Clear Sky Science · en
Modeling the uniform corrosion behavior of zinc in salt spray testing
Why this matters for everyday metal parts
From car bodies to power lines, many metal parts are protected by thin zinc coatings that slowly dissolve instead of the underlying steel. Engineers rely heavily on “salt-spray tests” to estimate how long these coatings will last in harsh, salty environments resembling winter roads or sea air. Yet these tests can be hard to interpret and don’t always give consistent, quantitative answers. This paper tackles that problem by building a physics-based computer model that predicts how fast zinc coatings wear away under salt-spray, aiming to turn a qualitative lab test into a more reliable design tool.
How zinc coatings protect metal
Zinc coatings act as sacrificial shields: they corrode first, keeping the steel beneath intact. In salty water, zinc dissolves into charged particles (ions), while oxygen from the air reacts to form a thin, initially patchy layer of corrosion products, mainly zinc hydroxide and zinc oxide. Over time, this layer grows and can partly slow further attack. In real salt-spray chambers, the surface is not submerged in a pool of water. Instead, a thin film of salty moisture constantly forms from sprayed droplets, thickens, and then runs off in cycles. This changing film controls how much oxygen and salt reach the metal and how quickly zinc ions build up, which in turn controls the corrosion rate.
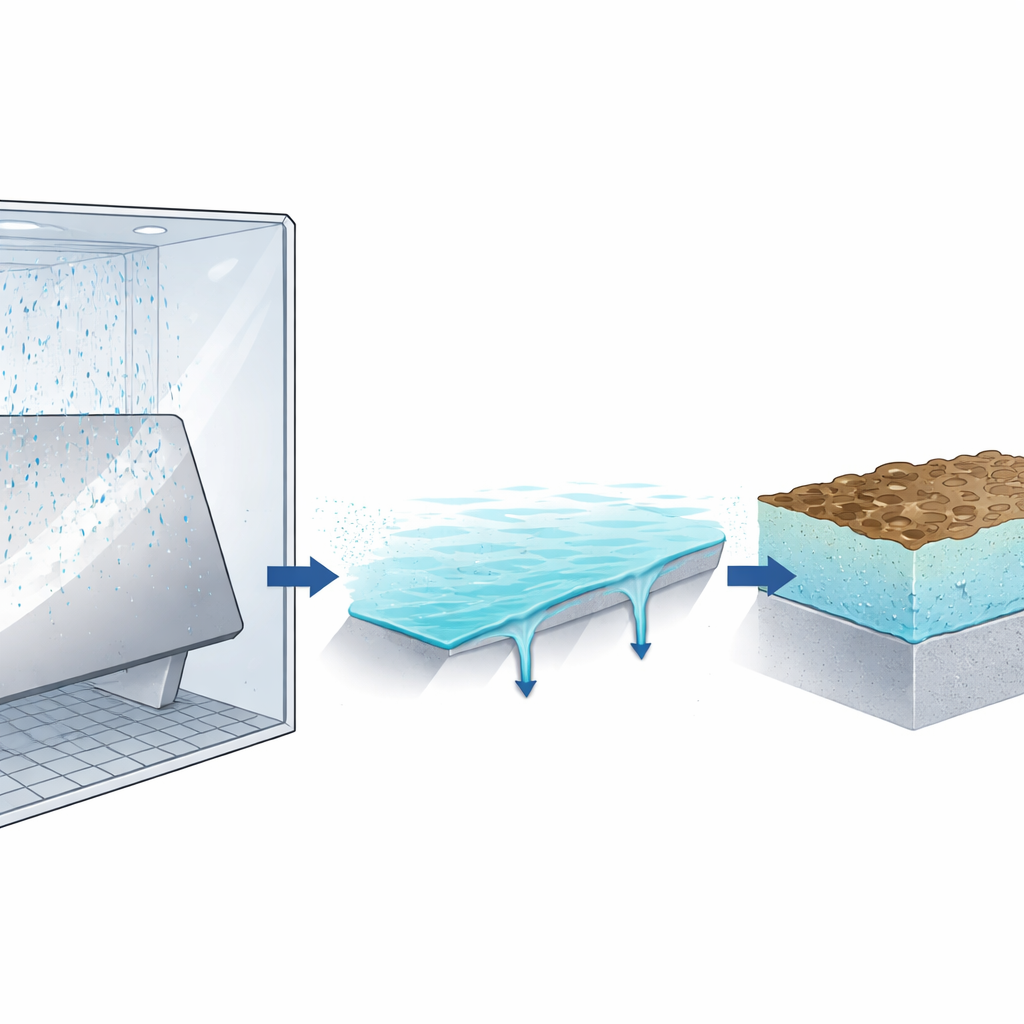
Building a corrosion model from the ground up
The authors developed a numerical model that connects three key pieces: the electrochemical reactions that dissolve zinc, the transport of ions and oxygen through the thin water layer, and the formation of solid corrosion products that create a growing barrier. They describe ion movement with a standard diffusion equation, simplify electrical effects, and treat corrosion as a blend of reaction-controlled and diffusion-controlled processes. A special relation, the Brønsted–Bjerrum equation, adjusts how quickly zinc hydroxide forms when the salt concentration in the water film becomes very high, as it often does in a thin, slowly drained layer. To keep the model realistic but manageable, the authors assume corrosion is uniform across the surface and focus on the zinc layer alone, not yet including later damage to the steel underneath.
Testing the model against real experiments
