Clear Sky Science · en
Early retinal synaptic dysfunction and proteomic remodeling precede neurodegeneration in a Parkinson’s disease model
Why your eye could reveal early signs of Parkinson’s
Parkinson’s disease is best known for causing tremors and movement problems, but long before these symptoms fully appear, subtle changes may be unfolding in the eye. Because the retina is essentially an outpost of the brain that we can look at non‑invasively, scientists are asking whether it might act as a window into early brain disease. This study in a Parkinson’s mouse model shows that the retina’s wiring and protein makeup begin to shift well before obvious nerve cell loss, raising the possibility that simple eye tests could one day help detect Parkinson’s earlier and track how it progresses.
Looking to the eye for early warning signals
The researchers worked with mice engineered to overproduce a mutant form of alpha‑synuclein, the key protein that clumps in the brains of people with Parkinson’s disease. They examined the animals at an early adult stage (six months) and a later stage (sixteen months), combining eye imaging, electrical tests of retinal function, and large‑scale protein measurements. Their central question was straightforward: do measurable retinal changes appear before large numbers of retinal nerve cells die, and if so, what molecular shifts accompany those changes?
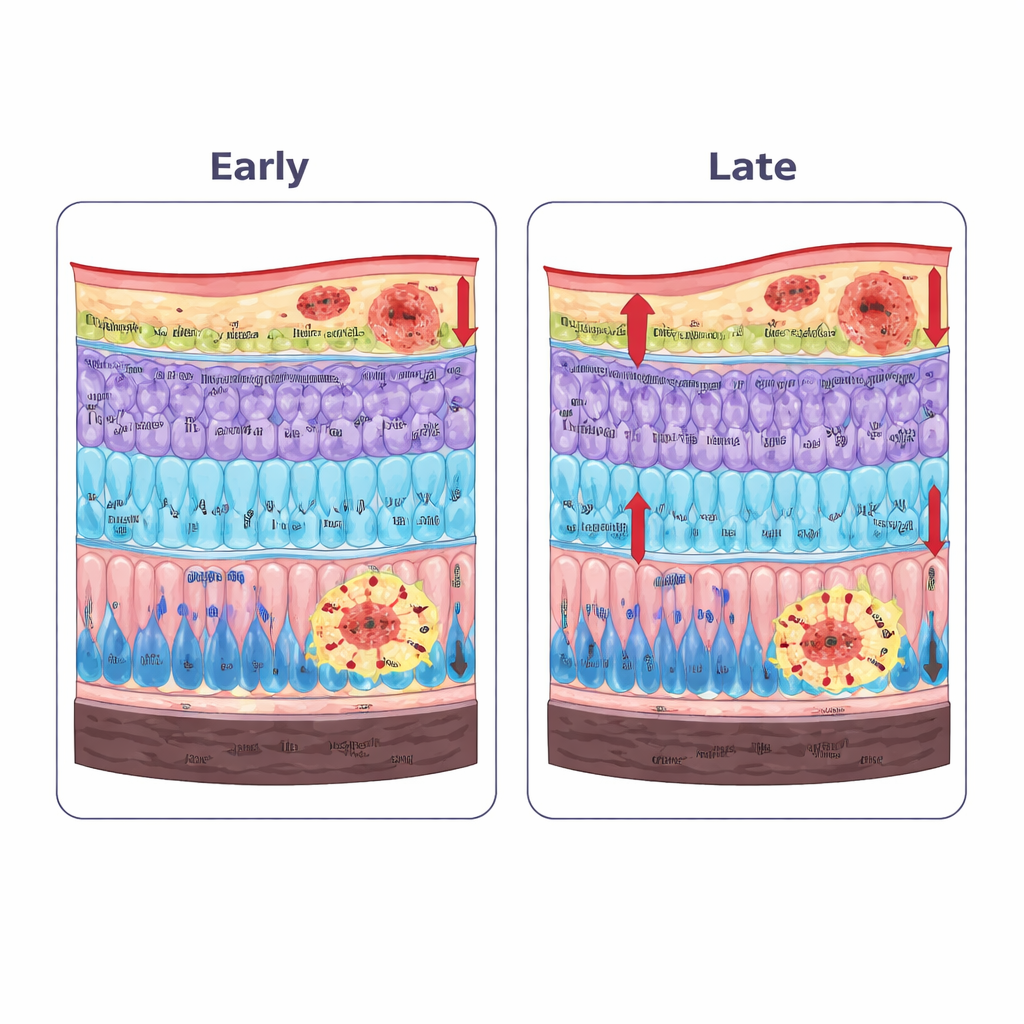
Subtle structural and electrical changes in the retina
High‑resolution eye scans, similar in principle to those already used in eye clinics, revealed that specific retinal layers changed thickness in the Parkinson’s mice. The layer containing nerve fibers and ganglion cells (which send visual signals to the brain) and the light‑sensing photoreceptor layer were already thinner by six months and became thinner still with age. In contrast, the inner plexiform layer—where many retinal neurons exchange signals—became thicker, a pattern consistent with swelling and remodeling driven by inflammation rather than simple cell loss. Electrical recordings from the retina showed that, early on, fine ripples in the signal linked to local “interneuron” circuits were reduced even when the main waves looked nearly normal. By sixteen months, both rod‑ and cone‑driven responses were clearly impaired, and activity related to the output ganglion cells had fallen, matching the earlier structural shifts.
Early protein buildup and stressed support cells
