Clear Sky Science · en
Decoding the impact of visual states on adaptive deep brain stimulation feedback signals in movement disorders
Why closing your eyes matters for brain implants
For people living with Parkinson’s disease or dystonia, deep brain stimulation (DBS) can act like a pacemaker for misfiring brain circuits. Newer “adaptive” DBS systems promise to automatically adjust stimulation in real time by listening to brain signals. This study shows that something as simple and normal as closing your eyes can strongly change those signals, meaning that future brain implants must first understand the person’s state before deciding how to stimulate.
Brain pacemakers that learn and adapt
Traditional DBS delivers constant electrical pulses to deep brain areas around the clock. While this can ease symptoms like tremor and stiffness, it may also waste battery power and occasionally cause side effects, because the stimulation does not change when the patient’s condition changes. Adaptive DBS aims to fix this by turning the current up or down based on “local field potentials,” tiny rhythmic voltages recorded from the same electrodes that deliver therapy. These rhythms are strongest in structures called the subthalamic nucleus (STN) and globus pallidus internus (GPi), key hubs in the movement network. 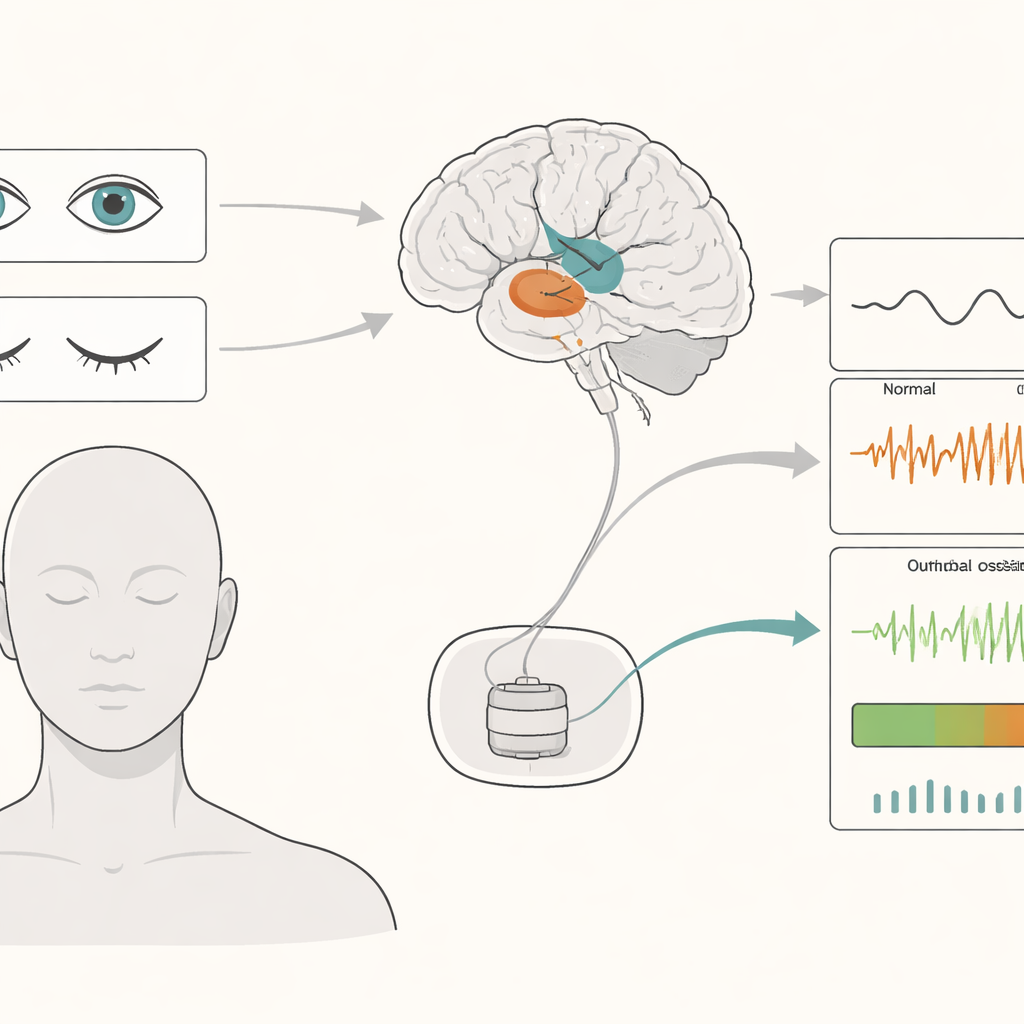
When vision rests, deep brain rhythms shift
The researchers followed 36 patients—18 with Parkinson’s disease and 18 with dystonia—during the days after DBS surgery. They recorded brain activity from STN or GPi electrodes and from the scalp while patients rested with eyes open, eyes closed, or asleep. Closing the eyes consistently boosted slow rhythms in the deep brain, especially in the theta and alpha ranges, which are the very signals many adaptive DBS systems watch. This boost was most pronounced in the STN, which is tightly connected to the outer “thinking” surface of the brain, and less marked in the GPi. Sleep showed a different pattern again, with particularly strong very slow waves.
Different disorders, different signal fingerprints
Parkinson’s disease and dystonia already have distinct rhythmic “fingerprints,” and the study found that eye closure altered these fingerprints in slightly different ways. In both disorders, low-frequency and alpha power dropped when patients opened their eyes again. But in Parkinson’s disease, an extra drop in theta power appeared that did not show up in dystonia. As a result, once the eyes were open, people with dystonia kept more theta activity than those with Parkinson’s. This highlights that the same deep brain signal can reflect both disease and ordinary changes in alertness, making it risky to treat any single rhythm as a simple on–off marker of symptoms.
