Clear Sky Science · en
Systematic evaluation of mitochondrial morphology regulators for amelioration of neuronal α-synucleinopathy
Why Tiny Powerhouses Matter in Brain Disease
Mitochondria, the cell’s “power plants,” are crucial for keeping nerve cells alive and their connections working. In brain disorders like Parkinson’s disease, these tiny structures often look damaged or broken apart, but it has been hard to know which changes are harmful and which switches we might safely flip to protect them. This study systematically tests key mitochondrial “shape regulators” in a lab model of Parkinson’s‑related protein buildup, using an artificial‑intelligence tool to measure mitochondrial shapes inside different parts of neurons. The work points to one particular regulator, called Fis1, as a promising target to keep mitochondria — and synapses — healthy without causing new side effects.
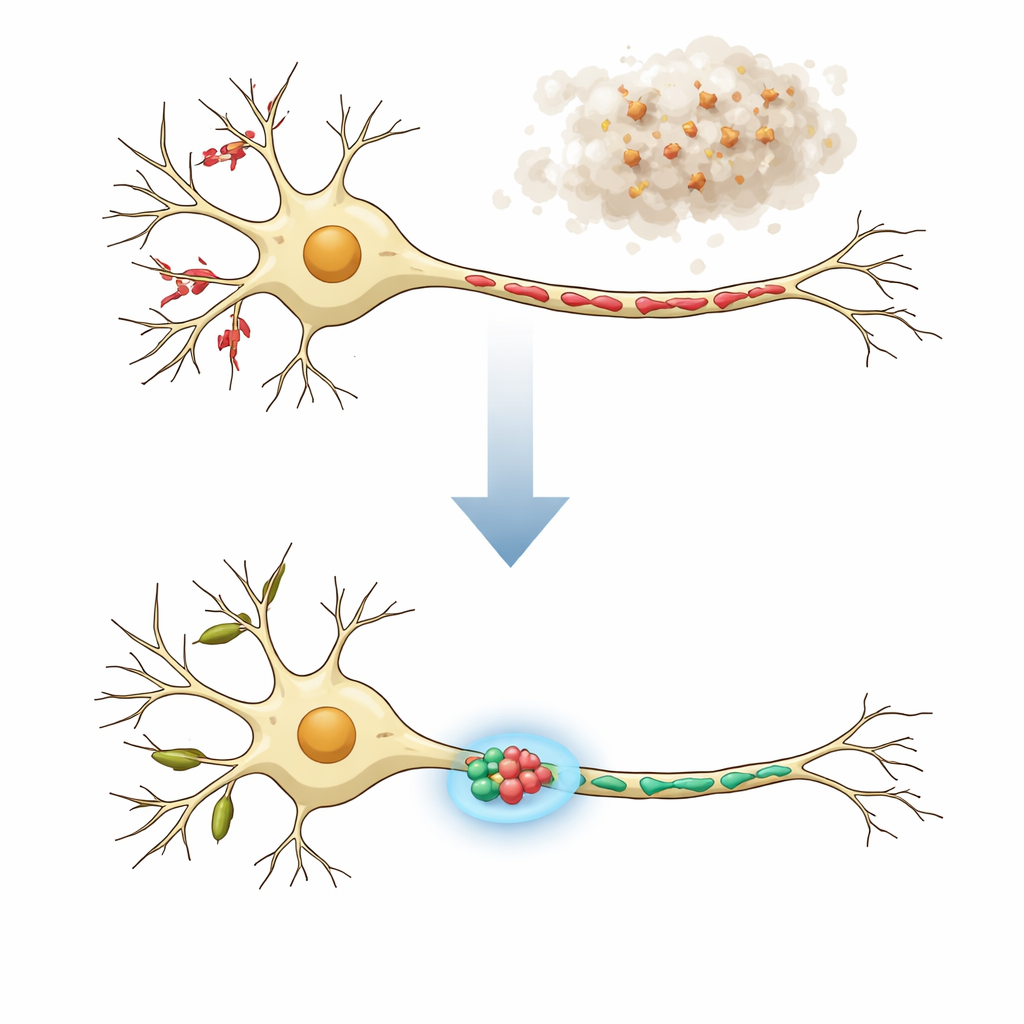
A Brain Cell’s Two Neighborhoods
Neurons are not uniform: their tree‑like branches (dendrites) and long cables (axons) have different jobs and host mitochondria of very different shapes. In healthy nerve cells, dendrites tend to contain long, tube‑like mitochondria that help support local protein production and flexible communication at receiving sites called dendritic spines. Axons, which send signals, instead carry many short mitochondria that supply energy and help control calcium levels at presynaptic terminals. In neurodegenerative diseases, both the function and structure of these mitochondria can be disturbed, showing up as fragments, swollen shapes, or beaded strings. The authors reasoned that truly effective treatments must restore mitochondria in both dendrites and axons back to their normal, compartment‑specific shapes.
