Clear Sky Science · en
Clinical utility of evoked potentials for programming subthalamic deep brain stimulation in Parkinsons disease
Why this brain pacemaker story matters
For many people with Parkinson’s disease, medications eventually stop working smoothly. They may swing between stiffness and uncontrolled movements, and fine‑tuning treatment becomes a frustrating cycle of clinic visits. This review article looks at how doctors can use the brain’s own electrical echoes—called evoked potentials—to program deep brain stimulation (DBS) in a faster, more objective way. In essence, it asks whether we can turn DBS from careful guesswork into a more data‑guided, personalized brain pacemaker.
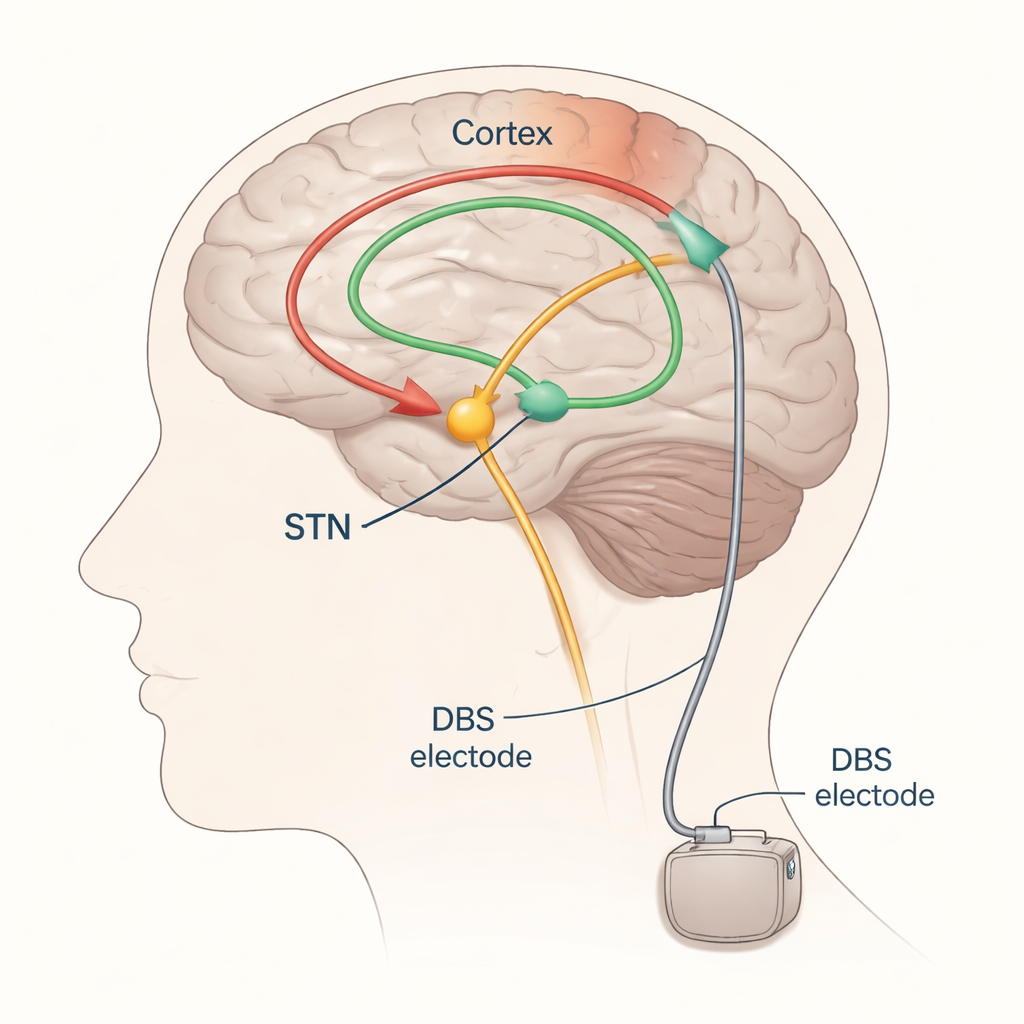
From trial-and-error to guided tuning
DBS for Parkinson’s most often targets a small structure deep in the brain called the subthalamic nucleus (STN). Electrodes are implanted here and connected to a pulse generator under the skin in the chest. Properly tuned, STN‑DBS can ease tremor, slowness, and stiffness, but if the electrical field spreads too far it can accidentally stimulate nearby pathways that control muscles or sensation, causing side effects like muscle pulling or tingling. Today, neurologists usually adjust DBS by slowly changing settings and watching the patient’s movements and side effects—an approach that is time‑consuming, depends on how the patient feels that day, and often requires several follow‑up visits.
Listening to the brain’s electrical echoes
The authors focus on evoked potentials—tiny, time‑locked electrical responses in the brain or muscles after a stimulus—as potential signposts for good and bad DBS settings. When the STN is stimulated, ripples of activity travel along nerve pathways and can be picked up on the scalp with EEG, in muscle activity with EMG, or occasionally with recording strips placed directly on the brain surface. By repeatedly stimulating at low frequencies and averaging the responses, researchers can reliably see distinct waves that appear at different delays (milliseconds) after each pulse. Early waves, arriving within about two thousandths of a second, mainly reflect activation of fast motor pathways running close to the electrode. Slightly later waves, around three milliseconds (often called the “P3” component), are thought to arise from a direct link between the frontal cortex and the STN known as the hyperdirect pathway. Still later waves likely travel through longer, looping circuits that include the thalamus and other deep nuclei.
