Clear Sky Science · en
Early-stage uranium-hydrogen corrosion kinetics and mechanism
Why this hidden metal damage matters
Modern energy technologies—from nuclear fuel to hydrogen storage and future fusion reactors—depend on metals that quietly endure harsh environments for years. One subtle threat is hydrogen, a tiny atom that can slip into metals and eventually turn them brittle or powdery. This paper peels back the curtain on how that damage starts in uranium, a key nuclear material, by watching the earliest moments of attack with a powerful optical microscope that can see height changes on the scale of billionths of a meter.
Watching metal change in real time
The researchers set out to answer a deceptively simple question: when uranium first meets hydrogen gas, what exactly happens at the surface, and when? For decades, scientists have mainly relied on pressure gauges and weight measurements to track how much hydrogen a sample absorbs overall. Those tools work well for later stages of damage, but they are effectively blind to the very first tiny defects that form. In this study, the team instead used white-light interferometry—an optical profilometry technique—to scan the metal surface repeatedly while it sat in hydrogen at a modest 50 °C and a fixed gas pressure. This approach let them build a time-lapse 3D map of the surface, capturing subtle bumps and pits as they emerged and grew.
The quiet wait before damage appears
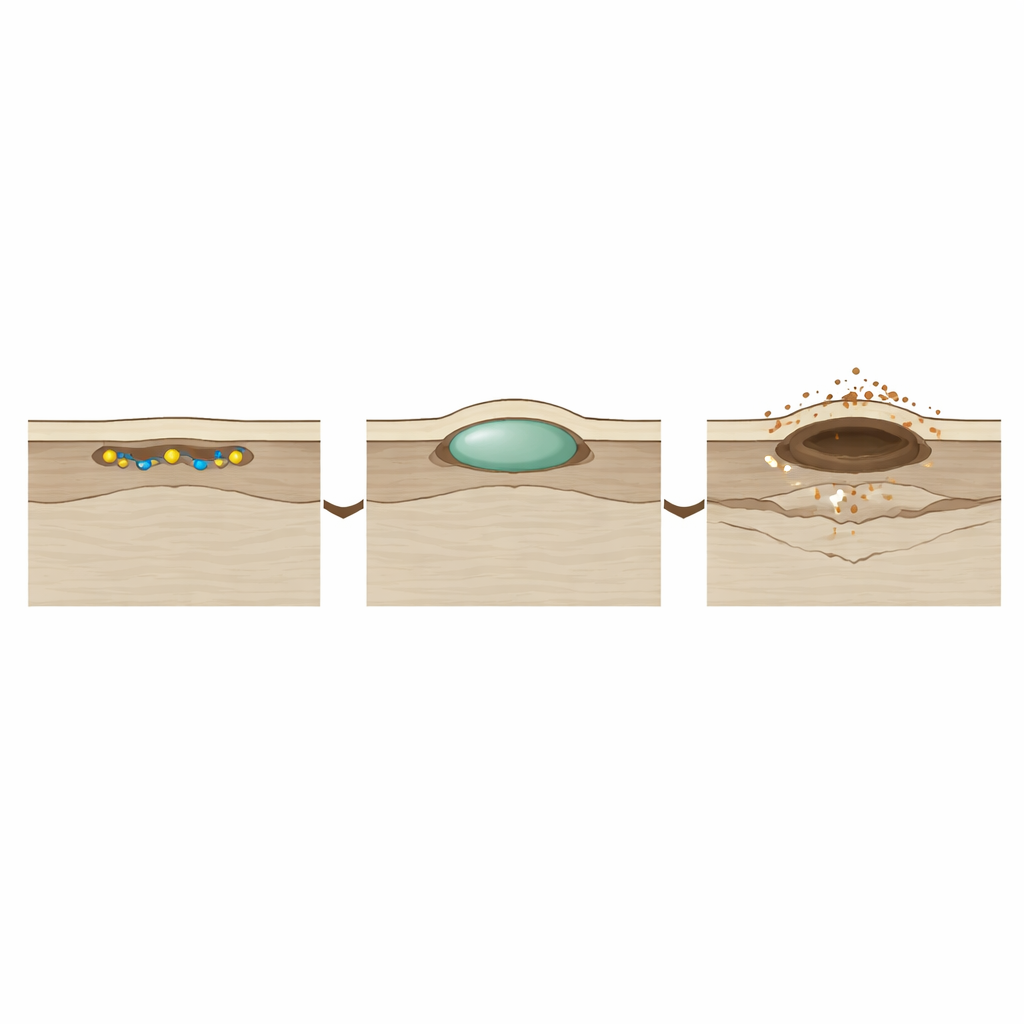
One of the most striking findings is that “nothing happens” for a surprisingly long time. After hydrogen is introduced, the uranium surface looks unchanged for about an hour. During this induction period, hydrogen is actually busy: atoms stick to the surface, work their way through a thin oxide film, and dissolve into the metal beneath. Only when enough hydrogen has built up locally—beyond what the metal can comfortably hold—does a tiny subsurface pocket of uranium hydride form, pushing the surface upward into a microscopic blister. The very first such blister in this experiment did not form at obvious defects like casting pores, hinting that subtle variations in surface oxide and impurities play a larger role than previously assumed.
From blisters to bursts and powder
Once the first blister appears, the story speeds up. The team tracked its height, width, and volume over time and saw rapid growth after the induction period. The blister initially remains intact, a smooth dome sitting just under the surface layer. But as the hydride pocket grows, it builds internal pressure against the overlying metal. When it reaches a critical size—about 40 micrometers across, roughly half the width of a human hair—the surface cracks and “spalls,” ejecting a burst of uranium hydride powder. At this moment, the surface profile suddenly becomes discontinuous, and the blister transforms into an open pit. After spallation, growth at that site becomes more linear and steady, and the damaged region can expand and merge with neighboring sites, carving out larger divots.
