Clear Sky Science · en
Inhibition of de novo ceramide synthesis mitigates alpha-synuclein pathology in a Parkinson’s disease mouse model
Why Fats in the Brain Matter for Parkinson’s
Parkinson’s disease is best known for its shaking hands and slowed movements, but deep inside the brain a quieter drama is unfolding. Nerve cells that control movement slowly die as a protein called alpha‑synuclein clumps into sticky aggregates. This study reveals that a particular family of fats, called ceramides, helps drive that damage—and that blocking their production can protect brain cells in mice and in human cell models. The work points to an unexpected, druggable pathway that might one day slow or prevent Parkinson’s rather than just easing its symptoms.
Clues from Brains After Death
To understand whether ceramides are involved in Parkinson’s and related conditions, the researchers first examined postmortem brain tissue from people who had Lewy body dementia, a disorder that shares the same toxic protein clumps seen in Parkinson’s. Using sensitive chemical measurements, they found that many ceramide types were markedly higher in the midbrain region than in healthy brains, especially forms with long fatty tails. They then re‑analyzed large genetic datasets from human brain cells and discovered that the genes that build and process ceramides were more active in dopamine‑producing neurons from Parkinson’s patients, and also in certain support cells such as astrocytes and oligodendrocytes. Together, these findings suggest that ceramide balance is disturbed across several cell types in the diseased brain.
Cleaning Up Protein Clumps in Cells
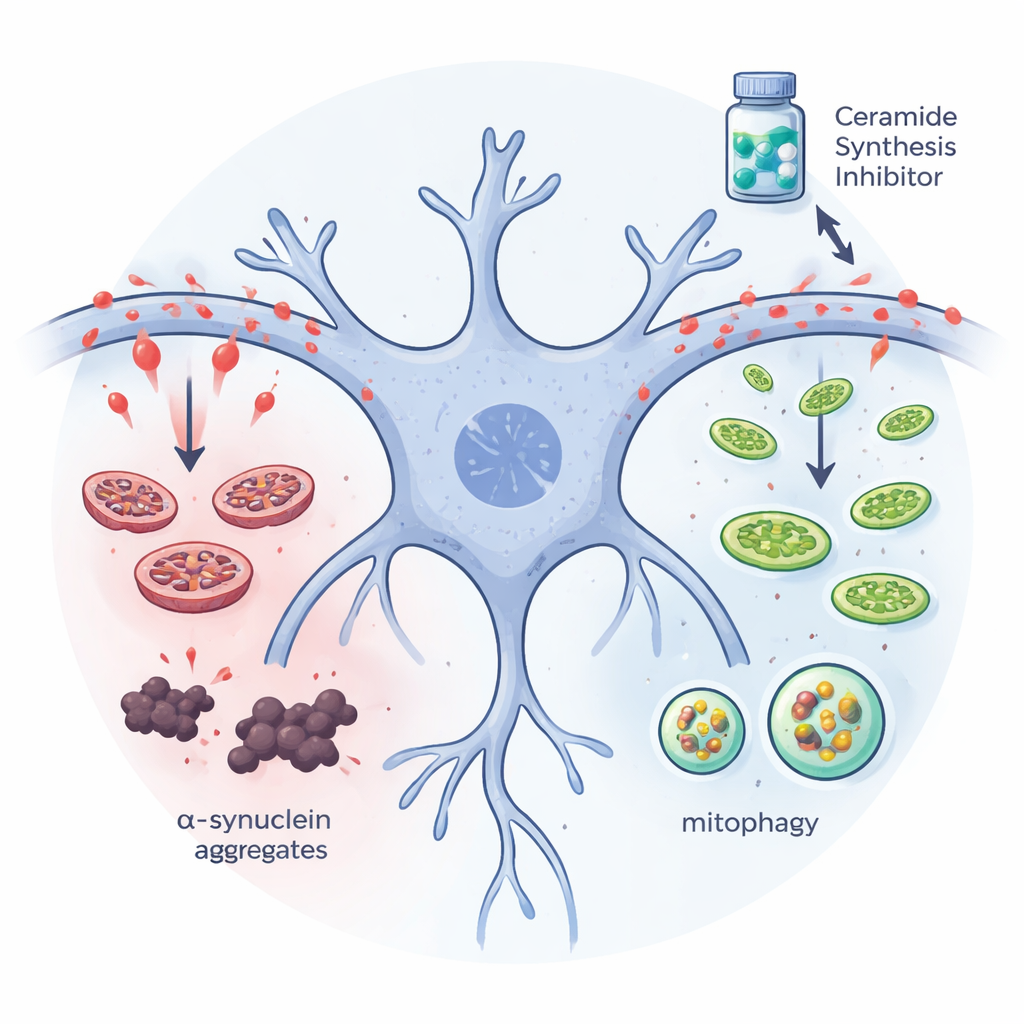
The team next asked whether lowering ceramide levels could reduce protein buildup in nerve‑like cells grown in the lab. They used human neuroblastoma cells engineered to overproduce a mutant form of alpha‑synuclein that readily clumps. When they shut down the first step of ceramide production—either by silencing a key enzyme gene or by adding a drug called myriocin—the amount of insoluble alpha‑synuclein aggregates dropped sharply. At the same time, markers of mitophagy, the cell’s specialized system for finding and recycling damaged mitochondria, went up, and the machinery that tags unwanted proteins for disposal became more active. These shifts suggest that excess ceramide normally blocks the cell’s cleanup crews, and that removing this lipid burden allows the cell to clear faulty mitochondria and protein clumps more efficiently.
Protecting Brain Function in Mice
