Clear Sky Science · en
The graded effect of propofol in electrophysiology-guided navigation during deep brain stimulation surgery
Why this matters for people with Parkinson’s
Deep brain stimulation (DBS) has transformed treatment for many people with Parkinson’s disease by easing tremors and stiffness when medications no longer suffice. But the benefits of DBS depend critically on placing thin electrodes within a tiny target deep in the brain. More and more hospitals now perform this surgery under full anesthesia with the drug propofol, sparing patients the stress of being awake. The question this study tackles is simple but crucial: does propofol make it harder for surgeons to navigate precisely to the right spot in the brain, and if so, how can they safely work around that?
Finding a moving target deep in the brain
To help guide DBS surgery, doctors use not only detailed brain scans but also real-time electrical recordings from individual brain cells. As a hair-thin recording wire moves down toward the subthalamic nucleus, a key target in Parkinson’s, the pattern and intensity of nerve-cell firing change in characteristic ways. These changes act like landmarks on a road map, telling the surgical team when they have entered or left the desired structure. The study analyzed 702 such recordings from 25 people with Parkinson’s, some operated on while awake with local numbing medicine and others under general anesthesia with propofol at different doses. The researchers then mapped every recording into a standard brain space to compare how these electrical landmarks shifted with different anesthesia levels.
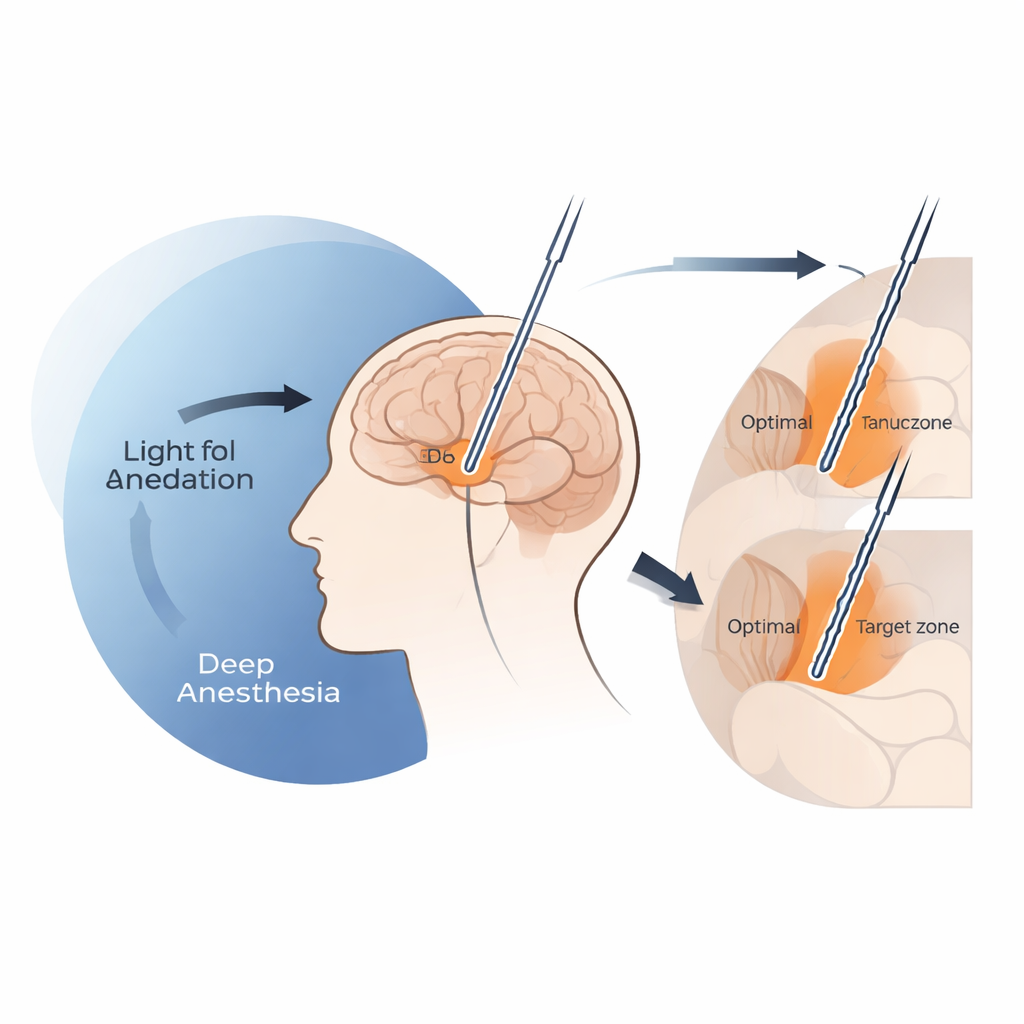
When too much sedation hides the brain’s signposts
The team found that deep propofol sedation can effectively dim the very signals surgeons rely on to know when they have reached the upper border of the subthalamic nucleus. When infusion rates of propofol rose above about 4 milligrams per kilogram per hour, the general background electrical activity near the entry to this structure dropped sharply, and clearly identifiable single-cell signals tended to appear only deeper along the track. Under these high-dose conditions, final DBS electrodes ended up systematically lower than the spot previously associated with the best movement improvement, a location often referred to as the clinical “sweet spot.” In contrast, when propofol doses stayed at or below that 4 mg/kg/h threshold, the spatial pattern of recordings closely resembled what was seen in awake patients, and electrode placement accuracy matched that of local anesthesia surgeries.
