Clear Sky Science · en
UQCRC1 deficiency impairs mitophagy via PINK1-dependent mechanisms in Parkinson’s disease
Why this research matters to everyday people
Parkinson’s disease is best known for its movement problems—tremor, stiffness, and slowness—but underneath those symptoms lies a struggle inside brain cells to make and clean up energy. This study explores how a little-known protein, UQCRC1, helps brain cells keep their “power plants” healthy, and how its failure may open the door to Parkinson’s. Understanding this hidden machinery could point the way to treatments that slow or even prevent the disease, rather than just easing symptoms.
A power problem inside brain cells
Our brain cells rely on mitochondria, tiny structures often described as cellular power plants, to produce the energy they need. UQCRC1 is an essential part of one of the mitochondrial engines that carry out this job. Earlier work showed that rare inherited mutations in UQCRC1 can cause Parkinson’s-like illness by stressing these engines. In this study, the authors asked a broader question: is reduced UQCRC1 also involved in the much more common, non-inherited (“idiopathic”) forms of Parkinson’s disease?
Finding a common signal in patient brain data
To answer this, the researchers pooled 19 publicly available datasets of brain tissue from people who had died with and without Parkinson’s disease. They focused on the substantia nigra, the deep brain region whose dopamine-producing nerve cells degenerate in Parkinson’s. Across 150 control cases and 185 Parkinson’s or related cases, they consistently found lower levels of UQCRC1 gene activity in Parkinson’s brains—about a 20% reduction on average, with little sign that the result was due to chance or publication bias. They confirmed this by measuring UQCRC1 protein in brain samples from two independent sources and in human nerve-like cells carrying a disease-linked UQCRC1 mutation; in each case, UQCRC1 levels were clearly lower in the Parkinson’s condition. 