Clear Sky Science · en
Surrogate modelling of corrosion inhibition finite element simulations using machine learning
Protecting Planes and Cars from Rust
Modern airplanes, cars and smartphones rely heavily on lightweight aluminum alloys. These metals resist rust better than ordinary steel, but in salty, humid or hot environments they can still corrode, threatening safety and shortening product lifetimes. Engineers use special paint-like coatings filled with protective particles to slow this damage, yet finding the best recipe is slow and expensive. This study shows how combining detailed computer simulations with machine learning can quickly guide the design of smarter, safer anti-corrosion coatings for a widely used aluminum alloy.
Why Traditional Protection Needs an Upgrade
For decades, the gold standard for protecting aluminum in aircraft and other demanding applications was based on compounds containing hexavalent chromium. These chemicals work extremely well but are toxic and increasingly restricted by environmental regulations. Researchers are now turning to “active” coatings that do more than just form a barrier. In these coatings, tiny pigment particles dissolve when a scratch or defect appears, releasing inhibitor ingredients that migrate to the exposed metal and help rebuild a protective layer. Lithium-based compounds have emerged as especially promising because they form a durable shield on aluminum surfaces. The challenge is figuring out which combination of pigment loading, coating thickness and defect geometry will reliably keep corrosion in check without years of trial-and-error testing.
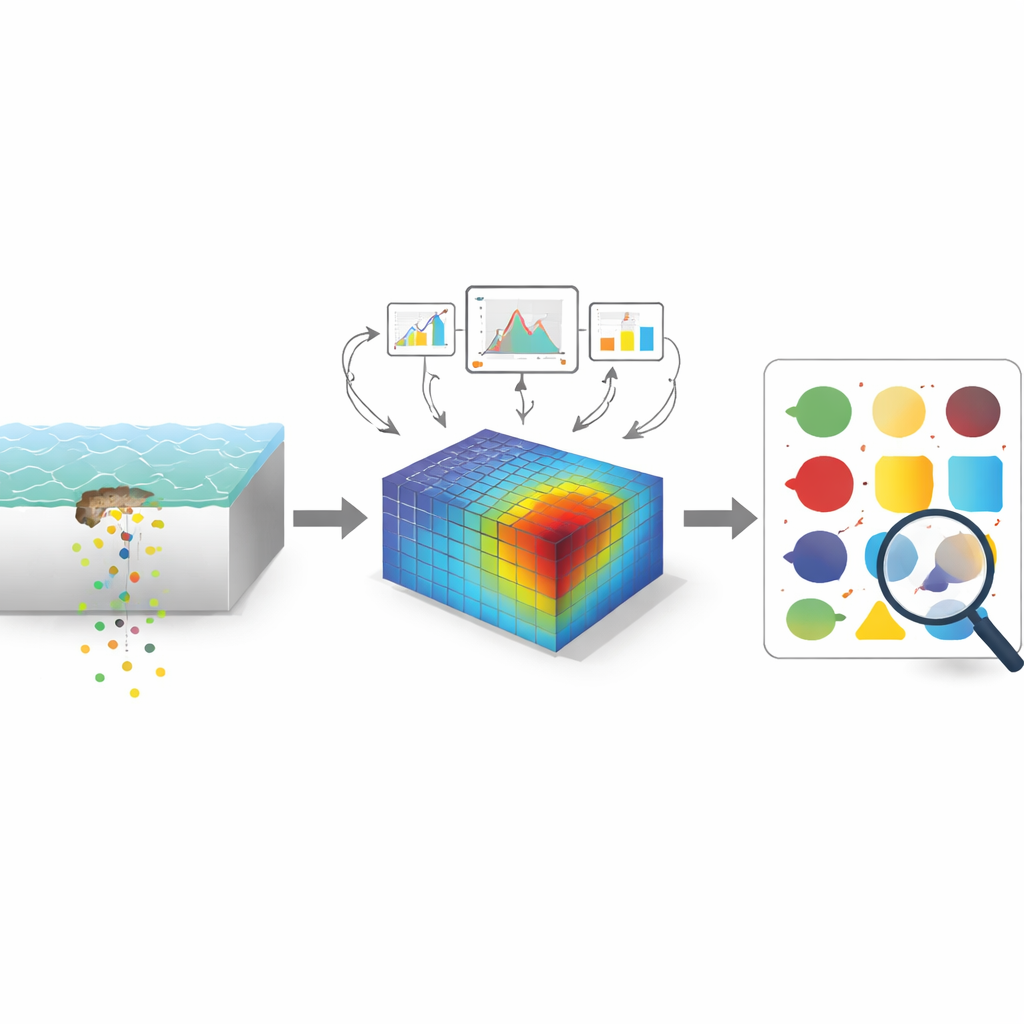
Using Virtual Experiments to Learn Faster
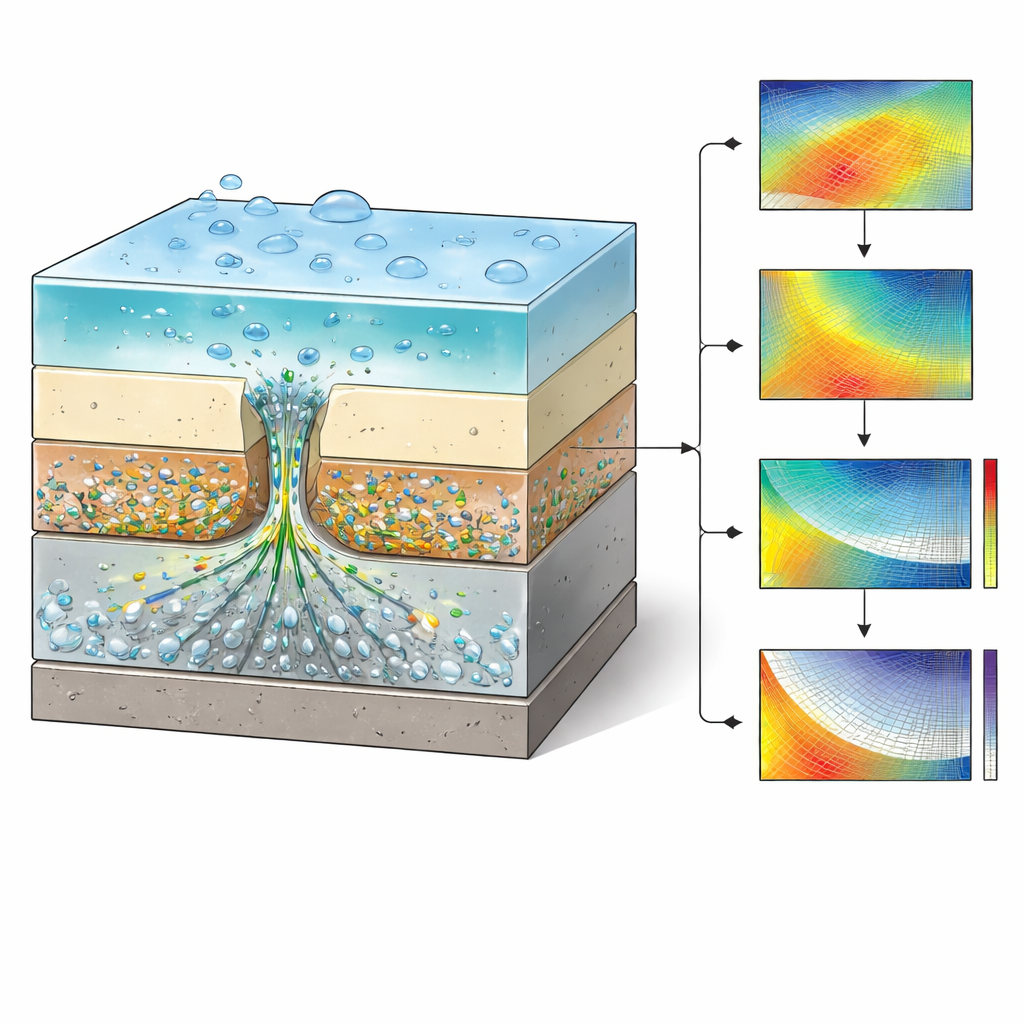
The authors built on an existing two-dimensional finite element model—essentially a detailed physics-based “virtual lab”—that tracks how lithium carbonate particles in a primer layer dissolve, move through tiny water pathways, and influence corrosion at a scratch in the coating. The simulated system mimics a common aerospace alloy, AA2024-T3, covered by a primer loaded with lithium pigment, a protective topcoat, and a thin layer of water on top. By systematically varying five controllable factors—scratch width and depth, primer thickness, water layer thickness, and starting pigment content—the team generated 231 virtual experiments. From each run they extracted two key outcomes at the most vulnerable point on the metal surface: how much inhibitor arrived there and how fast corrosion was proceeding, expressed as current density.
Teaching a Machine to Predict Corrosion
