Clear Sky Science · en
Tracking element-specific dissolution during pitting corrosion: an operando ICP-AES–electrochemical study of the CoCrFeMnNi Cantor alloy
Why Tiny Rust Spots Matter
From bridges and ships to chemical plants and future energy devices, many critical structures depend on metals that must survive harsh, salty, and acidic environments. Sometimes these metals fail not by slowly rusting everywhere at once, but by developing tiny, hidden cavities called pits that can suddenly grow and cause cracks. This study focuses on understanding how such pits start, grow, and heal on a promising new family of strong, corrosion‑resistant metals known as high‑entropy alloys, using a custom setup that can watch, in real time, which ingredients of the metal dissolve into the liquid.
A New Kind of Complex Metal
High‑entropy alloys are metallic “cocktails” made by mixing several elements in nearly equal amounts, rather than relying on one main ingredient like iron in steel. The CoCrFeMnNi “Cantor alloy” is one of the best‑known examples. It is strong, tough, and forms a protective surface film that usually shields it from attack. Yet, in real‑world service—such as in marine or chemical environments rich in chloride ions from salts—even this alloy can suffer localized corrosion. Understanding exactly how each of the five elements (cobalt, chromium, iron, manganese, and nickel) behaves when a pit forms is crucial for designing even better, longer‑lasting materials.
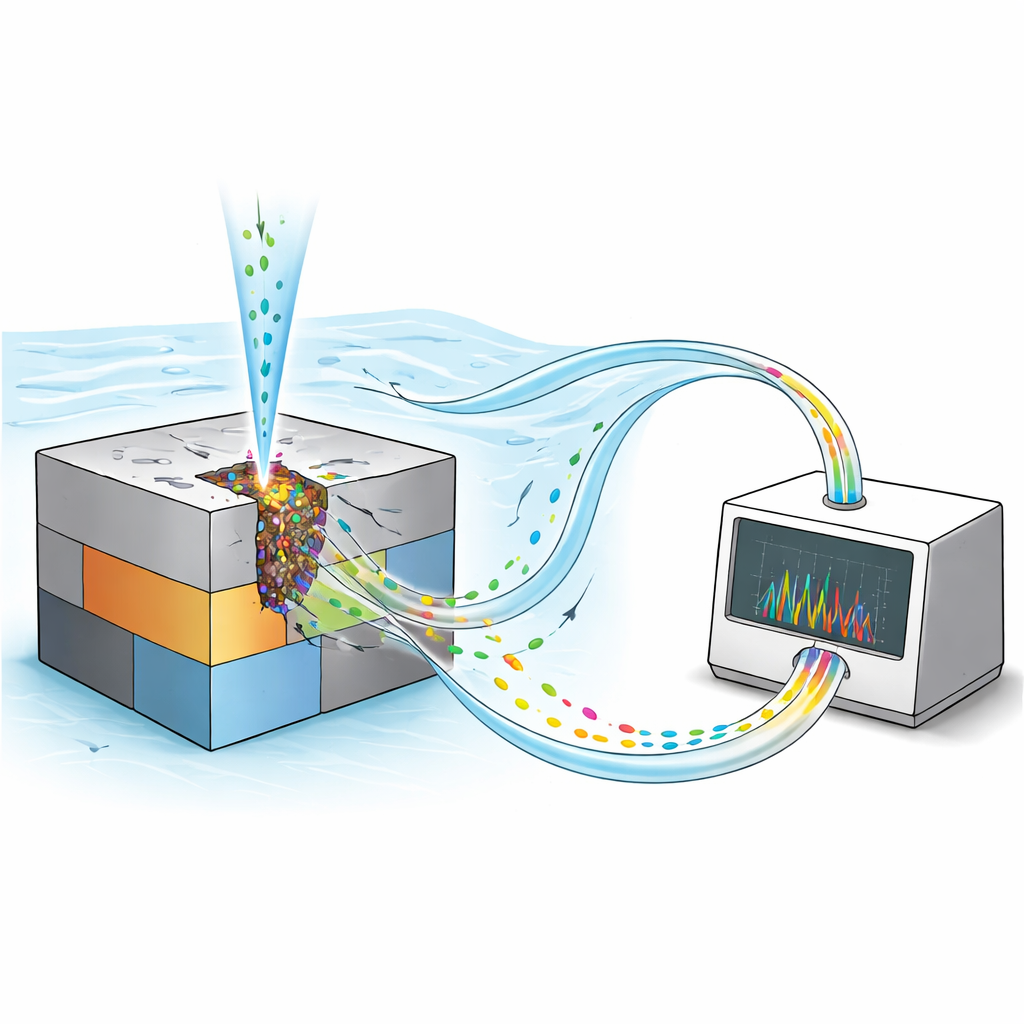
A Microscope for Dissolving Metals
Traditional corrosion experiments can tell us how much current flows when a metal corrodes, but not which element is leaving the surface at each moment. The researchers overcame this by combining two powerful techniques into a single “operando” platform. First, they used a tiny capillary to inject chloride ions onto a very small area of the alloy while holding the voltage constant, ensuring that pitting started in a controlled fashion rather than randomly across the surface. Second, they forced the surrounding acidic solution to flow past the metal and directly into an analytical instrument called an ICP‑AES, which can detect trace amounts of dissolved metals with high sensitivity. By converting these signals into time‑resolved dissolution rates, they could track how fast each element left the alloy during the life of a pit.
