Clear Sky Science · en
Olfactory sensory map is perturbed in a human wild-type α-synuclein overexpressing transgenic mouse model of Parkinson’s disease
Why Smell Loss Matters in Parkinson’s
Many people think of Parkinson’s disease as a movement disorder, but for most patients, the first thing to go wrong is their sense of smell. This loss can appear decades before tremor or stiffness, making it a powerful early warning sign. The study described here asks a basic yet unanswered question: what exactly goes wrong in the brain’s smell circuitry when Parkinson’s-related changes begin? By probing this question in a carefully engineered mouse model, the researchers uncover how a key Parkinson’s protein disrupts the brain’s internal “smell map” and point toward a new, easily accessible source of early disease clues.
The Brain’s Smell Map
Our noses contain millions of specialized smell cells that each respond to particular odor molecules. These cells send long, thin fibers into the front of the brain, where they converge into tiny hubs called glomeruli inside the olfactory bulb. Each odor type activates a specific pattern of glomeruli, forming a precise sensory map that allows the brain to tell coffee from cinnamon or smoke from perfume. In healthy mice, cells carrying one odor receptor (called M72) and another (called P2) reliably project to just two well-defined glomeruli per bulb, creating a highly ordered and reproducible map.
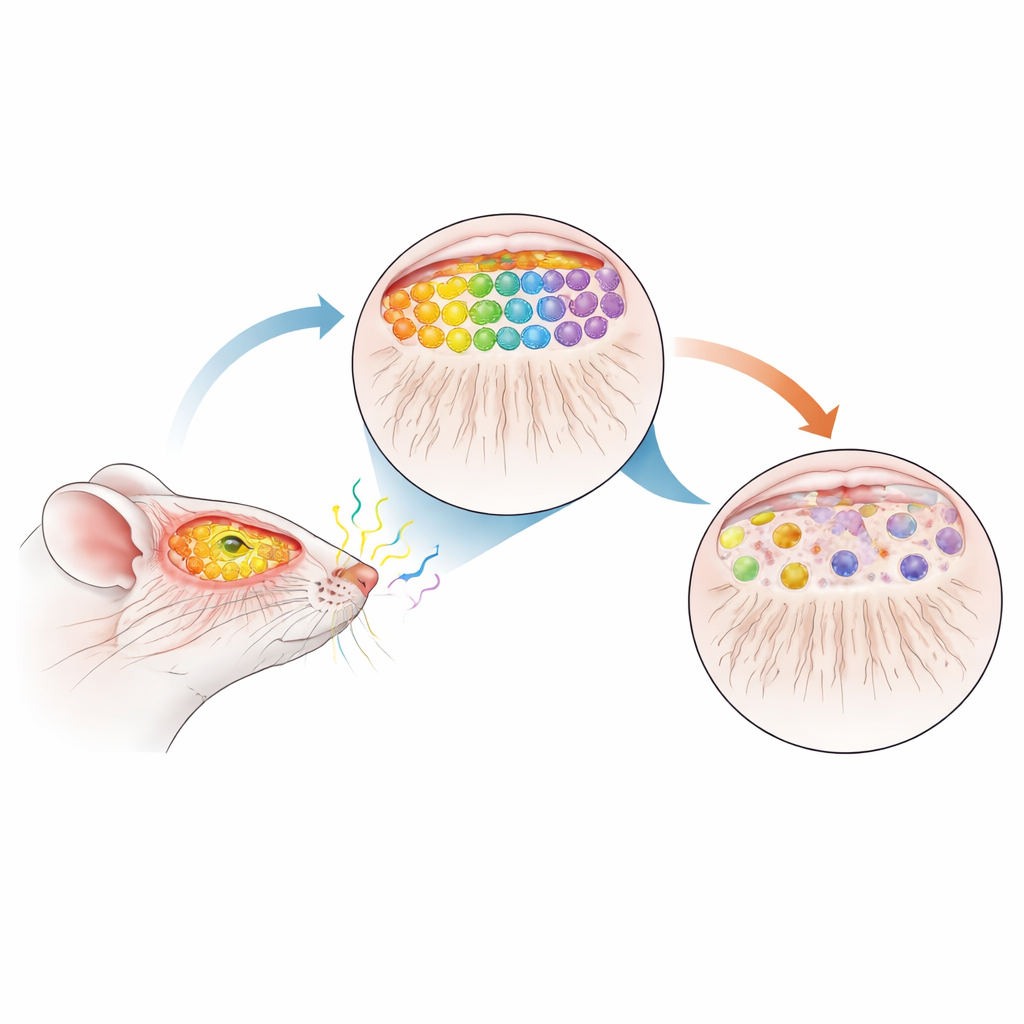
A Mouse Stand-In for Early Parkinson’s
To see how Parkinson’s disease might disturb this map, the team used mice that overproduce normal human α-synuclein, the same protein that clumps in the brains of people with Parkinson’s. They then crossed these animals with M72- and P2-tagged mice so they could visually track selected smell cells and their connections. Importantly, the extra α-synuclein turns on only after birth and gradually accumulates with age, mimicking the slow, progressive course of human disease. At 12 months of age—roughly midlife for a mouse and comparable to a prodromal, or pre-diagnosis, stage in humans—the researchers examined the animals’ olfactory systems in detail.
When the Smell Map Falls Apart
By staining the tagged neurons and following their paths, the scientists found a striking breakdown of the smell map in α-synuclein–overexpressing mice. The number of M72 and P2 neurons in the nose dropped sharply—about three-quarters loss for M72 and nearly half for P2—while their fibers to the bulb were greatly reduced. Instead of converging neatly onto two robust glomeruli, the remaining fibers often formed multiple, smaller glomeruli of varying size and position. These “extra” hubs did not appear in consistent locations from mouse to mouse, signaling a loss of the normal topographic precision. A broad marker of smell-cell terminals, VGLUT2, was also reduced by about 44% in the bulb, indicating that this degeneration extended far beyond the two tagged receptor types.
