Clear Sky Science · en
Pitting-corrosion inhibition in stainless steel by NaNO3: mechanistic insights on sulfide dissolution, depassivation, and active dissolution
Why tiny rust spots matter
From bridges and skyscrapers to cars and chemical plants, stainless steel is chosen because it usually shrugs off rust. Yet under the right conditions, it can suddenly develop tiny, deep holes called pits that can grow into dangerous cracks. This study asks a practical question for industry: can a common, relatively safe chemical—sodium nitrate—stop those pits from ever getting started, and if so, how? The answer could help keep critical infrastructure safer for longer while using additives that are affordable and less harmful to the environment. 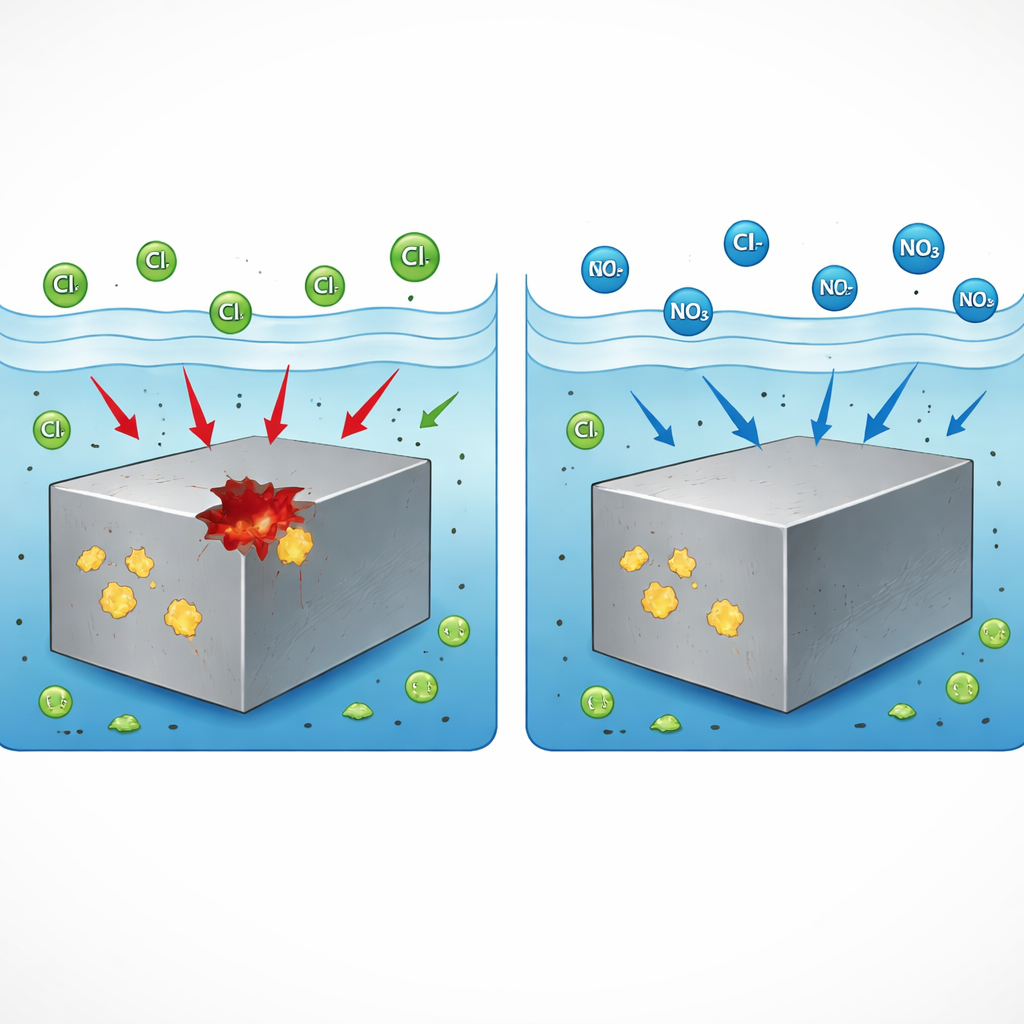
Hidden weak points inside stainless steel
Even the best stainless steels are not perfectly uniform. Scattered inside are microscopic particles rich in sulfur, known as sulfide inclusions. Two important kinds are based on manganese (MnS) and calcium (CaS). These inclusions behave like built‑in weak spots where pits preferentially start when chloride‑containing water, such as salty spray or cooling water, touches the metal. Earlier work showed that pit formation follows a consistent script: the sulfide particle begins to dissolve, the protective film on the nearby steel breaks down, and then the surrounding metal starts dissolving rapidly, digging a pit. The present study compares two commercial Type 304 stainless steels—one dominated by MnS inclusions and one by CaS‑based inclusions—to understand whether nitrate can interrupt this script at either or both types of weak spots.
