Clear Sky Science · en
Effect of iron and manganese on the corrosion resistance of contaminated secondary Al-Si-Mg cast alloys
Stronger Cars from Greener Metal
As industries race to cut carbon emissions, recycled aluminum is becoming a key ingredient in lighter cars, trains, and planes. But re‑melting scrap brings along unwanted chemical hitchhikers that can quietly eat away at metal parts over years of service. This study explores how two common impurities, iron and manganese, shape the long‑term corrosion of recycled Al‑Si‑Mg casting alloys, and shows how a small adjustment in their balance can turn vulnerable “dirty” aluminum into a durable, sustainable material.

Why Scrap Aluminum Misbehaves
Recycled aluminum saves up to 90% of the energy needed for primary metal, but it is much harder to clean. Elements like iron, manganese, copper, and magnesium remain in the melt and combine into tiny hard particles within the metal. These particles, known as intermetallics, can act like microscopic batteries when the alloy meets salty water: some regions behave as tiny cathodes, others as anodes, and the resulting currents drive local dissolution, pits, and cracks. Among these particles, thin plate‑shaped compounds rich in iron are especially harmful, accelerating localized corrosion in everyday environments such as road salt spray and marine air.
Designing Three Test Alloys
The researchers cast three versions of a popular automotive alloy, AlSi7Mg0.3, by varying iron and manganese. Alloy A had relatively low iron and little manganese; Alloy B had high iron but still little manganese; Alloy C kept the same high iron as Alloy B but added more manganese, raising the Mn/Fe ratio. Microscopy showed that Alloy B, with high iron and low manganese, formed many long, plate‑like iron‑rich particles. Alloy C, by contrast, transformed most of these plates into more compact, twisted “Chinese‑script” particles that mixed iron and manganese. At the same time, the overall grain pattern and silicon network in the aluminum became finer and more uniform, a change known to influence how corrosion spreads.
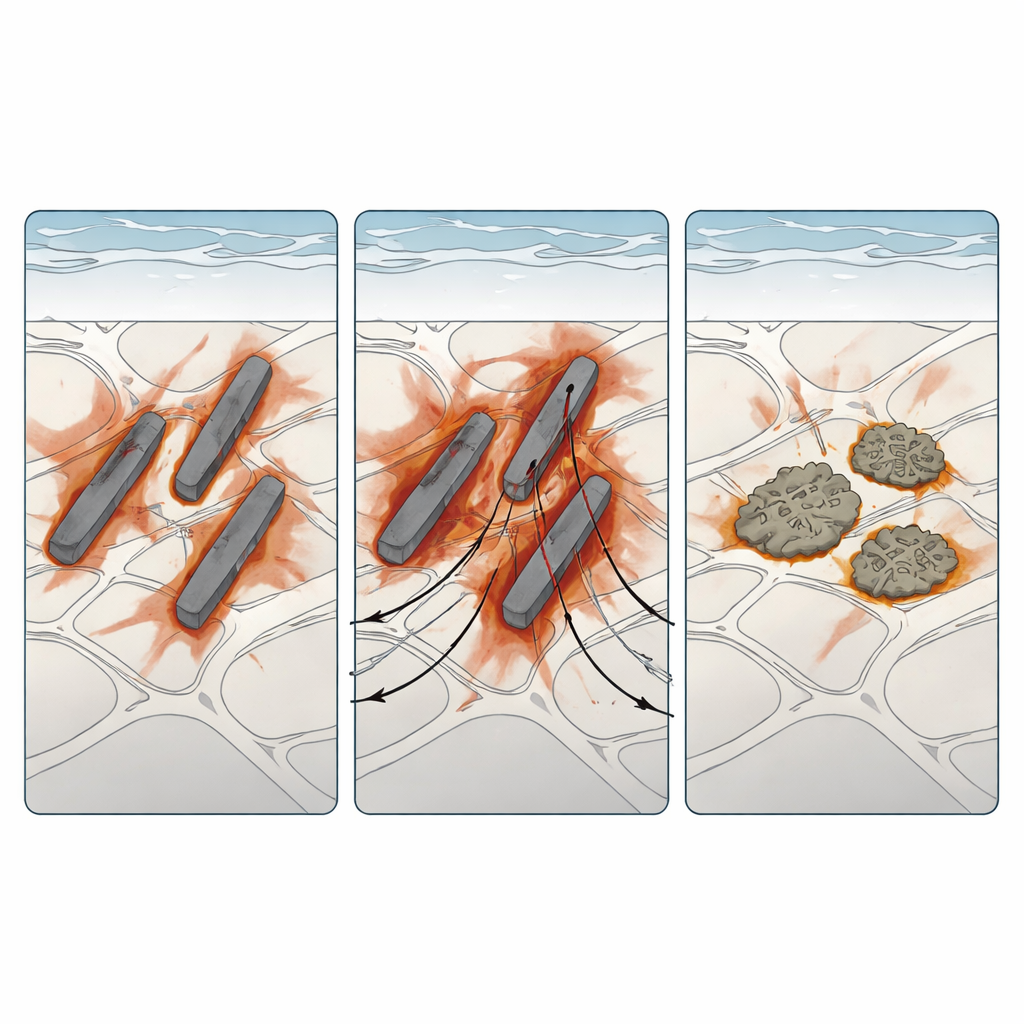
Watching Metal Rot in Salty Conditions
To probe how these microstructures behaved in corrosive environments, the team used electrochemical tests in salt solutions, long salt‑spray exposures that mimic thin atmospheric moisture films, and high‑resolution imaging of attacked cross‑sections. When submerged in a standard sodium chloride solution, all three alloys showed similar average electrochemical signatures, meaning that bulk tests