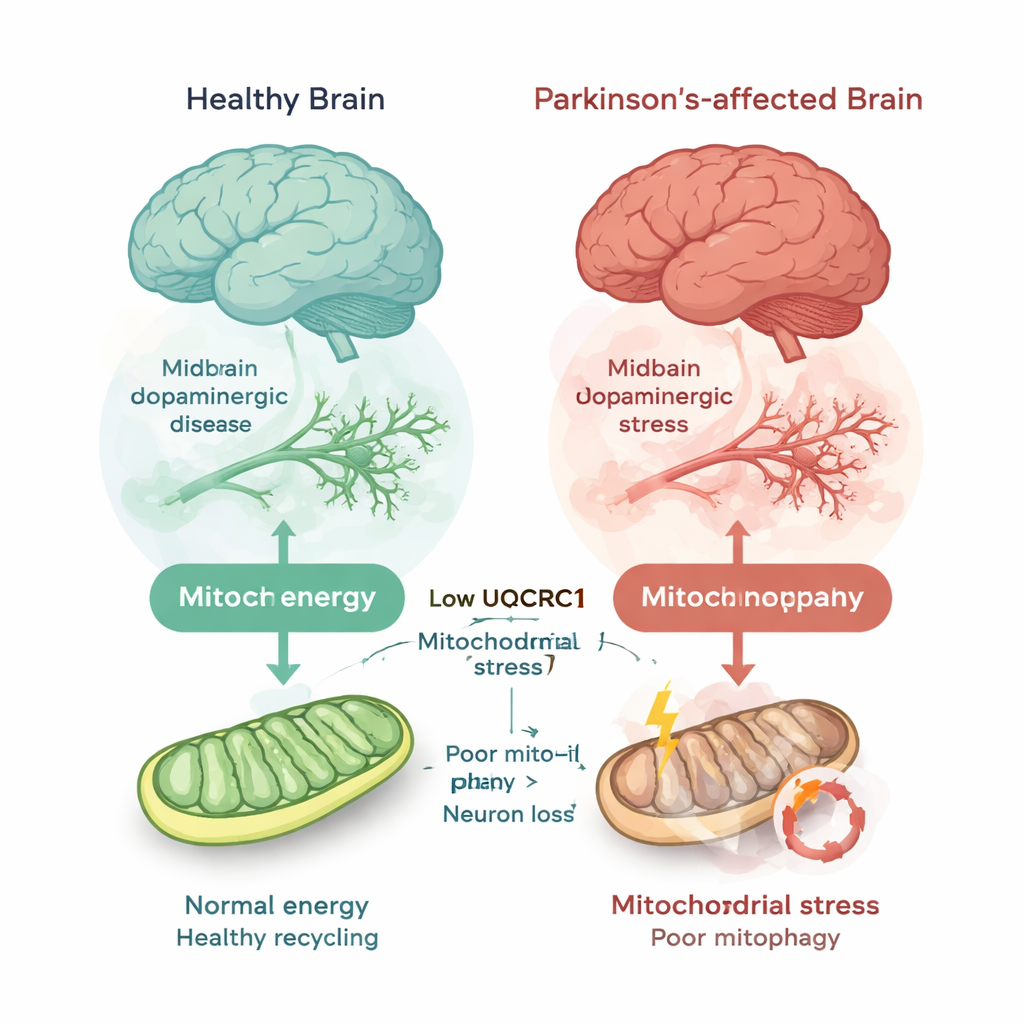
When cleanup crews falter
Mitochondria do not just make energy; they also need regular inspection and removal when damaged. Cells use a quality-control process called mitophagy to tag worn-out mitochondria and send them to a recycling compartment. The team used fluorescent reporters in human cells and fruit flies to watch this process in action. Under stress that normally triggers mitophagy, cells with UQCRC1 mutations or reduced UQCRC1 made far fewer “mitolysosomes,” the structures that mark successful disposal of damaged mitochondria. In fruit fly dopamine neurons, reducing UQCRC1 likewise led to fewer recycled mitochondria, mirroring defects seen when a core autophagy gene was blocked. These findings show that UQCRC1 loss does not simply weaken energy production—it also disrupts the cleanup of faulty power plants.
A missing switch and a promising target
The researchers then turned to PINK1, a protein that acts as a sensor and switch for mitophagy. When mitochondria are stressed, PINK1 accumulates on their surface, recruits another protein called Parkin, and kick-starts the tagging process that leads to recycling. Meta-analysis of patient brain data revealed that PINK1, but not Parkin, was also reduced in Parkinson’s midbrain, by roughly 22%. In both human cells and flies with UQCRC1 problems, PINK1 levels fell, and early steps of the PINK1–Parkin pathway—Parkin moving onto mitochondria and tagging them—were blunted. Strikingly, boosting PINK1 in flies restored their climbing ability and normalized mitophagy, suggesting that dialing this switch back up can compensate for UQCRC1 loss. 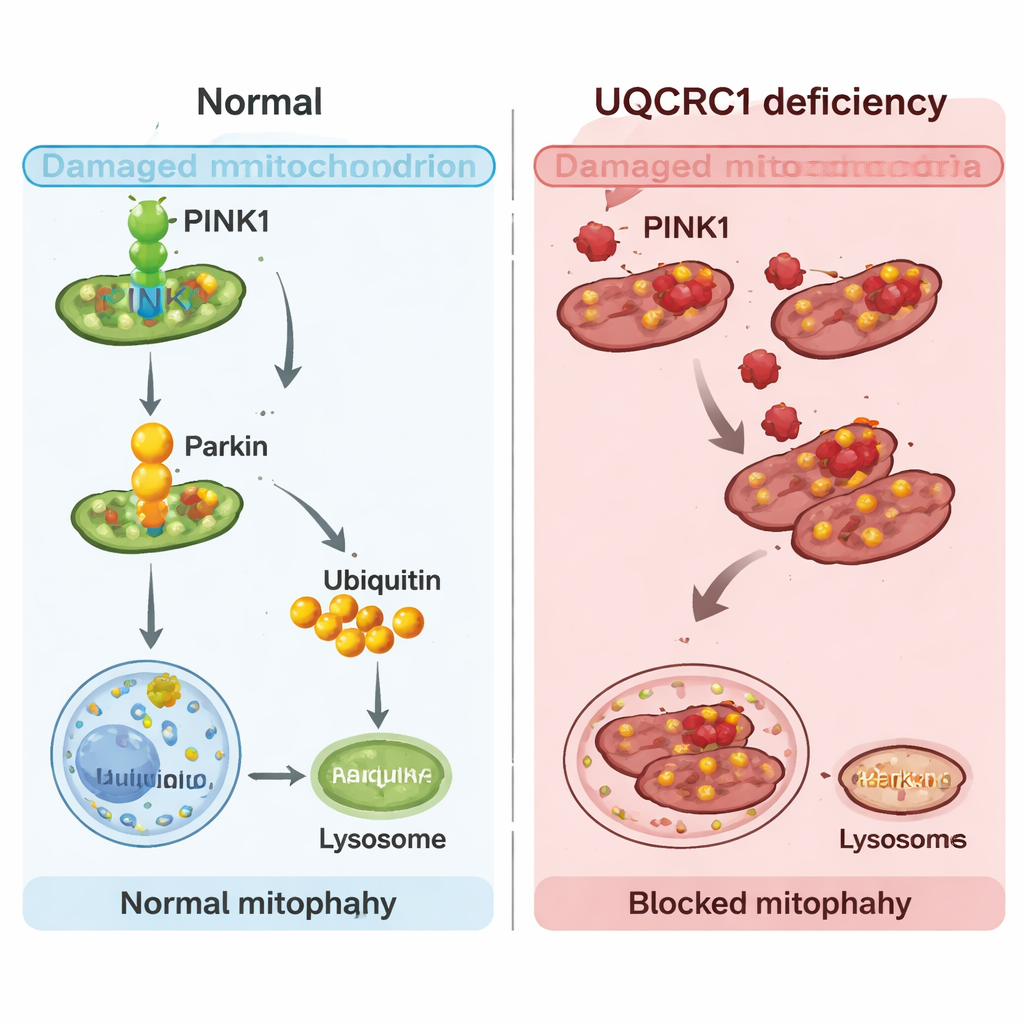