The most critical test was whether this strategy helps in a living brain. The researchers treated a well‑established mouse model that overproduces mutant human alpha‑synuclein and gradually develops movement problems and memory issues. Starting in mid‑life, some mice received myriocin injections for several months, while others got a harmless vehicle. Myriocin clearly lowered ceramide levels in the animals’ blood and midbrain. Behavior tests showed that treated mice walked farther and performed better on a simple maze that depends on spatial working memory. Brain slices from these animals revealed that more dopamine‑producing neurons survived in key regions, and that the amount of phosphorylated, aggregation‑prone alpha‑synuclein was reduced. Large‑scale gene‑expression profiling of the midbrain further showed that myriocin dialed down inflammatory pathways while restoring genes tied to synaptic communication and healthy mitochondrial maintenance.
Human Neurons and Mini‑Brains Agree
To bring the results closer to patients, the team used neurons and three‑dimensional midbrain "organoids" grown from induced pluripotent stem cells derived from people with Parkinson’s. In patient‑derived neurons carrying a fluorescent sensor for mitophagy, myriocin treatment boosted the signal that marks damaged mitochondria being delivered to cellular recycling centers and improved the architecture of the mitochondrial network. In midbrain organoids, myriocin preserved dopamine‑producing cells and reduced harmful alpha‑synuclein aggregates. When the researchers added extra ceramides to these mini‑brains, the opposite happened: more protein clumps formed and dopamine neurons were lost, especially in organoids from Parkinson’s donors. These experiments support a direct, damaging role for ceramide buildup in human‑relevant tissue.
What This Could Mean for Future Treatments
For non‑specialists, the core message is straightforward: in multiple Parkinson’s models, too much of a certain brain fat appears to poison cells by promoting protein clumps, faulty mitochondria, and chronic inflammation. Blocking the main pathway that makes these fats, using the experimental drug myriocin, eased these problems, preserved vulnerable neurons, and improved behavior in mice, while also rescuing human neurons in a dish. The authors stress that long‑term safety and dosing in people are unknown, and Parkinson’s has many causes beyond ceramides. Still, the work opens a fresh line of attack: instead of only boosting the fading dopamine signal, future therapies might also normalize lipid metabolism and cellular cleanup, giving brain cells a better chance to resist the slow march of neurodegeneration.
Citation: Lee, E., Park, My., Park, M. et al. Inhibition of de novo ceramide synthesis mitigates alpha-synuclein pathology in a Parkinson’s disease mouse model. npj Parkinsons Dis. 12, 49 (2026). https://doi.org/10.1038/s41531-026-01263-5
Keywords: Parkinson’s disease, ceramide, alpha-synuclein, mitophagy, neurodegeneration