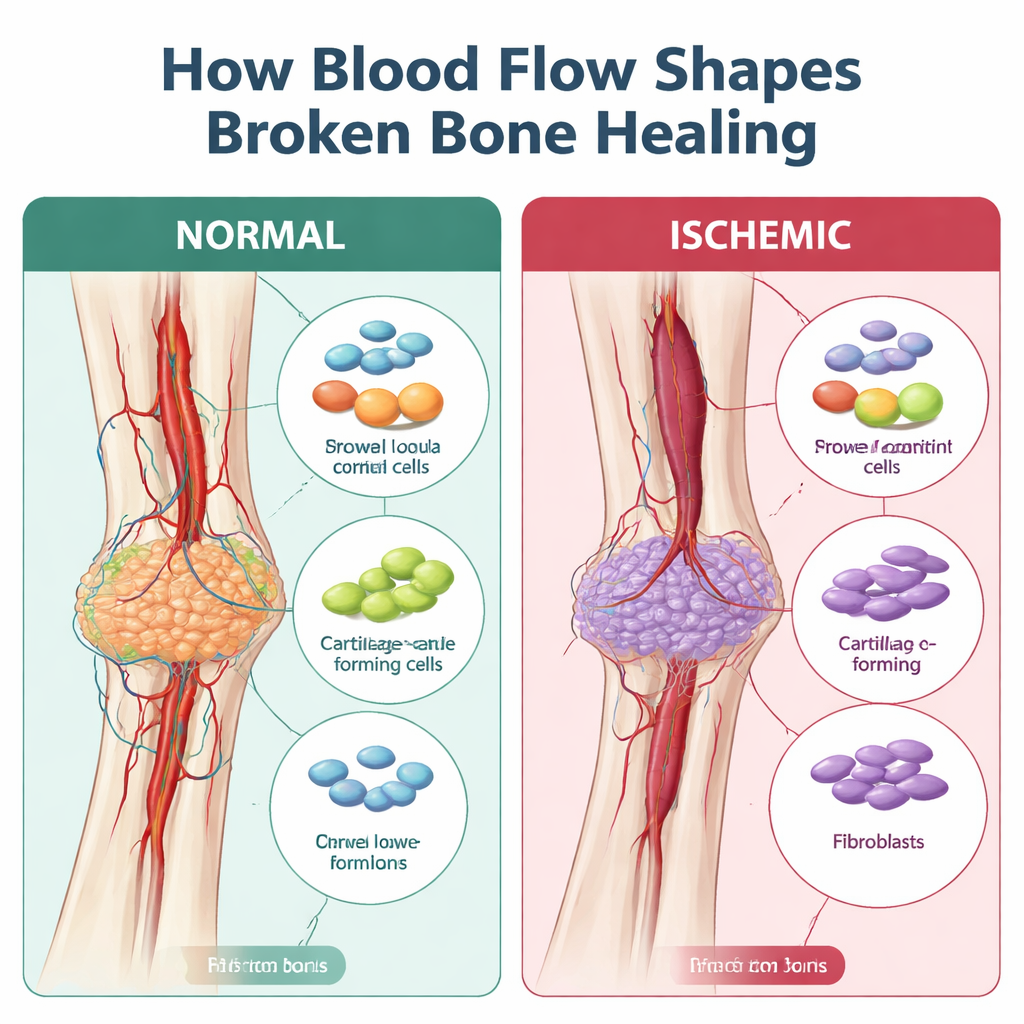
When healing turns into scarring instead of rebuilding
Bone normally heals by building a soft bridge of cartilage that is gradually replaced by hard bone. This bridge, called the callus, depends on a supply of stem-like support cells, immune cells, and blood vessels that coordinate the repair. In ischemic fractures, earlier work showed that the callus tends to be smaller, weaker, and filled with fibrous tissue, more like a scar than a sturdy patch of new bone. What was missing was a detailed map of which cells go wrong, and when, during this detour from proper healing.
Single cells reveal a detour into fibrosis
The researchers used a mouse model in which a leg artery is surgically damaged at the time of fracture, closely mimicking the loss of blood flow seen in severe injuries or in people with vascular disease or who smoke. They then applied single-cell RNA sequencing, a technique that reads out the active genes in thousands of individual cells, to fracture calluses four and seven days after injury. In well-perfused fractures, the early callus was packed with immune cells that soon gave way to expanding stromal cells—the support cells that become cartilage and bone. Under ischemic conditions, however, there were far fewer cartilage-forming and bone-forming cells and many more fibroblasts, the cells that lay down fibrous tissue. Computational “trajectory” analysis showed that, instead of smoothly maturing from early progenitors into cartilage, stromal cells in ischemic bone often diverted through a fibroblast-like state, matching the more scar-like callus seen under the microscope.