Testing drug-like activators of the cleanup pathway
Because directly adding PINK1 is not practical in patients, the team tested small molecules known to enhance PINK1 activity: kinetin and MTK458. In fruit flies with UQCRC1 deficiency, feeding these compounds improved movement and protected vulnerable dopamine neurons. In human nerve-like cells with reduced UQCRC1, MTK458 lengthened shrunken nerve projections and revived defective mitophagy. These benefits depended on PINK1, supporting the idea that carefully activating this pathway can help cells cope with mitochondrial stress caused by low UQCRC1.
What this could mean for future Parkinson’s treatments
Altogether, the study links a drop in UQCRC1 to a chain reaction: stressed mitochondria, weaker PINK1-driven cleanup, and ultimately loss of dopamine neurons. For a layperson, this means that some cases of Parkinson’s may stem from power plants that not only run poorly but also pile up as trash the cell cannot clear. While more work is needed to map every step and test safety in people, the results highlight PINK1 activators as potential precision medicines for patients whose Parkinson’s is marked by mitochondrial defects like UQCRC1 deficiency. Instead of just masking symptoms, such treatments would aim to restore the cell’s own ability to maintain and renew its energy factories.
Citation: Li, JL., Huang, SY., Huang, PY. et al. UQCRC1 deficiency impairs mitophagy via PINK1-dependent mechanisms in Parkinson’s disease. npj Parkinsons Dis. 12, 48 (2026). https://doi.org/10.1038/s41531-026-01262-6
Keywords: Parkinson’s disease, mitochondria, mitophagy, PINK1, UQCRC1