Clear Sky Science · en
Deep neurobehavioral phenotyping uncovers neural fingerprints of locomotor deficits in Parkinson’s disease
Why walking troubles in Parkinson’s matter
For many people with Parkinson’s disease, one of the most frightening symptoms is when the feet suddenly feel glued to the floor, or walking slows to a near standstill. These movement breakdowns, from subtle slowness to full "freezing" of gait, greatly increase the risk of falls and loss of independence, yet current drugs and brain stimulation often fail to prevent them. This study set out to uncover the hidden brain patterns that accompany such walking problems, with the long-term goal of designing smarter, on‑demand therapies.
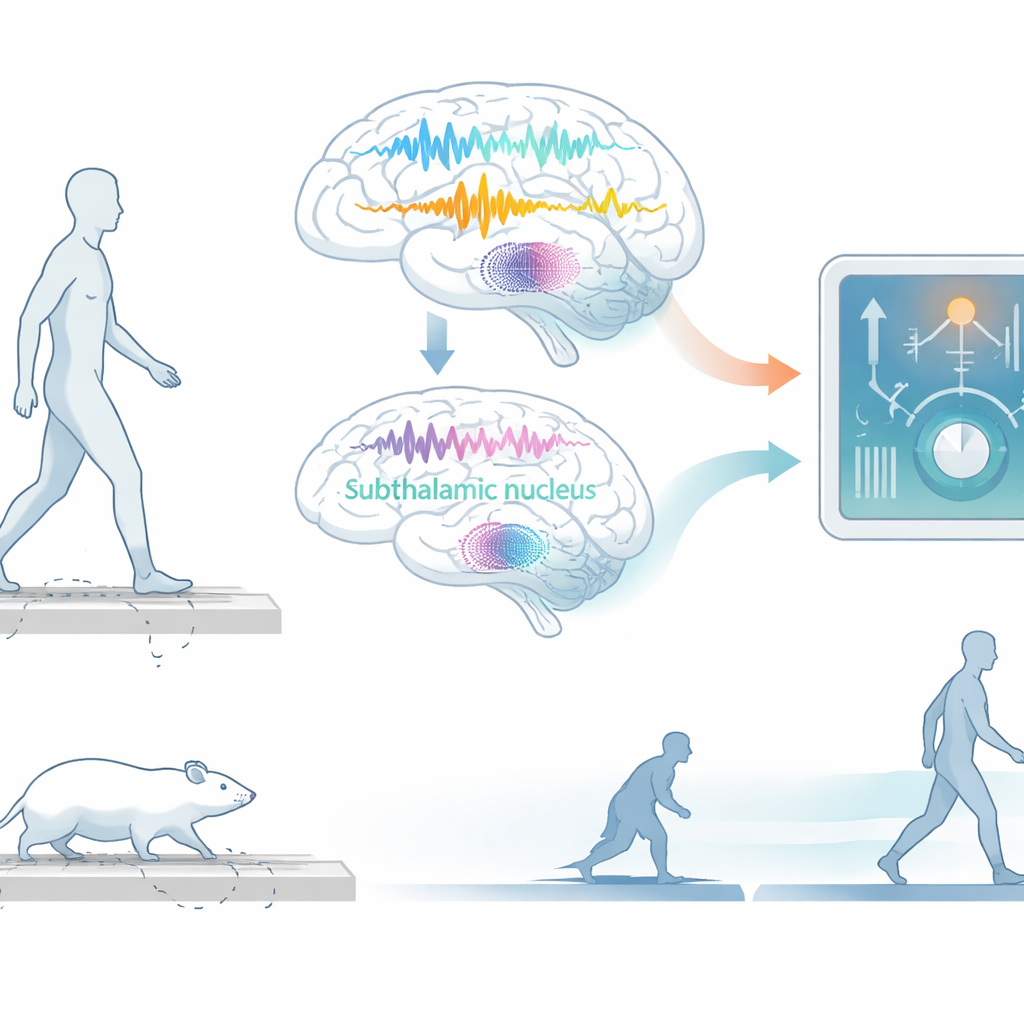
Watching movement in fine detail
The researchers began with a well‑established rat model of Parkinson’s disease. They trained rats to walk back and forth along a runway while tracking every tiny movement of their hind legs in three dimensions and recording electrical activity from the movement areas of the brain. Each moment of behavior was labeled as one of three states: normal walking, small purposeful movements while staying in place, or akinesia—a near‑complete halt in movement that resembles freezing episodes seen in patients. This created a rich “neurobehavioral” dataset that lined up brain rhythms with what the body was doing from instant to instant.
Finding patterns in brain waves
To make sense of this high‑dimensional data, the team applied both classic statistics and modern deep‑learning tools. They first confirmed known signatures: rats with Parkinson‑like damage spent more time in akinesia than healthy animals, and their brain signals showed stronger activity in a frequency range related to abnormal beta and low gamma rhythms. But when the scientists asked which features best distinguished the three movement states, new players emerged. Measures called Hjorth complexity and Hjorth mobility—time‑domain summaries of how irregular and how fast the signal changes—turned out to be powerful markers. In the affected hemisphere, higher complexity and lower mobility were tightly linked to the onset of akinesia, while other features, such as high‑frequency gamma power, tracked active movement.