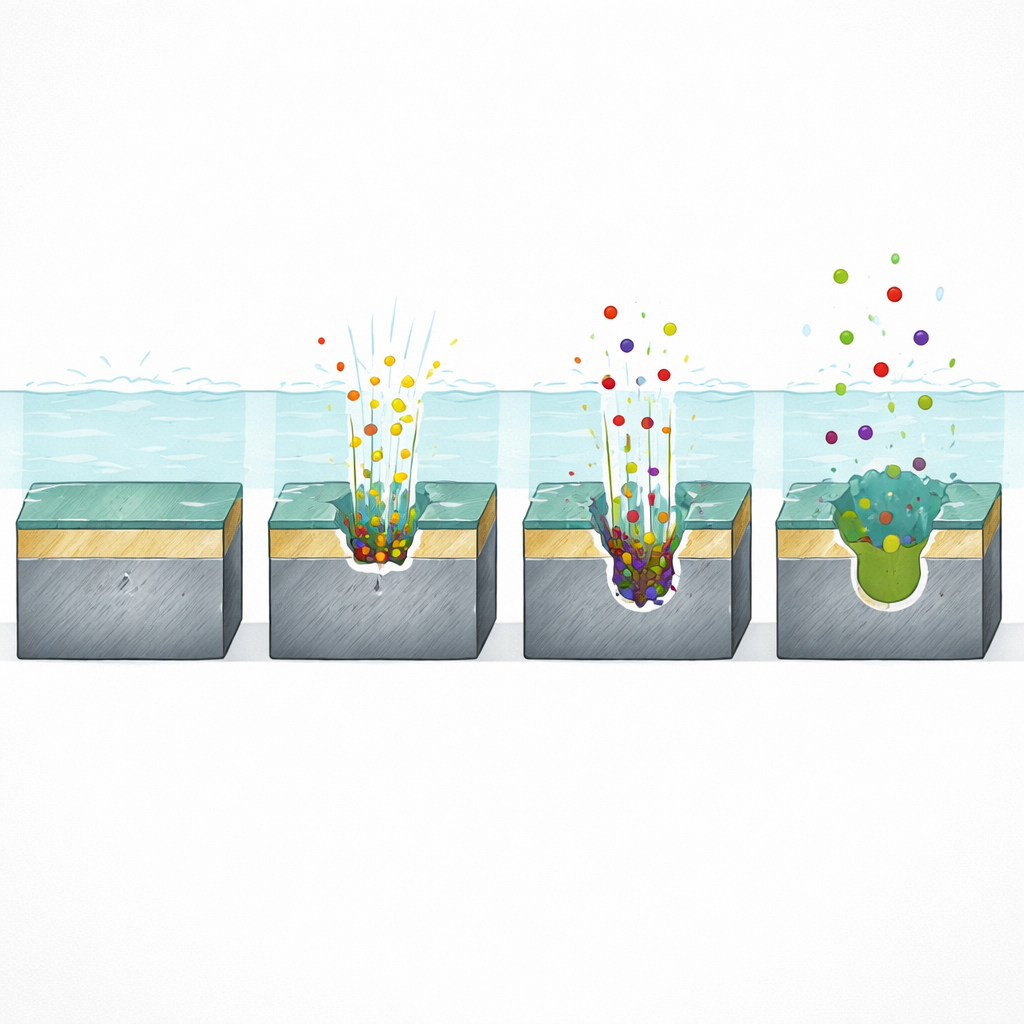
Following a Pit’s Life Story
With this setup, the team identified four clear stages in the pit’s life: incubation, initiation, propagation, and repassivation. During incubation, little happens—the protective film remains intact while chloride builds up locally. At initiation, a brief surge in both current and dissolution reveals that the film breaks and one or more pits suddenly appear. As the pit propagates, current levels off to a quasi‑steady value while the cavity deepens. Finally, in the repassivation stage, after chloride injection stops, the current slowly falls as the pit and surrounding area attempt to rebuild their protective film, even though trapped chloride inside the cavities delays full healing.
Each Ingredient Plays a Different Role
Because the alloy contains five elements in nearly equal proportions, one might expect them to dissolve at the same rate throughout the pit event. Instead, the measurements revealed subtle but important differences. Cobalt and iron contributed slightly more to dissolution right at initiation, suggesting that they are preferentially removed when the protective film first breaks. Chromium, by contrast, dissolved less than the others during active pit growth, meaning it tended to accumulate in the surface film. During repassivation, chromium’s dissolution signal became comparatively stronger, consistent with its central role in building and rebuilding the protective, chromium‑rich oxide that helps the alloy resist further attack. At the same time, the total electrical charge consumed during healing was much larger than expected for a simple, compact film, implying a slow, repeated cycle of oxide formation and partial dissolution inside the pit.
What This Means for Safer Structures
To a non‑specialist, the main message is that the way a metal fails often depends on a delicate, time‑dependent tug‑of‑war between its ingredients and the surrounding environment. This work shows that even within a single tiny pit, different elements take turns leading the action: some leave first, others help rebuild protection. By directly watching which atoms dissolve and when, the new method gives engineers a more detailed recipe for designing high‑entropy alloys that are less prone to dangerous pitting. It also provides rich, quantitative data that can feed computer models and machine‑learning tools aimed at predicting corrosion behavior, ultimately helping us build safer, longer‑lasting infrastructure and devices.
Citation: Hou, Y., Gharbi, O., Xie, C. et al. Tracking element-specific dissolution during pitting corrosion: an operando ICP-AES–electrochemical study of the CoCrFeMnNi Cantor alloy. npj Mater Degrad 10, 33 (2026). https://doi.org/10.1038/s41529-026-00747-2
Keywords: pitting corrosion, high-entropy alloys, localized dissolution, chromium passivation, operando ICP-AES