Building a Parkinson’s‑Like Stress Test
To mimic a key feature of Parkinson’s disease and related disorders, the researchers exposed mouse cortical neurons in culture to preformed fibrils of alpha‑synuclein, a protein that can clump into harmful aggregates. Within days, these fibrils seeded abnormal alpha‑synuclein buildup in the cells and led to pronounced mitochondrial fragmentation in both dendrites and axons. Using MitoVis, a deep‑learning image analysis system, the team could automatically separate dendrites from axons in microscope images and measure the length, area, and shape of hundreds of mitochondria per image about ten times faster than manual tracing. This high‑throughput approach confirmed that the disease‑like condition shortened mitochondria and made them rounder, dovetailing with reports from animal models and patient tissue.
Testing the Mitochondrial Shape Switches
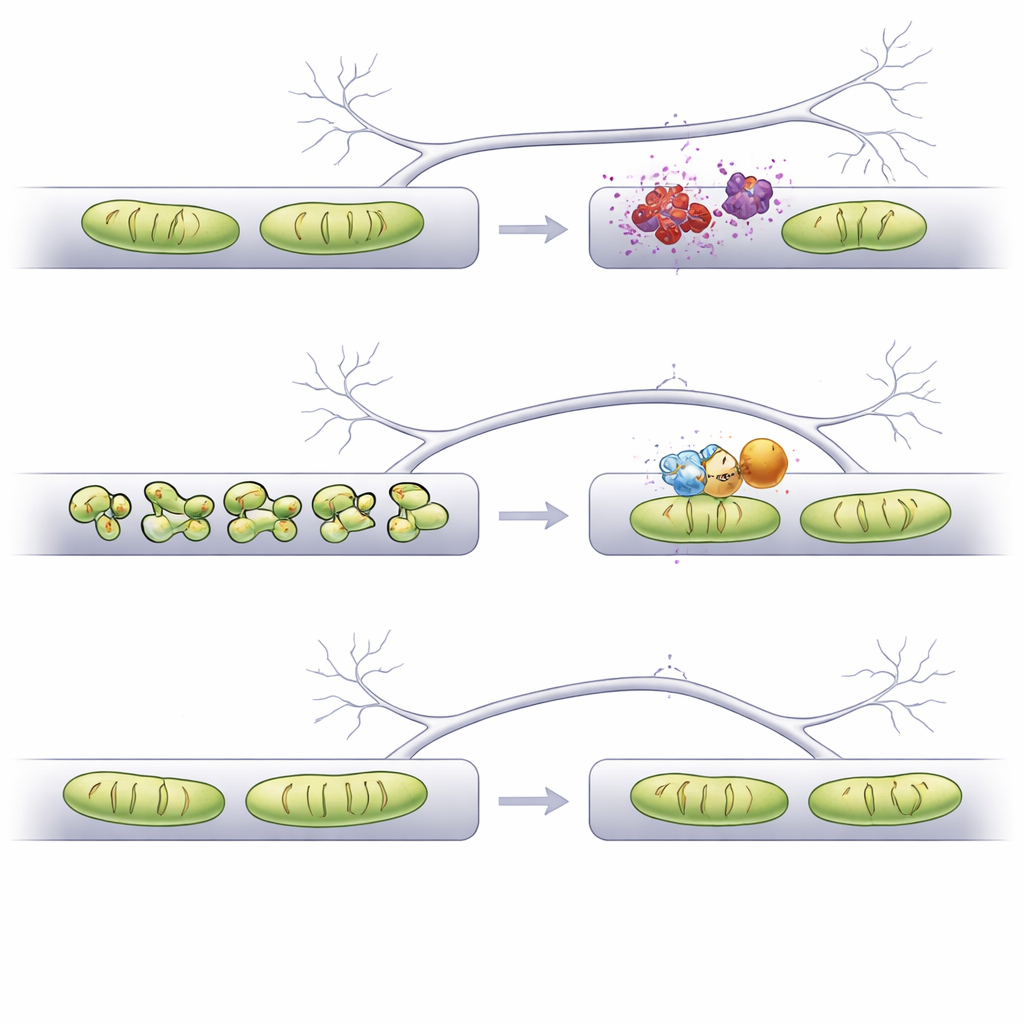
The team then asked whether nudging specific fusion‑and‑fission proteins could prevent this damage. Boosting two fusion proteins (Mfn1 and Mfn2) or knocking down a fission protein (Mff) did protect dendritic mitochondria from alpha‑synuclein‑induced shortening and also kept dendritic spines from being lost. However, these same manipulations made axonal mitochondria overly long, which previous work had linked to problems with signal release and axon branching. In contrast, dialing down a different fission protein, Fis1, restored mitochondrial length in both dendrites and axons to near‑normal values without over‑elongation. Importantly, in this setup Fis1 reduction did not kill neurons, and, like the other interventions, it preserved the density of dendritic spines that would otherwise shrink under alpha‑synuclein stress.
Keeping Calcium in Balance