Measuring the pace of damage
Because the interferometry scans provide precise diameters for each growing site, the researchers could calculate how fast the damage front moves sideways along the surface. For the conditions tested, the advancing edge of a hydride site after spallation traveled at about 0.91 micrometers per minute. They repeated similar measurements at other temperatures and compared their results with classic hydrogen–uranium data obtained decades earlier from pressure-based experiments. Remarkably, the new surface-based rates lined up well with those older bulk measurements, lending strong support to both the interferometry method and existing mathematical models of uranium hydriding. By the end of just over four hours, nearly 43 percent of the observed surface area had transformed into hydride-related damage.
Inside the hidden pockets of change
To understand what these blisters and pits look like beneath the surface, the team used advanced electron microscopes and focused ion beams to slice through individual damage sites and image them in 3D. They found that early hydride pockets form compact, flattened (oblate) regions just under the surface, closely following the metal–hydride boundary. After spallation, the loss of the top layer reduces confinement, and the hydride beneath can fracture and form layered, more open structures that accelerate further reaction. X-ray diffraction of the collected powder showed that two different crystal forms of uranium hydride are present, with slightly different densities. This suggests that which form appears where may influence how fast individual sites grow and how severe the damage becomes.
What this work tells us about safety
For non-specialists, the key message is that uranium does not crumble under hydrogen all at once; it passes through a long “silent” stage before visible damage, followed by rapid growth once tiny subsurface pockets reach a critical size and burst. By directly watching this process unfold with ultra-precise optical mapping, the authors provide the first detailed, quantitative view of how and how fast these early defects form, grow, and merge. Their results validate modern corrosion models and establish white-light interferometry as a powerful tool for predicting and eventually managing hydrogen-driven damage in nuclear materials and related technologies.
Citation: Shittu, J., Siekhaus, W., Sun, TC. et al. Early-stage uranium-hydrogen corrosion kinetics and mechanism. npj Mater Degrad 10, 35 (2026). https://doi.org/10.1038/s41529-026-00751-6
Keywords: uranium corrosion, hydrogen embrittlement, metal hydrides, surface profiling, nuclear materials