Testing common additives in salty water
The researchers immersed the steels in a simple salt solution and slowly forced the metal toward more corrosive conditions while watching when stable pits formed. They compared three additives at realistic concentrations: ammonium, nitrite, and nitrate, all as sodium or ammonium salts. Only nitrate had a striking effect. In both the MnS‑rich and CaS‑rich steels, adding a modest amount of sodium nitrate completely prevented stable pits from forming over the tested range, while the other two additives offered no improvement. Microscopy confirmed that, without nitrate, pits indeed started at sulfide inclusions, whereas with nitrate, those inclusions no longer triggered damaging holes. This showed that nitrate is a broadly effective pit inhibitor for different sulfide types, not just a special case. 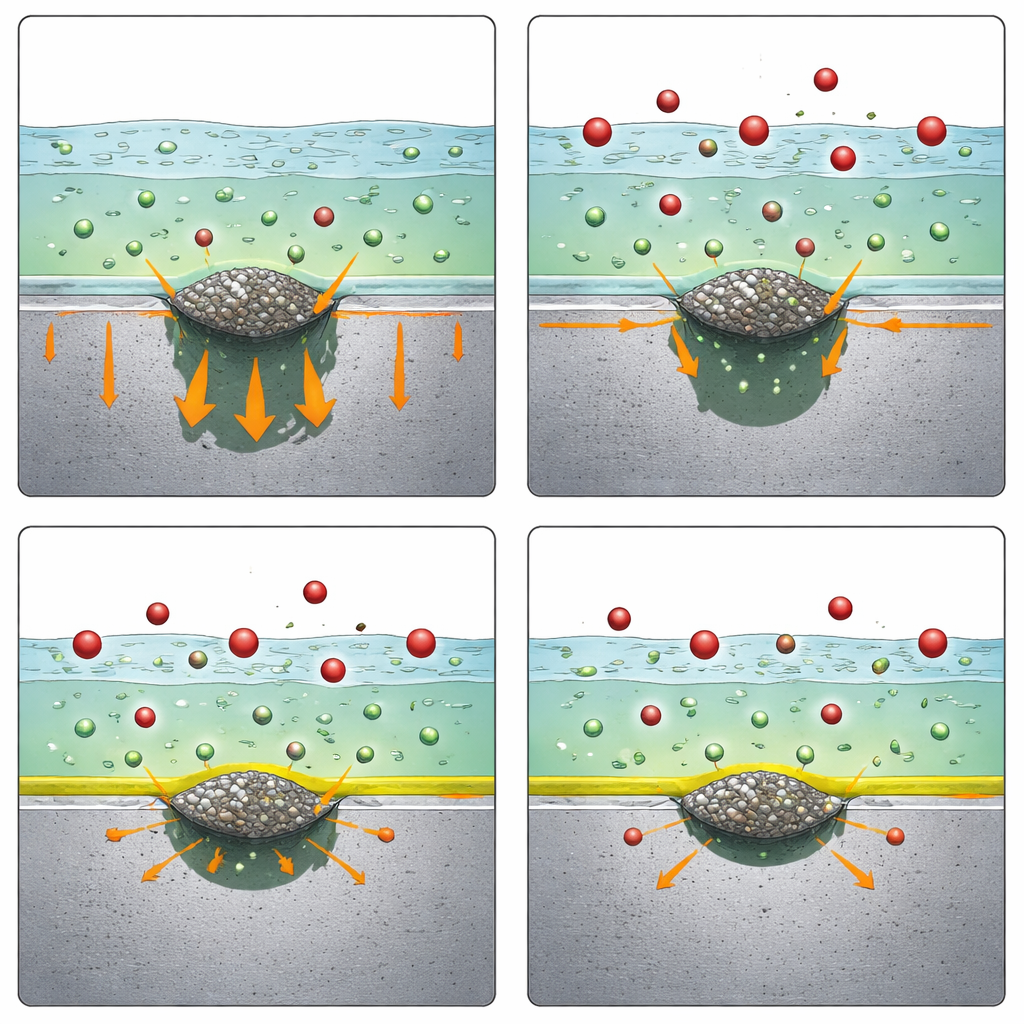
Looking closely at what nitrate does—and does not—change
To pinpoint how nitrate works, the team zoomed in on the early stages of pit formation around single MnS particles using tiny electrodes and high‑resolution imaging. They observed that nitrate did not stop the MnS particles themselves from dissolving, nor did it change the small trenches that form where the inclusion meets the surrounding steel. Nitrate also did not shift the acidity level at which the steel’s protective film fails in chloride solution. All of this indicated that the first steps of pit initiation—weakening and disruption around the inclusion—still proceed much the same in the presence of nitrate.
Slowing the final rush of metal loss
The crucial difference emerged when the researchers recreated the harsh environment inside an already‑formed pit: very acidic, chloride‑rich conditions. Using strong acid solutions that mimic the chemistry deep in pits, they found that the steel normally shows two distinct surges of rapid metal dissolution as the voltage is increased. Adding nitrate consistently reduced the first surge in dissolution, both in plain acid and in acid that also contained sulfur species similar to those released from dissolving MnS. Surface observations showed uniform attack but at a clearly lower rate. Additional tests, varying acidity and chloride levels, suggested that nitrate’s effect could not be explained simply by diluting acidity, displacing chloride, or stabilizing salt crusts. Instead, the results pointed toward nitrate helping chromium within the alloy form a stabilizing surface layer, which in turn slows the early, most critical phase of active metal loss.
What this means for real‑world steels
Put simply, nitrate does not prevent the first chemical nibbles at tiny sulfide particles inside stainless steel, but it does slow the final, runaway phase where a harmless imperfection turns into a dangerous pit. By damping that burst of metal loss in acidic, salty micro‑environments, sodium nitrate keeps the steel closer to a passive, self‑protecting state, even when sulfide inclusions and chloride ions are present. Because nitrate is relatively inexpensive, less toxic than many alternatives, and already used in industrial water systems, understanding this mechanism strengthens the case for its careful use as a corrosion‑control additive to extend the service life of stainless steel structures and equipment.
Citation: Amatsuka, S., Nishimoto, M. & Muto, I. Pitting-corrosion inhibition in stainless steel by NaNO3: mechanistic insights on sulfide dissolution, depassivation, and active dissolution. npj Mater Degrad 10, 40 (2026). https://doi.org/10.1038/s41529-026-00753-4
Keywords: stainless steel corrosion, pitting inhibition, sodium nitrate, sulfide inclusions, chloride solutions