Clear Sky Science · en
LRRK2R1627P mutation amplifies environmental risk factors induced chronic inflammation and α-synuclein aggregation in the gut of rats
Why the Gut Matters in a Brain Disease
Parkinson’s disease is best known for tremors and movement problems, but growing evidence suggests that its roots may begin far from the brain, deep in the gut. This study explores how a specific genetic change linked to Parkinson’s in Asian populations can, together with aging and exposure to toxins, gradually damage the intestine of rats. By watching what happens in the gut over the animals’ lifetimes, the researchers trace how everyday immune defenses can tip into chronic inflammation, setting the stage for disease-related protein buildup that may eventually threaten the brain.
A Risky Gene in the Digestive Tract
The scientists focused on a variant of a gene called LRRK2, long associated with Parkinson’s disease and certain inflammatory bowel conditions. They created rats carrying the analogous mutation, named LRRK2R1627P, and compared them with normal rats throughout life. Although the amount of LRRK2 gene message in the gut was unchanged, the total LRRK2 protein and one of its key activity markers were reduced, indicating that the mutation weakens this protein’s normal function in the intestine. This subtle molecular change did not cause dramatic early damage, but it quietly altered the way intestinal cells renew and organize themselves as the animals grew older.
Aging Gut Under Slow, Persistent Strain
As months passed, rats with the mutation developed clear signs of disturbed gut structure. Their small intestines became shorter, and the fine finger-like villi and pockets (crypts) that absorb nutrients grew smaller. Certain specialized cells in the lining that produce protective mucus and antimicrobial substances—goblet cells and Paneth cells—were