Clear Sky Science · en
Grinding-induced degradation in the pitting corrosion resistance of stainless steel: insights into passive film and MnS
Why everyday metal finishes matter
From kitchen sinks and elevators to chemical plants and bridges, stainless steel is trusted because it usually shrugs off rust. Yet many of these parts are ground or polished before use, and a seemingly simple choice—fine versus coarse grinding—can quietly shorten their service life. This study looks inside the surface of a common stainless steel to reveal how heavy grinding changes tiny features in the metal and makes it more likely to develop dangerous pin‑like corrosion pits in salty environments.
Tiny weak spots hiding in clean metal
Stainless steel resists corrosion because it naturally forms an ultra‑thin protective film of oxide on its surface. However, the metal is not perfectly uniform. It contains microscopic particles rich in manganese and sulfur, known as MnS inclusions. Previous work has shown that these inclusions are frequent starting points for pitting corrosion in salt solutions. The authors studied a standard Type 304 stainless steel with either low or high sulfur content, and prepared surfaces with three finishes: a mirror‑polished surface, a moderately ground surface using fine abrasive paper, and a heavily ground surface using coarse paper that leaves deep grooves.
How surface finish changes pit resistance
When the samples were exposed to salt solutions and their behavior was tracked electrochemically, a clear pattern emerged. Mirror‑polished and moderately ground surfaces showed almost the same resistance to pit formation: their protective films held up to similar voltages before breaking down. In contrast, the heavily ground surfaces showed pit formation at noticeably lower voltages, especially in the high‑sulfur steel that contains many MnS particles. Microscopic images confirmed that, in all cases, pits formed at or around MnS inclusions; areas of the steel intentionally prepared without these particles did not pit under the same conditions, even when the surface was ground. This means that MnS inclusions are essential triggers, and grinding mainly changes how vulnerable these triggers become.
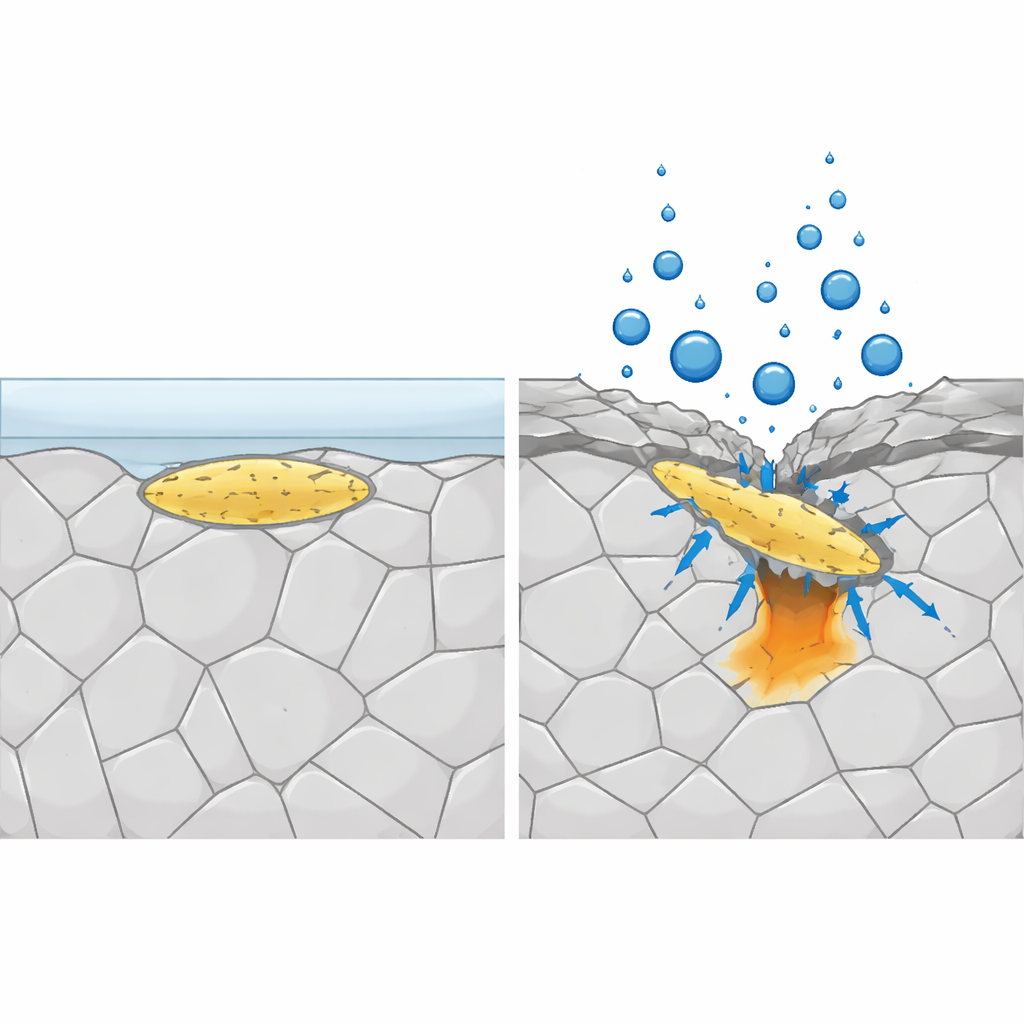
What heavy grinding really does to the surface
