Clear Sky Science · en
Introducing PIGMO, a novel PIGmented MOuse model of Parkinson’s disease
Why coloring brain cells may matter to you
Parkinson’s disease is best known for its tremors and stiffness, but deep inside the brain it is linked to a dark pigment that slowly builds up in certain nerve cells. The new study introduces a mouse model called PIGMO that deliberately turns those key cells brown‑black, much like in the human brain. By doing so in a controlled and gradual way, researchers can watch Parkinson‑like damage unfold step by step and test treatments aimed not just at easing symptoms, but at slowing or stopping the disease itself. 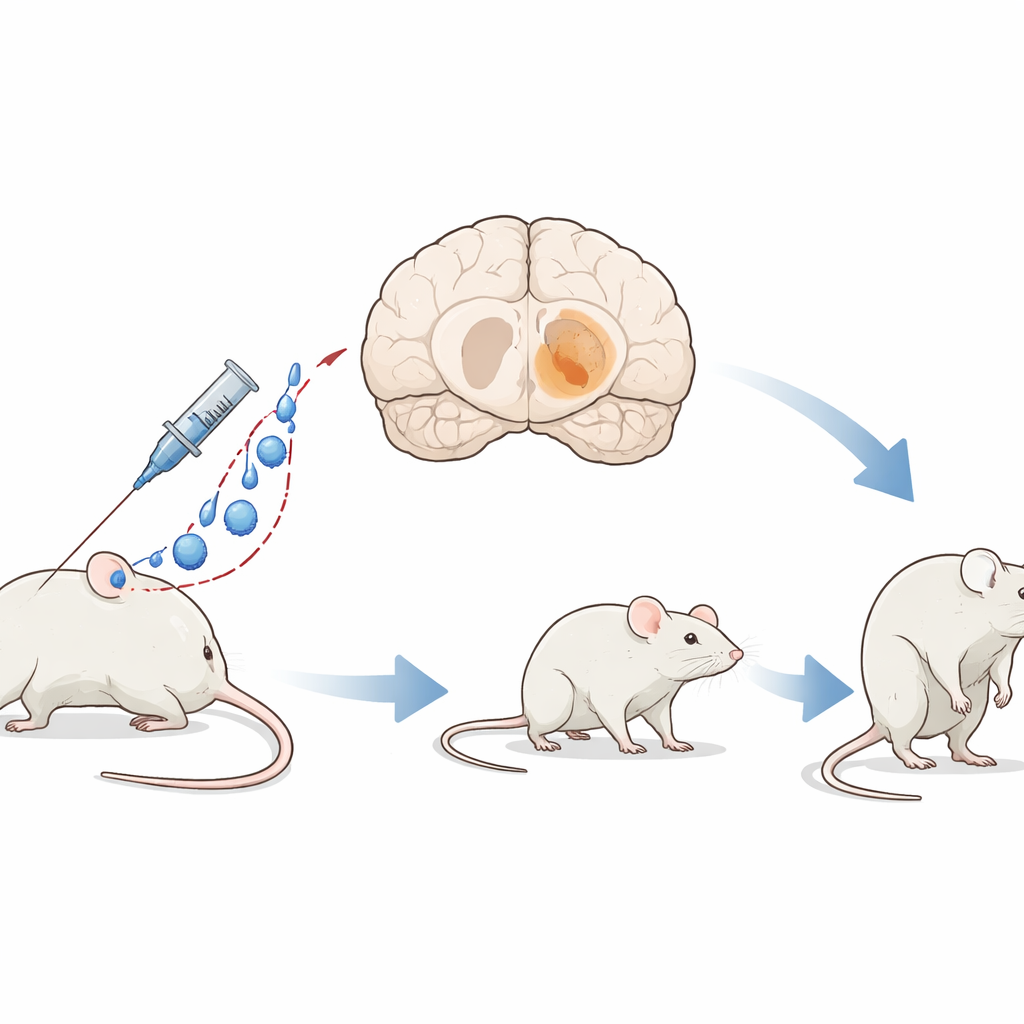
Building a darker brain without brain surgery
To create PIGMO, the team used a modified gene‑delivery virus that can travel through the bloodstream, cross the blood–brain barrier, and enter brain cells. This virus carries the recipe for human tyrosinase, an enzyme that makes neuromelanin, the dark pigment found in the nerve cells most affected in Parkinson’s disease. Instead of drilling into the skull, the researchers injected the virus into a vein behind the eye, letting it spread widely and bilaterally through the brain. This simple, surgery‑free approach produced a mouse whose deep movement centers slowly darken and age in ways that closely resemble the human condition.
Stepwise darkening of key movement centers
Once the virus was delivered, pigment began to appear in three brain hubs that control movement and alertness: the substantia nigra, the ventral tegmental area, and the locus coeruleus. After one month, only a few cells showed faint color, but by four months these regions were visibly dark to the naked eye. The pigment continued to build over eight and twelve months, although the total number of pigmented cells in some areas began to fall, hinting that the most heavily loaded cells were starting to die. Not all nerve cells were equally affected: specific subtypes within the substantia nigra took up far more pigment than others, echoing the selective vulnerability seen in people with Parkinson’s disease.