reduced, while proteins that help neighboring cells form tight, leak-resistant junctions also fell. Under the microscope, these junctions became shorter or wider, suggesting a weakened barrier. Importantly, the overall architecture did not collapse; instead, the gut showed a more insidious pattern of reduced renewal and protection, which can make it more vulnerable to irritation and infection. 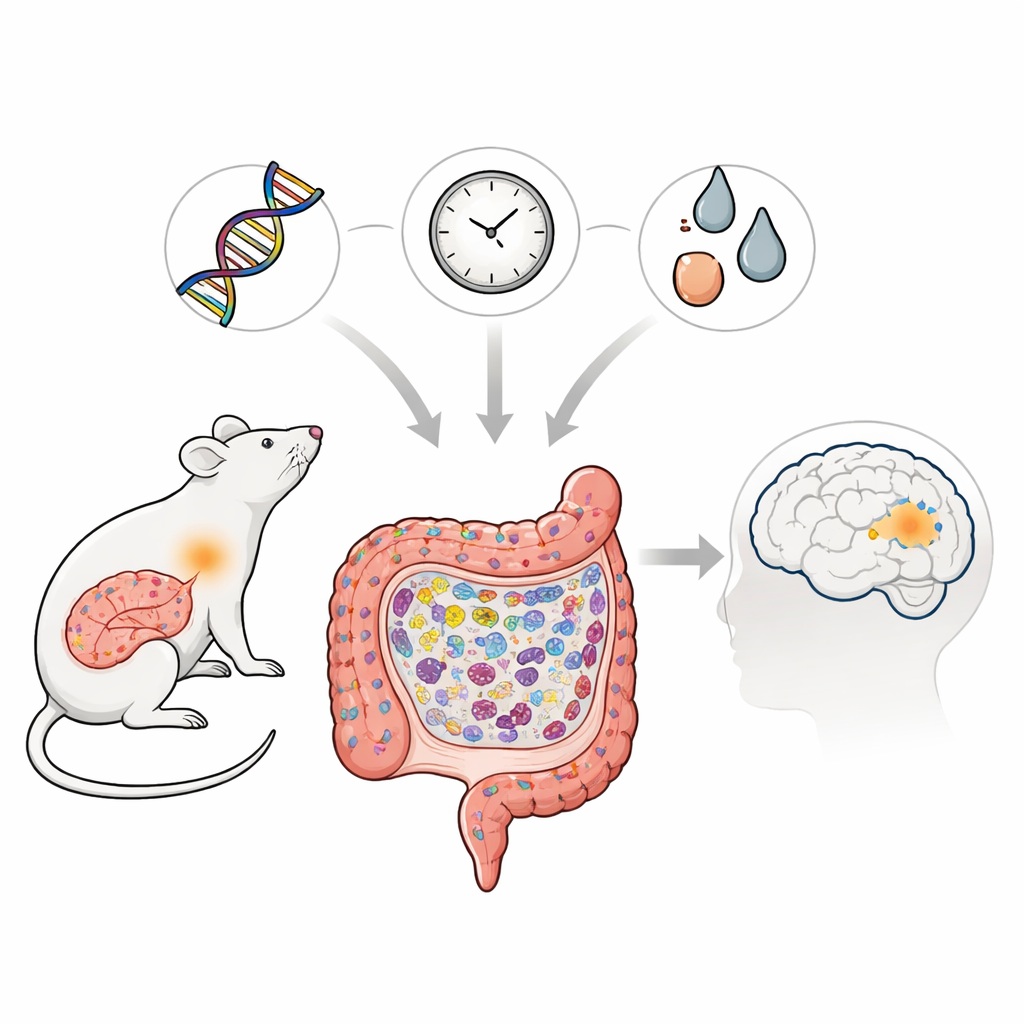
Immune System on a Hair Trigger
To understand why the intestine was drifting out of balance, the team examined gene activity and immune cells in the gut wall. They found that pathways linked to a sensor called TLR4 and its partner NF-κB were overactive in older mutant rats. These sensors normally help the body detect harmful microbes, but here they drove an accumulation of “fight-first” immune cells known as M1 macrophages. These cells pumped out inflammatory molecules, turning the gut into a chronically irritated environment. Strikingly, the Parkinson’s-related protein α-synuclein, in its disease-associated phosphorylated form, began to build up not in gut neurons but inside these activated macrophages in the small intestine, especially in older animals.
Extra Sensitive to Environmental Hits
The genetic mutation alone was not the whole story. When young rats were briefly exposed to bacterial toxins (LPS), which stimulate TLR4, animals with the LRRK2 mutation developed much more severe intestinal inflammation than their normal counterparts. Their gut lining shed more cells, barrier proteins dropped further, and pro-inflammatory macrophages surged, again accumulating abnormal α-synuclein. This suggests that people carrying similar mutations may be especially sensitive to environmental insults that disturb the gut, such as certain infections or toxins, amplifying their long-term risk.
Turning Down the Alarm Signal