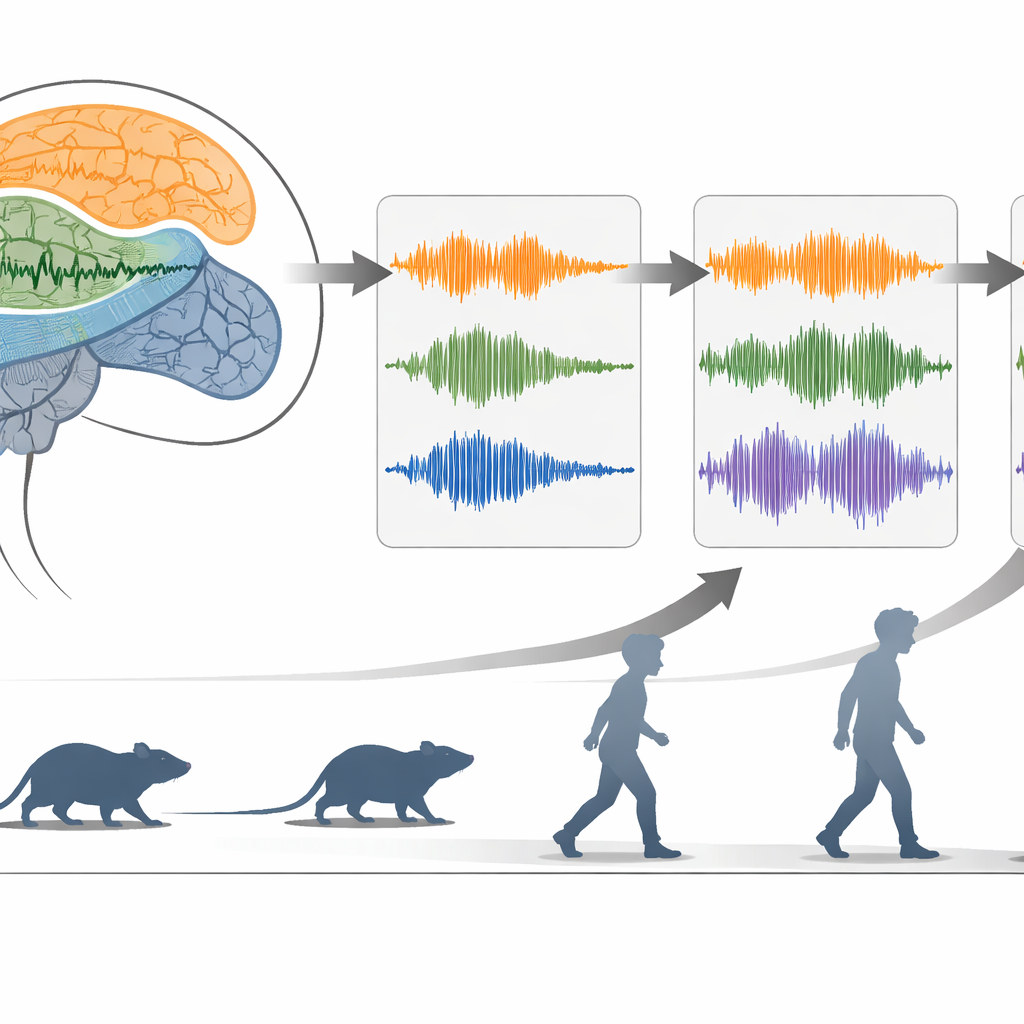
Zooming in on the moment movement stops
Using neural networks that learn compact “maps” of the data, the researchers could place each brief time window into a low‑dimensional space where walking, stationary movement, and akinesia occupied distinct regions. Within this space, the Hjorth measures and the abnormal beta–low gamma band strongly shaped where akinetic episodes landed. When the team focused on the seconds around the start of an akinetic episode, they saw a consistent sequence: complexity and beta‑like power ramped up just before and at the moment of stopping, while mobility and gamma power faded. Crucially, these measures can be computed quickly without heavy frequency analysis, making them attractive candidates for real‑time monitoring.
From rats to people with freezing of gait
The group then asked whether similar neural fingerprints appear in humans. They analyzed detailed motion capture and recordings from electrodes implanted in a deep brain structure, the subthalamic nucleus, in two people with Parkinson’s who experienced freezing of gait. In one participant, the same pattern seen in rats emerged during freezing episodes: increased Hjorth complexity and beta power, alongside decreased Hjorth mobility. Although gamma behaved somewhat differently between species, the core signal that movement was sliding into a pathological state looked strikingly similar in cortex of rats and subthalamic nucleus of this patient, suggesting a shared underlying mechanism across the motor network.
What this could mean for future therapies
By fusing detailed motion tracking with advanced analysis of brain waves, this work identifies simple, computationally light “fingerprints” of when walking in Parkinson’s disease is about to break down. For a layperson, this means that doctors and engineers may soon be able to build brain stimulators that watch for these fingerprints in real time and respond only when needed, nudging the system away from a freezing episode before it fully takes hold. While larger human studies are still required, the approach opens a promising path toward personalized, closed‑loop treatments for disabling gait problems in Parkinson’s disease.
Citation: Garulli, E.L., Merk, T., El Hasbani, G. et al. Deep neurobehavioral phenotyping uncovers neural fingerprints of locomotor deficits in Parkinson’s disease. npj Parkinsons Dis. 12, 65 (2026). https://doi.org/10.1038/s41531-026-01280-4
Keywords: Parkinson’s disease, freezing of gait, brain rhythms, deep brain stimulation, neurobehavioral phenotyping