Brain networks talk more when eyes are shut
Beyond the strength of the rhythms themselves, the team also looked at how tightly deep structures and the cortex move in sync. Using a method that focuses on genuine communication rather than simple shared noise, they found that closing the eyes increased coordinated low-frequency and alpha activity between STN and central scalp regions over the sensorimotor cortex. GPi also showed stronger alpha coupling to the cortex with eyes closed, though more modestly and without clear regional differences. These findings suggest that resting with eyes closed changes not just local activity but the broader communication patterns of the movement network.
Teaching machines to recognize internal states
Because future implants will likely use algorithms to read brain signals, the researchers tested whether simple machine learning models could tell eyes-open from eyes-closed based only on these rhythms. By feeding multiple frequency bands into classifiers, they could identify the eye state with about 88 percent accuracy using STN signals and 77 percent using GPi signals, in both Parkinson’s disease and dystonia. Nonlinear models that can capture more complex patterns performed even better, and the best decoding came from sensorimotor regions most directly linked to movement. 
Toward smarter, context-aware brain stimulation
In plain terms, the study shows that ordinary, healthy changes in how awake or visually engaged a person is can strongly alter the very brain waves that adaptive DBS systems rely on. If an implant simply reacts whenever a rhythm crosses a fixed threshold, it may misread normal state changes as disease flares and over- or under-stimulate. The authors argue that next-generation DBS should first detect the person’s context—such as eyes open versus closed, sleep versus wakefulness—and then interpret disease signals differently depending on that state. Such “state-aware” brain pacemakers could deliver more precise help, avoid unnecessary stimulation, and better support people with movement disorders as they go about their daily lives.
Citation: Zhu, GY., Merk, T., Butenko, K. et al. Decoding the impact of visual states on adaptive deep brain stimulation feedback signals in movement disorders. npj Parkinsons Dis. 12, 61 (2026). https://doi.org/10.1038/s41531-026-01273-3
Keywords: adaptive deep brain stimulation, Parkinson’s disease, dystonia, brain oscillations, brain–computer interfaces