To tune the model, the team first simulated a simpler case: pure zinc immersed in a dilute salt solution. They adjusted three uncertain quantities—the rate at which zinc hydroxide precipitates, how easily zinc ions move through the film, and how porous the oxide layer is—until the simulations matched published measurements of corrosion depth, oxide thickness, and zinc released into the liquid. This calibration showed, for example, that faster precipitation thickens the oxide layer and slows corrosion by limiting oxygen access. Once calibrated, the same parameters were applied to a much more realistic neutral salt-spray test, mimicking seawater from the Kerch Strait. Here, the model captured an important shift: corrosion starts out governed mainly by surface reactions but, as the oxide layer and ion concentration grow, it becomes limited by how fast species can diffuse through the increasingly clogged layer.
Why the water film’s motion matters
A distinctive feature of salt-spray testing is the restless behavior of the thin water film. Spray droplets gradually thicken the layer until gravity and surface forces cause portions to run off, carrying dissolved zinc with them and briefly thinning the film. The authors included this by letting the film thickness grow at a chosen rate and then periodically reset to a smaller value, based on measured run-off periods and specimen angles. Simulations revealed that higher spray rates and larger tilt angles generally increase corrosion early on, by keeping the surface well supplied with fresh solution. Longer intervals between run-off events give more time for zinc ions to accumulate, which strengthens diffusion barriers and can slow corrosion later. When these film dynamics are included, and the zinc oxide layer is assumed moderately porous, the model reproduces measured corrosion rates from salt-spray experiments typically within about 20 percent.

What the study means for real-world durability
In plain terms, the study shows that how the salty water film grows, concentrates, and drains off a zinc-coated surface is just as important as the chemistry of zinc itself in determining how fast the coating disappears. A water film that is periodically refreshed prevents zinc ions from building up too much and can sustain higher corrosion rates, while a dense, unbroken oxide crust can slow corrosion but may eventually crack or flake away. By capturing these trade-offs in a single, relatively efficient model, the work provides a foundation for predicting the lifetime of zinc-coated parts more quantitatively and for extending the approach to more complex, industry-standard corrosion tests that include drying cycles, changing temperatures, and ultimately the onset of steel corrosion once the zinc is consumed.
Citation: Chen, C., Hofmann, M. & Wallmersperger, T. Modeling the uniform corrosion behavior of zinc in salt spray testing. npj Mater Degrad 10, 37 (2026). https://doi.org/10.1038/s41529-026-00749-0
Keywords: zinc corrosion, salt spray testing, zinc coatings, corrosion modeling, electrolyte film