Finding the sweet spot and avoiding trouble
Across many studies, a consistent picture emerges: contacts on the DBS lead that produce a strong, short‑latency P3 tend to lie in the most helpful part of the STN and are linked to better improvement in Parkinsonian motor symptoms and a wider “therapeutic window” before side effects appear. In contrast, very early waves tied to motor pathways and certain 8–10 millisecond responses often signal that the stimulation is spreading into neighboring structures, increasing the risk of muscle contractions or other unwanted effects. Separate measurements of motor evoked potentials (EMG responses in face and limb muscles) and somatosensory evoked potentials (EEG patterns related to touch pathways) can reveal subtle activation of the internal capsule or the medial lemniscus—fiber bundles that carry motor commands and sensory information. Detecting these changes early allows clinicians to steer current away from risky areas by choosing different contacts or adjusting pulse shape and polarity.
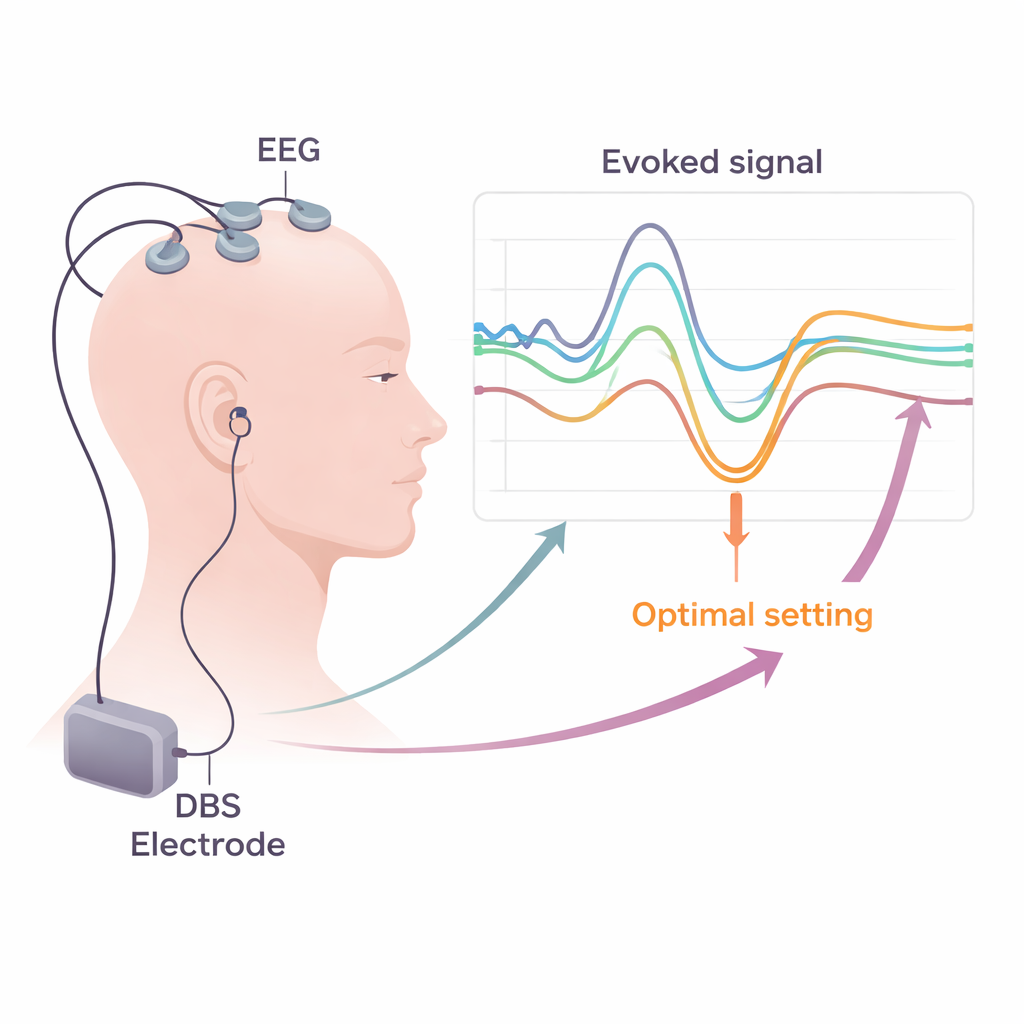
Making the lab tools practical in the clinic
Turning these insights into routine care requires practical solutions. The review explains how standard hospital EEG and EMG systems can already record most of the needed signals, as long as they use high sampling rates and careful methods to remove electrical “noise” from the DBS pulses. The authors discuss strategies for reducing artifacts, such as clever re‑referencing, template subtraction, and advanced computer algorithms that separate true brain activity from stimulation leftovers. They argue that short, structured testing sessions—combining brief periods of low‑frequency DBS, scalp recordings, and muscle monitoring during rest and light contraction—could be folded into the usual post‑surgical programming week. Evoked‑potential maps of each contact could then be combined with imaging and other biomarkers to guide which contacts and settings to use long‑term.
What this means for people with Parkinson’s
The article concludes that evoked potentials are promising candidates to make DBS programming more precise, efficient, and predictable. In particular, the short‑latency P3 wave appears to be a strong marker that stimulation is hitting the right circuit to relieve Parkinson’s motor symptoms, while motor and sensory evoked responses can warn when current is leaking into pathways that cause side effects. Although more work is needed to standardize techniques and prove benefits in everyday practice, this approach aims toward a future where doctors can “listen” to the brain during programming, quickly home in on each patient’s sweet spot, and reduce the trial‑and‑error burden for people living with Parkinson’s disease.
Citation: Hale, B., Latorre, A., Rocchi, L. et al. Clinical utility of evoked potentials for programming subthalamic deep brain stimulation in Parkinsons disease. npj Parkinsons Dis. 12, 54 (2026). https://doi.org/10.1038/s41531-026-01274-2
Keywords: Parkinson’s disease, deep brain stimulation, evoked potentials, brain mapping, neurophysiology