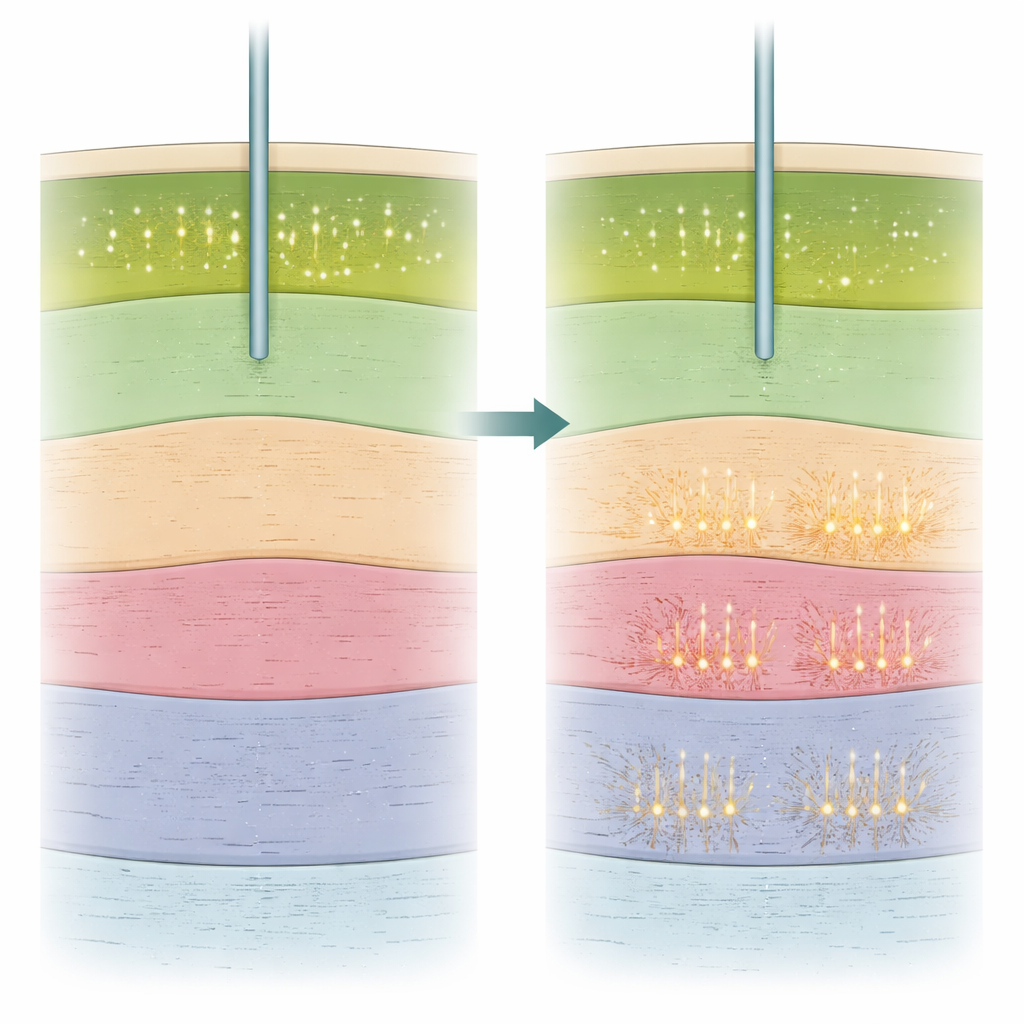
A new signal that still works under anesthesia
Beyond whether cells were firing, the researchers examined how they were firing. They calculated several simple features of each neuron’s activity, including how fast it fired and how much it fired in brief bursts versus more evenly over time. One measure, called the burst index, turned out to be especially informative. This index was higher in the subthalamic nucleus than in the neighboring substantia nigra region, allowing the team to distinguish the two even under general anesthesia. Importantly, the closer a recording site lay to the clinical sweet spot, the higher its burst index tended to be, both in awake and asleep surgeries. Other measures, such as average firing rate or the regularity of the spikes, were less reliable at telling the two structures apart. The team also used electroencephalography (EEG) from the scalp to gauge how deeply sedated each patient was and showed that deeper sedation went hand-in-hand with more blurred borders at the top of the target and more bursty firing overall.
Practical tips for safer “asleep” brain surgery
Because this analysis came from routine clinical practice rather than a randomized trial, it cannot dictate one “correct” anesthesia recipe. Still, the patterns it reveals offer practical guidance. Keeping propofol infusion at or below roughly 4 mg/kg/h during the recording phase appears to preserve the key electrical cues needed for navigation, allowing the surgical team to enjoy the comfort and efficiency of general anesthesia without losing accuracy. When higher doses are unavoidable—for example, in very anxious patients—the burst index can serve as a backup marker, helping to identify the lower boundary of the target and warn that recordings may be distorted by heavy sedation. Combining this information with careful imaging and test stimulation may prevent electrodes from being placed too deep.
What this means for people facing DBS
For patients considering DBS, the study offers reassurance and nuance. It shows that surgery under general anesthesia with propofol is feasible and can still provide high-quality guidance from brain recordings, as long as drug levels are carefully controlled. At moderate doses, the brain’s electrical landmarks remain readable, and electrodes can be steered toward the same sweet spot that benefits patients in awake procedures. At very high doses, however, those landmarks fade and the risk of placing electrodes slightly too deep increases. Recognizing this, anesthesiologists and neurosurgeons can adjust propofol dosing and pay special attention to burst-like firing patterns to keep DBS surgery both comfortable and precisely targeted.
Citation: Issabekov, G., Al-Fatly, B., Mousavi, M. et al. The graded effect of propofol in electrophysiology-guided navigation during deep brain stimulation surgery. npj Parkinsons Dis. 12, 64 (2026). https://doi.org/10.1038/s41531-025-01243-1
Keywords: deep brain stimulation, Parkinson’s disease, propofol anesthesia, neural recordings, brain surgery navigation