From pigment to clumps and dying connections
As pigment levels rose past a certain threshold, the affected cells began to form dense clumps of the protein alpha‑synuclein—structures that closely resemble the Lewy bodies found in human patients. These inclusions appeared first in the substantia nigra after about four months and later in the other pigmented regions, becoming more common with time. In parallel, the long projections from these cells to the striatum, a key relay for movement commands, steadily thinned out. Measurements showed a marked loss of dopamine‑rich fibers and a shrinking population of healthy dopamine cells, particularly in the substantia nigra, with “ghost” cells—pigmented but functionally compromised—being especially prone to disappear. 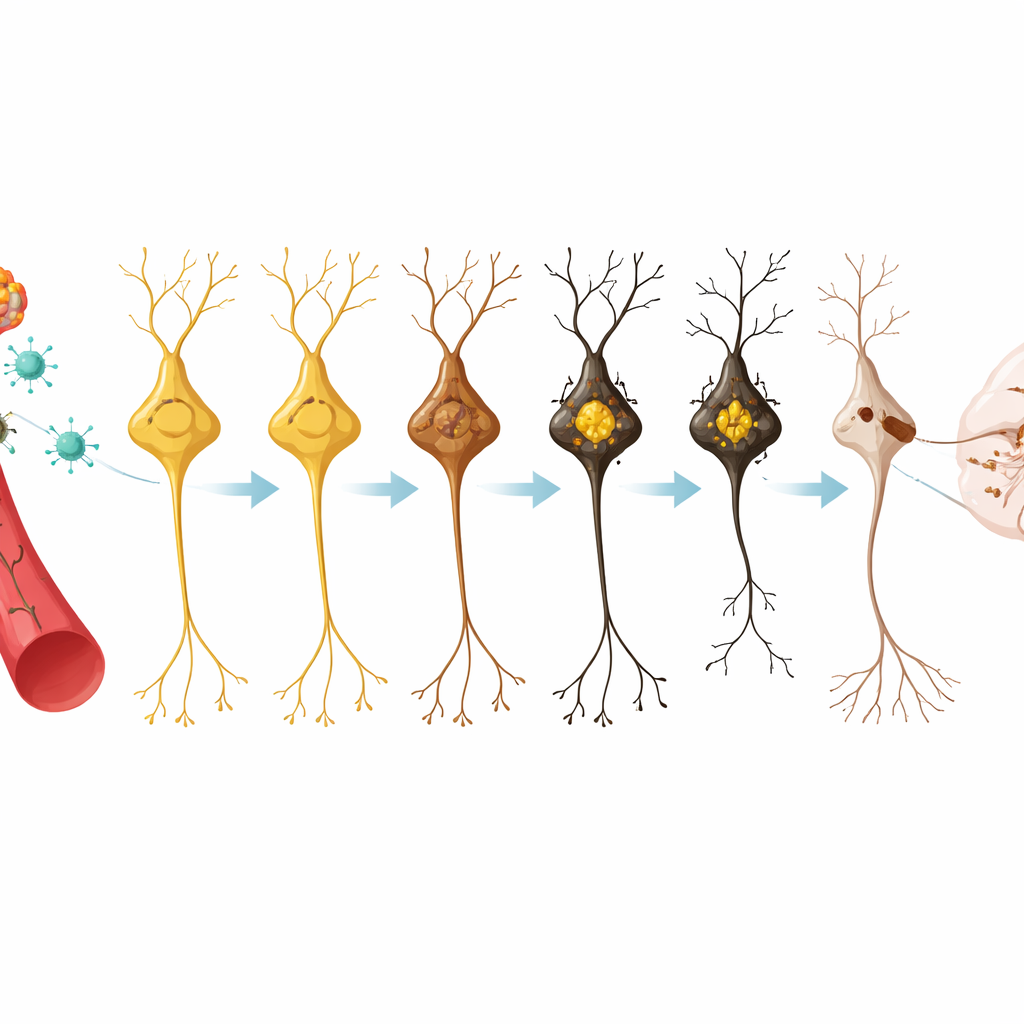
Watching movement falter over time
The outward behavior of the PIGMO mice reflected this inner damage. Animals were tested on a rotating rod and in a simple posture test that measures rigidity. Early on, their performance was similar to that of control mice, but as months passed and pigment, clumps, and cell loss mounted, the PIGMO mice showed clear motor problems. By twelve months, they stayed on the rotating rod for much shorter times and were markedly stiffer, mirroring the gradual onset and progression of movement difficulties in human Parkinson’s disease.
A new test bed for slowing Parkinson’s disease
For non‑specialists, the key message is that the PIGMO mouse brings researchers closer to a realistic, progressive version of Parkinson’s disease in the lab. By turning on pigment production in the very cells that fail in patients—and doing so across the whole brain without surgery—this model reproduces pigment buildup, protein clumping, nerve fiber loss, and motor symptoms in a predictable timeline. That long window, from first pigment specks to clear disability, offers a crucial chance to test new drugs or gene therapies after the disease has quietly begun but before damage is irreversible, potentially speeding the search for treatments that truly slow or prevent Parkinson’s progression.
Citation: Chocarro, J., Marana, S., Espelosin, M. et al. Introducing PIGMO, a novel PIGmented MOuse model of Parkinson’s disease. npj Parkinsons Dis. 12, 72 (2026). https://doi.org/10.1038/s41531-026-01289-9
Keywords: Parkinson’s disease, animal model, neuromelanin, gene therapy, dopamine neurons