Linking Broken Circuits to Lost Smell
The structural damage translated into measurable smell problems. Older α-synuclein mice took longer to locate hidden food, showed poorer ability to tell one odor from another, and failed to prefer areas marked with their own scent—behavioral tests that together reveal hyposmia and impaired odor discrimination. They also needed much stronger odor concentrations to respond, revealing reduced sensitivity. These deficits did not appear in younger mice, in which the tagged neurons and their maps still looked largely normal, tying the timing of functional loss to the later breakdown of the sensory map rather than to early development. Notably, while the problematic protein was abundant in the olfactory bulb, it was not detectable in the nose’s sensory lining itself, pointing to changes within the bulb’s circuitry—particularly in mitral cells that relay smell information onward—as the likely drivers of map disruption.
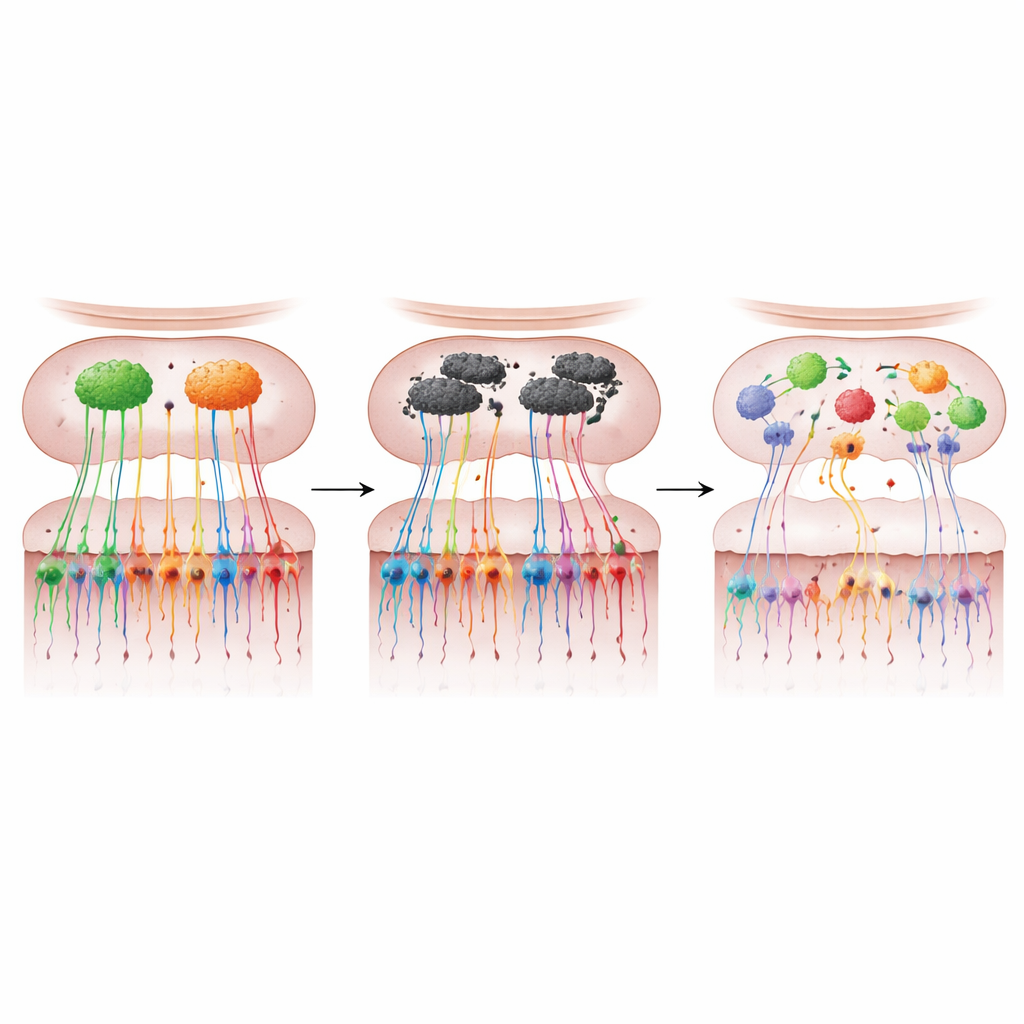
Smell Cells as Windows into Early Disease
Taken together, the work supports a clear message for non-specialists: in this Parkinson’s-like mouse model, overproduction of α-synuclein in the olfactory bulb leads to both loss and miswiring of smell neurons, warping the brain’s odor map and causing early smell problems. Because the nose’s sensory cells are developmentally related to the brain yet can be sampled with minimally invasive swabs or biopsies, the authors argue that similar changes in people could provide a powerful early biomarker of Parkinson’s disease. Mapping the molecular fingerprints of these cells, they suggest, may not only help flag individuals at risk years before movement symptoms emerge, but also offer fresh insight into how the disease starts and spreads—and how it might be slowed or prevented.
Citation: Biju, K.C., Hernandez, E.T., Stallings, A.M. et al. Olfactory sensory map is perturbed in a human wild-type α-synuclein overexpressing transgenic mouse model of Parkinson’s disease. npj Parkinsons Dis. 12, 70 (2026). https://doi.org/10.1038/s41531-026-01288-w
Keywords: Parkinson’s disease, olfactory dysfunction, alpha-synuclein, sensory map, biomarkers