Microscopic staining of retinal tissue showed that the mutant alpha‑synuclein did not stay confined to the brain. Its phosphorylated, aggregation‑prone form accumulated in the outer plexiform layer, where photoreceptors connect to downstream cells. At the same time, support cells in the retina became reactive. An early increase in a structural protein called GFAP was seen mainly in astrocytes near the inner surface of the retina, and by the later stage, tall GFAP‑positive processes from Müller cells spanned multiple layers—hallmarks of chronic stress and inflammation. While the numbers and basic shapes of key interneurons appeared largely preserved, the specialized “ribbon” structures at photoreceptor synapses, marked by the protein CtBP2, were already reduced at six months and declined further with age, pointing to early breakdown in the communication points between cells.
A shifting molecular landscape inside the eye
To understand these changes at a systems level, the team cataloged more than 4,000 retinal proteins and compared Parkinson’s and normal mice at both ages. The overall protein profiles separated cleanly by disease status, indicating that Parkinson’s‑related processes shaped the retinal proteome more strongly than normal aging. Alpha‑synuclein itself was higher at both stages, confirming the tissue staining. Proteins involved in handling oxidative stress, such as certain calcium‑binding and metal‑binding molecules, were consistently elevated, suggesting a persistent reaction to damaging reactive molecules. Other proteins linked to the cell’s internal scaffolding and to protective chaperone functions—especially members of the crystallin family—showed stage‑dependent shifts: some were boosted early and then dropped, while others rose later, implying an initial attempt at compensation that gives way to broader remodeling as disease advances. Network analyses highlighted crystallins and cytoskeletal components as central hubs in this evolving protein interaction map.
What this means for people living with Parkinson’s
Taken together, the findings support a simple idea for non‑experts: in this Parkinson’s model, the retina starts to malfunction and rewire itself well before large numbers of nerve cells die. Protein clumps, inflammation, and stress responses first disrupt the synapses—the tiny contact points between cells—especially where light‑sensing photoreceptors hand off signals. These early changes alter how the retina processes visual information and eventually lead to measurable vision problems. Because similar eye imaging and electrical tests are already used in clinics, and because the retina can be sampled in a relatively accessible way, this work strengthens the case for developing retinal measurements as early warning signs and progression markers for Parkinson’s disease, with the long‑term goal of enabling treatment before irreversible damage occurs.
Citation: Moon, CE., Lee, S.J., Shin, H. et al. Early retinal synaptic dysfunction and proteomic remodeling precede neurodegeneration in a Parkinson’s disease model. npj Parkinsons Dis. 12, 47 (2026). https://doi.org/10.1038/s41531-026-01261-7
Keywords: Parkinson’s disease, retina, alpha-synuclein, neurodegeneration, biomarkers