Next, the researchers trained decision-tree-based machine learning models, particularly an algorithm known as XGBoost, to act as a “surrogate” for the heavy physics simulations. The model learned to predict inhibitor concentration and corrosion rate from the five input factors. Careful cross-checking, in which the data were repeatedly split into training and testing portions, showed that the machine learning approach reproduced the virtual experiments with good accuracy, especially for inhibitor concentration. Compared with a simple neural network tested as a baseline, the tree-based methods performed markedly better on this modest-sized dataset. An analysis of which inputs mattered most revealed that the thickness of the water layer on top of the coating and the amount of pigment in the primer were the dominant levers controlling protection, while scratch depth played only a minor role under the studied conditions.
Testing the Model’s Limits and Using It for Design
To see how well their surrogate would perform on new situations, the team created nine fresh simulation cases that spanned the range of coating designs but were not used during training. For most of these “blind test” cases, the machine learning predictions of inhibitor arrival and corrosion rate agreed well with the full physics model, though accuracy declined at the edges of the explored design space, where fewer examples were available for learning. Finally, the authors used the trained model as a rapid design tool: they scanned through different pigment levels and primer thicknesses for a typical defect and identified where the inhibitor concentration would exceed a known threshold needed to suppress corrosion, and where the associated corrosion current began to drop sharply. This showed, for example, that thicker primers or higher pigment loadings can push the system into a safer operating regime.
What This Means for Real-World Materials
In simple terms, this work demonstrates that a machine can learn the essential lessons of many complex corrosion simulations and then provide near-instant guidance on how to tune a coating recipe. Instead of running hundreds of time-consuming computer models or laboratory tests for every new design, engineers can use such surrogate models to narrow down promising combinations of pigment content, coating thickness and expected service conditions. While the approach still inherits any simplifications made in the underlying physics model and should not be used far outside the trained range, it offers a powerful shortcut. Ultimately, this kind of digital toolkit could help researchers replace hazardous chemicals and bring safer, longer-lasting protective coatings for aluminum alloys to market more quickly.
Citation: Sahlmann, L., Abdelrahman, N., Meeusen, M. et al. Surrogate modelling of corrosion inhibition finite element simulations using machine learning. npj Mater Degrad 10, 38 (2026). https://doi.org/10.1038/s41529-026-00760-5
Keywords: corrosion protection, aluminum alloys, protective coatings, machine learning, finite element modeling