alone could not distinguish them. Microscopy told a different story: corrosion preferentially chewed along the fine Al‑Si regions and around intermetallic particles, with deeper pits forming near the iron‑rich plates. Under salt‑spray, Alloy B degraded the fastest, with widespread dark corrosion products and deep trenches around plate‑like particles. Alloy C, despite having the same iron level, corroded more slowly; its manganese‑rich “Chinese‑script” particles largely kept their shape, with only limited edge attack and shallower surrounding damage.
Simulating Microscopic Corrosion Pathways
Experiments were paired with computer simulations that modeled micro‑galvanic corrosion at the scale of individual phases. Using a finite‑element approach, the authors represented the alloy as a mixture of aluminum matrix and connected Al‑Si regions in a thin salt film. They fed in measured electrochemical behavior for each phase and tracked how current concentrated and the corrosion front moved over time. The model reproduced what the microscopes saw: currents focused along the Al‑Si network, driving selective dissolution there while leaving the primary aluminum relatively untouched, a classic intergranular attack pattern. Larger or more continuous impurity particles intensified local currents, explaining why long iron‑rich plates in Alloy B were so damaging compared with the more stable manganese‑modified shapes in Alloy C.
A Practical Recipe for Tougher Recycled Alloys
By combining imaging, electrochemical testing, salt‑spray exposure, and physics‑based simulation, the study pinpoints a sweet spot for balancing iron and manganese in contaminated recycled Al‑Si‑Mg cast alloys. Keeping the Mn/Fe ratio in the range of roughly 0.3 to 0.6 suppresses the formation of highly active plate‑like iron compounds and promotes more benign “Chinese‑script” particles that are less galvanically aggressive and remain mostly intact as corrosion proceeds. For a lay reader, the takeaway is simple: with careful tuning of impurity levels rather than costly purification, foundries can turn mixed scrap into aluminum castings that are both greener and more durable, making lightweight components for cars and other vehicles last longer in harsh, salty environments.
Citation: Li, Q., Gazenbiller, E., Jarren, L.C. et al. Effect of iron and manganese on the corrosion resistance of contaminated secondary Al-Si-Mg cast alloys. npj Mater Degrad 10, 42 (2026). https://doi.org/10.1038/s41529-026-00767-y
Keywords: recycled aluminum, corrosion, impurities, iron and manganese, automotive alloys