Clear Sky Science · en
Achieving a corrosion-resistant Mg-Ca lean alloy by solidification control to sequester parts-per-million-level Fe impurity
Why Protecting Light Metals Matters
Magnesium is one of the lightest structural metals we have, making it attractive for cars, planes, portable electronics, and even medical implants. But there is a catch: magnesium dissolves in salty, wet environments far more quickly than common metals like steel or aluminum. This study explores a clever way to make a very simple magnesium–calcium metal blend resist corrosion better than even ultra‑pure magnesium, not by removing impurities, but by trapping them in the right kind of microscopic cages during solidification.
A Tiny Impurity with a Big Impact
Even when magnesium is produced to high purity standards, it still contains trace amounts of iron—only a few parts per million. That sounds negligible, but it is enough to create tiny iron‑rich particles that act like miniature batteries when the metal is placed in salt water. These particles draw electrons from the surrounding magnesium, speeding up metal loss and also boosting the production of hydrogen gas bubbles. Conventional wisdom has been to remove iron as much as possible, or to buy ultra‑high‑purity magnesium, which is expensive and hard to use in industry.
Using Calcium to Build Microscopic Cages
Earlier work showed that adding a pinch of calcium to magnesium (around one‑tenth of a percent by weight) dramatically slows corrosion, because new microscopic compounds containing calcium, magnesium, and silicon form inside the metal. In this study, the researchers focused on a specific magnesium–0.1% calcium blend and asked a deeper question: how does the speed at which the melted alloy is cooled and solidified affect where the iron ends up, and therefore how fast the alloy corrodes? To answer this, they cast the same alloy using four different cooling rates, from very slow to very fast, and then examined the resulting microstructures in detail using electron microscopes and mapping techniques.
Slow Cooling and Hidden Iron
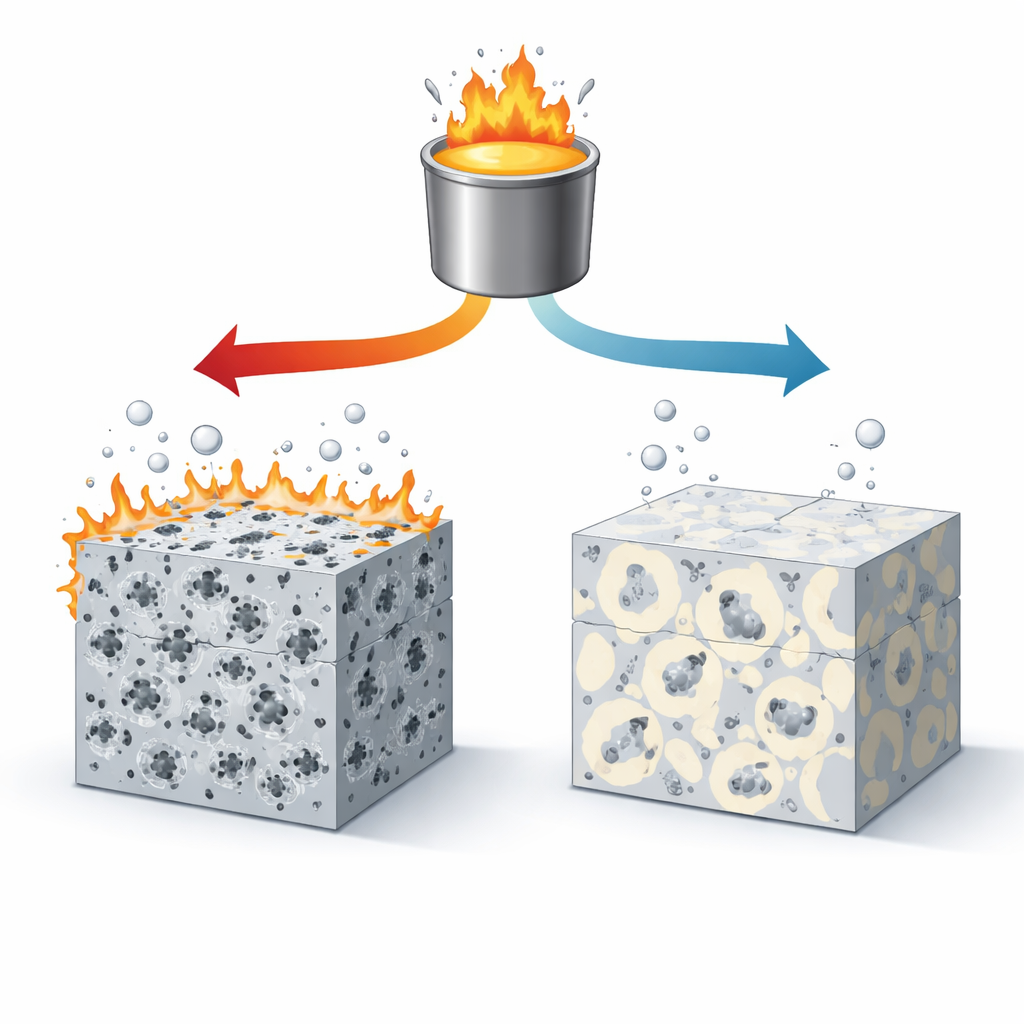
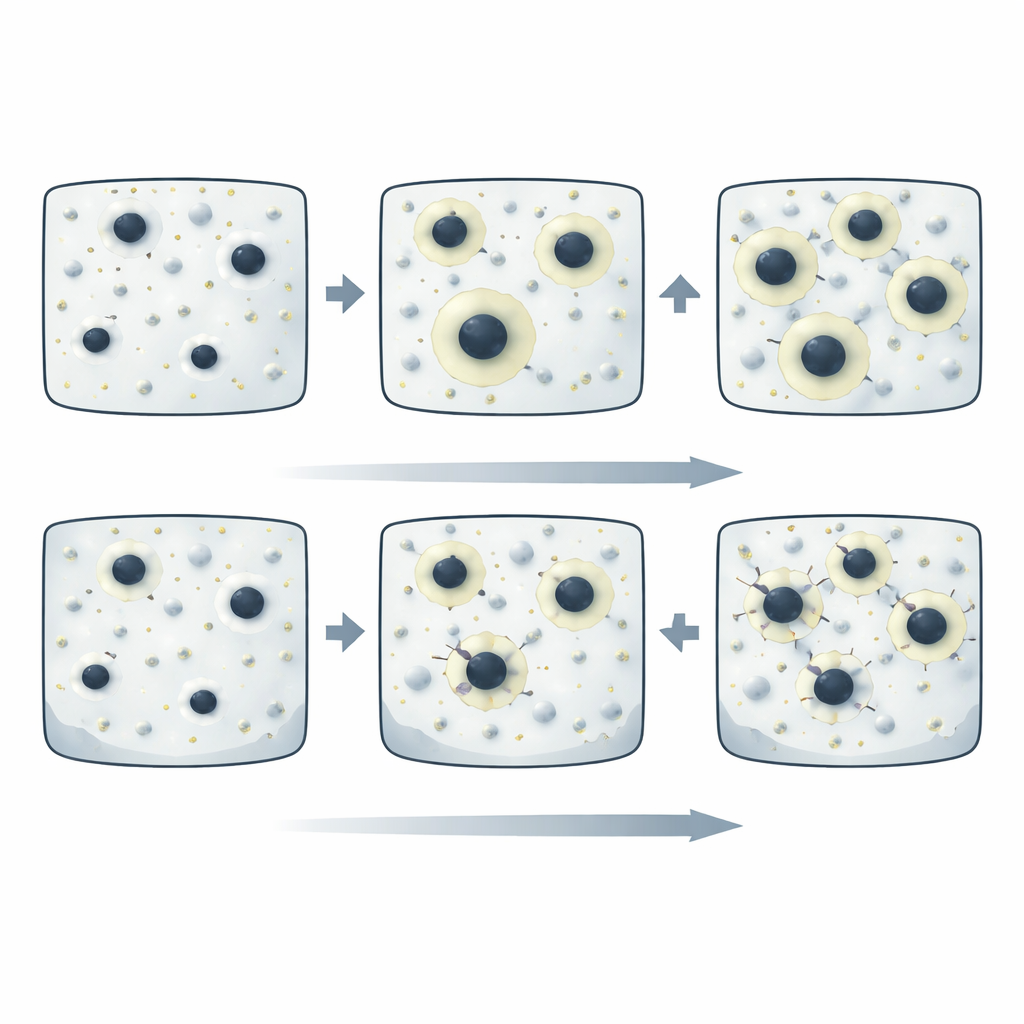
When the alloy was cooled slowly, the team found relatively large particles of the calcium–magnesium–silicon compound spread through the metal. Crucially, many of the iron‑rich particles were completely enclosed inside these larger particles, like seeds trapped in fruit. This encapsulation meant the iron had little direct contact with the surrounding magnesium. In corrosion tests using salty water similar to seawater, these slowly cooled samples produced extremely little hydrogen gas and lost metal at rates thousands of times lower than ordinary high‑purity magnesium. The corrosion was gentle and fairly uniform, with only shallow pits and a protective surface film that became more resistant over time.