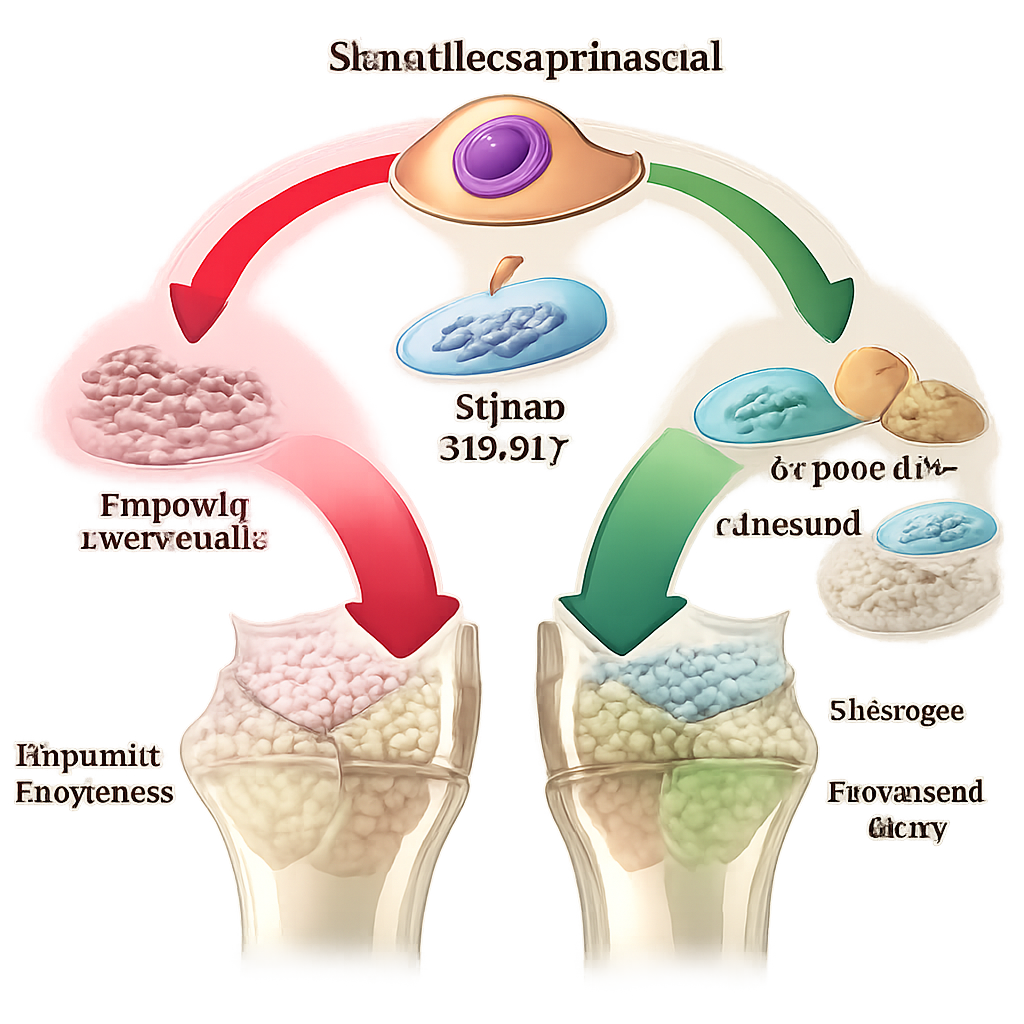
A molecular brake on bone-building cells
Digging into the gene activity of these cells, the team found a strong signal of cellular stress in ischemic calluses, including high levels of heat shock proteins. Among the most striking changes was a surge in activity of a gene called Cdk8, which encodes a regulatory enzyme that sits in a major transcription complex and can slow or redirect how cells respond to signals. Cdk8 has been known to block cell differentiation in other contexts. Here, it was especially elevated in early stromal progenitors in ischemic bone. Human mesenchymal stromal cells grown in low-oxygen lab conditions—mimicking poor blood flow—also ramped up CDK8, linking oxygen shortage directly to this potential “brake” on cartilage and bone formation.
Turning off CDK8 to rescue repair
The researchers next asked whether blocking CDK8 could free stromal cells to resume proper healing. In cell culture, treating mouse and human progenitor cells with selective CDK8 inhibitors boosted key cartilage genes and increased production of cartilage matrix, and also enhanced mineralization when the same cells were pushed toward becoming bone. The team then tested an orally available CDK8/19 inhibitor called SNX631-6 in mice with ischemic fractures. When given during the early, cartilage-forming window, the drug increased the amount of cartilage in the callus. When treatment was extended into the later transition from cartilage to bone, the calluses showed more total bone, higher mineral content, and larger overall size on micro–CT scans and tissue sections, indicating more robust structural repair.
From lab insight to possible new therapies
Taken together, the results suggest that ischemia steers healing cells toward a stressed, fibrotic fate in part by dialing up CDK8, which restrains their ability to become cartilage and bone. Blocking this enzyme appears to release that brake, allowing more cartilage to form early and more bone to be laid down later, even when blood supply is compromised. Because CDK8 inhibitors are already being tested in cancer patients, they may one day be repurposed to help people at high risk of poor fracture healing—such as older adults, smokers, or those with vascular disease—regrow strong bone instead of fragile scar tissue.
Citation: Capobianco, C.A., Song, M.J., Farrell, E.C. et al. Inhibition of CDK8 rescues impaired ischemic fracture healing. npj Regen Med 11, 12 (2026). https://doi.org/10.1038/s41536-026-00456-z
Keywords: bone fracture healing, ischemia, CDK8 inhibitor, mesenchymal stromal cells, fibrosis