At first glance, one might assume that the roughness of a ground surface alone explains its poorer performance. The researchers used advanced tools to test this idea, including scanning probe methods, electron microscopy, and surface chemical analysis. They found that grinding does make the protective film slightly thinner and more uneven, and that scratches respond more actively in corrosive conditions. But the overall chemical makeup of the film, including its beneficial chromium enrichment, changed very little. Instead, the most striking differences appeared in the steel layer just beneath the surface and in the shape of MnS inclusions themselves. Coarse grinding produced a thick, severely deformed layer with dense microstructural defects and caused the elongated MnS particles to be bent, cracked, partially removed, and pressed deeper into the metal. These damaged inclusions often sat at the bottoms of grinding grooves, where micro‑gaps could trap solution.
From hidden damage to growing pits
By isolating single MnS inclusions in tiny test areas, the team watched how pits start and evolve. On lightly finished surfaces, pits tended to nucleate at the edge of an intact inclusion where it meets the steel and then grow in a typical “lacy” fashion. On heavily ground surfaces, pit initiation voltages were lower, and pits were strongly associated with intersections between inclusions and deep grinding marks. Cracks in the inclusions and crevice‑like spaces around embedded fragments appeared to concentrate aggressive species from the solution, such as chloride and sulfur, and to hinder the healing of the protective film. Interestingly, the overall rate at which MnS dissolved did not increase dramatically with grinding, suggesting that the geometry and mechanical damage around inclusions, rather than faster chemical dissolution, are what make pits easier to start.
What this means for real‑world stainless steel
For designers and maintenance engineers, these findings underline that not all “ground” stainless steel surfaces are equal. Moderate grinding that leaves a relatively shallow, even texture can preserve pit resistance close to that of a polished surface. In contrast, aggressive grinding that cuts deeper than the width of MnS particles deforms and buries them, creating microscopic crevices that act as pit launchpads in salty environments. The study shows that the drop in performance cannot be blamed solely on a slightly weaker protective film; it stems mainly from how heavy grinding reshapes the tiny inclusions that are already the Achilles’ heel of stainless steel. Choosing gentler finishing processes and avoiding overly coarse abrasives can therefore help stainless steels live up to their rust‑resistant reputation in service.
Citation: Wang, S., Nishimoto, M. & Muto, I. Grinding-induced degradation in the pitting corrosion resistance of stainless steel: insights into passive film and MnS. npj Mater Degrad 10, 34 (2026). https://doi.org/10.1038/s41529-026-00750-7
Keywords: stainless steel, pitting corrosion, surface grinding, manganese sulfide inclusions, passive film