Because mitochondria in axons help buffer calcium during electrical activity, the researchers tested whether changing their shape altered this delicate balance. They used a fluorescent calcium sensor targeted to mitochondria in presynaptic boutons and stimulated axons with brief bursts of action potentials. In the alpha‑synuclein condition, calcium handling in slightly shortened axonal mitochondria looked similar to normal. But when axonal mitochondria were made excessively long by Mfn1 overexpression or Mff knockdown, they soaked up more calcium than usual after stimulation. This heightened uptake could disrupt presynaptic function. By contrast, Fis1 knockdown, which normalized but did not overshoot mitochondrial length, preserved mitochondrial calcium responses that closely matched healthy controls, suggesting fewer hidden trade‑offs.
What This Could Mean for Future Treatments
Taken together, the study shows that simply making mitochondria longer is not enough — restoring their shape to the right size in the right compartment is key. Using an AI‑assisted imaging workflow, the authors identify Fis1 as an especially attractive target: its downregulation keeps mitochondria structurally stable in both dendrites and axons, prevents the loss of dendritic spines, and avoids abnormal calcium handling in presynaptic terminals. These findings support the idea that carefully tuning mitochondrial shape, potentially with drugs or antisense molecules aimed at Fis1, could help protect vulnerable synapses in Parkinson’s‑related alpha‑synucleinopathy and possibly other brain diseases where mitochondria go awry.
Citation: Kim, S.Y., Choi, J., Jang, D.C. et al. Systematic evaluation of mitochondrial morphology regulators for amelioration of neuronal α-synucleinopathy. npj Parkinsons Dis. 12, 58 (2026). https://doi.org/10.1038/s41531-026-01277-z
Keywords: Parkinson’s disease, mitochondria, alpha-synuclein, synaptic dysfunction, neurodegeneration