Because TLR4 sat at the center of this inflammatory storm, the team tested a drug, TAK-242, that specifically dampens TLR4 signaling. Given over several months to middle-aged rats, the inhibitor largely restored the small intestine’s length, villus and crypt size, mucus-producing cells, and barrier proteins. It reduced overactive macrophages, lowered inflammatory molecules, and sharply cut the buildup of abnormal α-synuclein in the gut. At the same time, it corrected a disturbed microbial community: diversity increased, an overgrowth of Lactobacillus subsided, and several beneficial bacterial groups rebounded, while predicted microbial functions shifted away from disease-associated patterns. 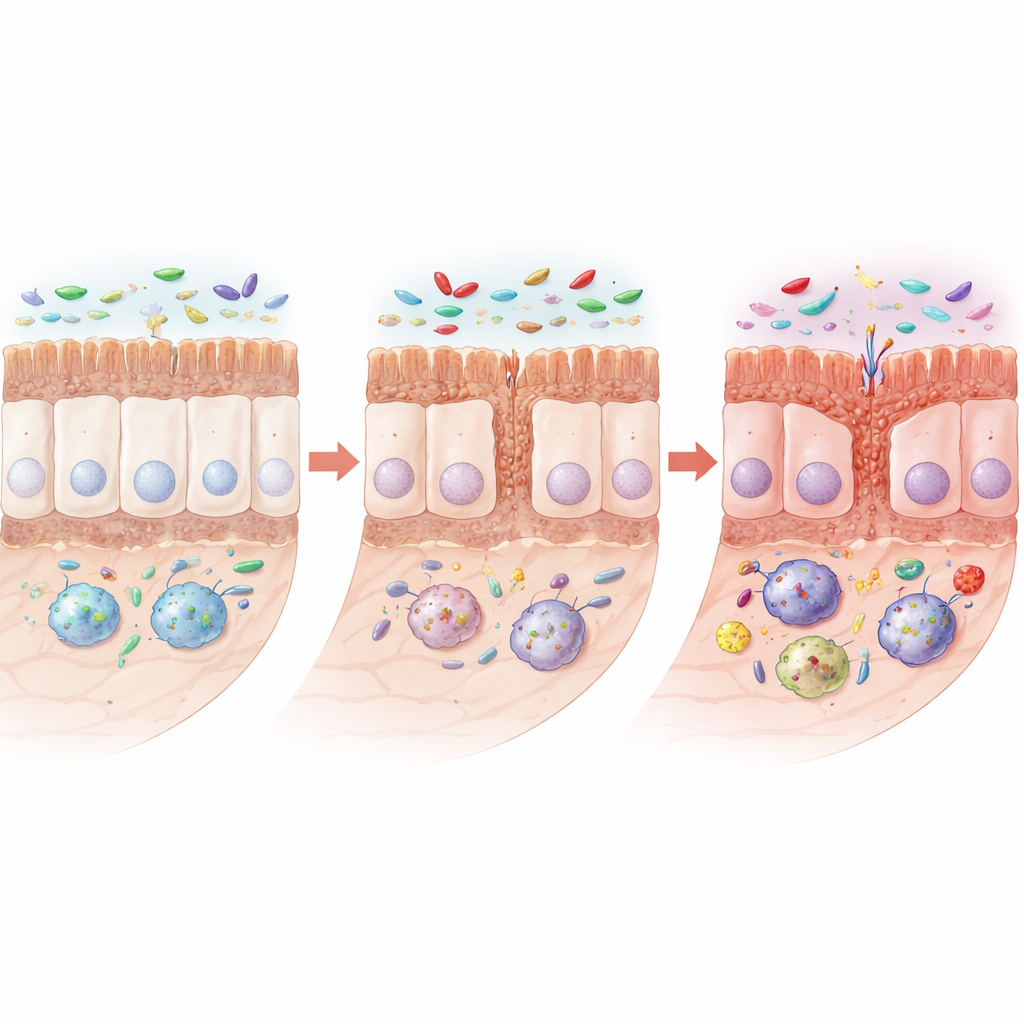
What This Means for Parkinson’s Risk
To a non-specialist, the message is that a “Parkinson’s gene” can quietly reshape the gut’s immune defenses over time, especially in the presence of aging and environmental stress. In these rats, the result is a chronic, low-level inflammation that weakens the intestinal barrier, disturbs resident microbes, and causes Parkinson’s-linked protein to accumulate in immune cells—not yet full-blown Parkinson’s, but a biological setting that could favor later brain disease. By showing that blocking a single gut immune pathway can reverse many of these changes, the study highlights the intestine as a practical early target: protecting gut health and taming gut inflammation may help delay or prevent Parkinson’s in people who carry genetic risks.
Citation: Pang, S., Lu, J., Wang, Y. et al. LRRK2R1627P mutation amplifies environmental risk factors induced chronic inflammation and α-synuclein aggregation in the gut of rats. npj Parkinsons Dis. 12, 68 (2026). https://doi.org/10.1038/s41531-026-01281-3
Keywords: Parkinson’s disease, gut inflammation, LRRK2 mutation, microbiome, innate immunity