Fast Cooling and Exposed Trouble Spots
As the same alloy was cooled faster and faster, the calcium‑rich compounds became smaller and more finely divided. They no longer grew large enough to wrap themselves around many of the iron‑rich particles. Microscopy revealed numerous iron‑rich specks sitting in direct contact with the magnesium, or only partially covered. In saltwater exposure, these uncovered spots became highly active sites where corrosion started quickly, digging deep cavities and filament‑like attack paths across the surface. Hydrogen gas was generated much more rapidly, and electrochemical measurements showed stronger cathodic activity and weaker, less protective surface films.
How Cooling Control Beats Ultra‑Pure Metal
The key insight from this work is that corrosion behavior is dictated less by how much iron is present and more by how that iron is arranged inside the metal. With a small calcium addition and sufficiently slow cooling—slower than about 5 kelvins per second—the metal’s internal structure naturally cages iron inside benign compounds. These cages block the tiny electrochemical “short circuits” that would otherwise cause rapid attack. Under these conditions, the simple magnesium–calcium alloy outperforms even ultra‑high‑purity magnesium in harsh salt solutions, while starting from cheaper, commercially pure feedstock.
What This Means for Real‑World Uses
For engineers and manufacturers, the study offers a practical recipe: rather than relying solely on expensive, ultra‑clean raw magnesium, they can tailor both alloy composition and casting conditions to disarm harmful impurities. By adding a trace of calcium and using casting processes that keep cooling rates low enough to form encapsulating particles, it is possible to produce lightweight magnesium components that last far longer in corrosive environments. This strategy could benefit everything from automotive parts and energy‑storage anodes to biodegradable medical devices, where controlled and predictable corrosion is essential.
Citation: Qi, Y., Deng, M., Rong, J. et al. Achieving a corrosion-resistant Mg-Ca lean alloy by solidification control to sequester parts-per-million-level Fe impurity. npj Mater Degrad 10, 41 (2026). https://doi.org/10.1038/s41529-026-00755-2
Keywords: magnesium alloys, corrosion resistance, microalloying, solidification cooling rate, lightweight materials